Industrial Scale-Up of 2,6-Dimethylpyridine-3-Boric Acid via Novel One-Pot Catalysis
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for synthesizing heterocyclic building blocks, particularly pyridine derivatives which serve as critical scaffolds in modern drug discovery. Patent CN112174993A introduces a transformative preparation method for 2,6-dimethylpyridine-3-boric acid, a valuable intermediate for Suzuki-Miyaura cross-coupling reactions. This innovation addresses the longstanding challenges associated with the functionalization of electron-deficient pyridine rings, shifting away from hazardous cryogenic lithiation towards a thermally stable, palladium-catalyzed borylation protocol. By enabling a seamless one-pot conversion from the bromo-precursor to the final boronic acid, this technology offers a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to optimize their manufacturing portfolios. The strategic implementation of this route not only enhances process safety but also aligns with green chemistry principles by reducing solvent usage and eliminating intermediate isolation steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,6-dimethylpyridine-3-boric acid relied heavily on halogen-lithium exchange strategies using reagents such as n-butyllithium at extremely low temperatures, typically around -78°C. This conventional approach presents severe operational bottlenecks for large-scale manufacturing, primarily due to the immense energy costs required to maintain cryogenic conditions over extended reaction periods. Furthermore, organolithium reagents are pyrophoric and highly moisture-sensitive, necessitating specialized equipment and rigorous safety protocols that drastically increase capital expenditure and operational complexity. The sensitivity of the intermediate lithiated species often leads to inconsistent yields and the formation of difficult-to-remove byproducts, complicating downstream purification and negatively impacting the overall cost reduction in API manufacturing. These factors collectively render the traditional lithiation route economically unviable for the production of high-purity pyridine derivatives on a multi-ton scale.
The Novel Approach
In stark contrast, the methodology disclosed in CN112174993A utilizes a Miyaura borylation strategy that operates under significantly milder and more scalable thermal conditions, typically between 80°C and 120°C. This novel approach leverages the synergy between a palladium catalyst, a diboron reagent like pinacol diboride, and a mild base to effect the carbon-boron bond formation efficiently. The true breakthrough lies in the telescoped one-pot design, where the intermediate boronic ester is hydrolyzed in situ without isolation, thereby streamlining the workflow and minimizing material loss. This transition from batch-wise isolation to a continuous flow-like logic within a single vessel represents a paradigm shift in process chemistry, facilitating the commercial scale-up of complex heterocyclic intermediates with unprecedented ease. By removing the need for cryogenic infrastructure, manufacturers can utilize standard stainless steel reactors, significantly lowering the barrier to entry for high-volume production.
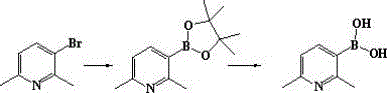
Mechanistic Insights into Palladium-Catalyzed Miyaura Borylation
The core of this synthetic advancement rests on the well-established yet meticulously optimized palladium catalytic cycle, which initiates with the oxidative addition of the aryl bromide to the active Pd(0) species. In the context of 2,6-dimethyl-3-bromopyridine, the steric hindrance provided by the ortho-methyl groups can traditionally impede catalyst approach; however, the use of bulky phosphine ligands or specific catalyst precursors like PdCl2(dppf) effectively overcomes this barrier. Following oxidative addition, the transmetallation step occurs where the boron moiety from the diboron reagent is transferred to the palladium center, a process facilitated by the presence of the base which activates the diboron species. The cycle concludes with reductive elimination, releasing the boronic ester product and regenerating the active catalyst, ensuring high turnover numbers and minimal metal residue in the final product. Understanding these mechanistic nuances is crucial for reducing lead time for high-purity boronic acids as it allows for precise tuning of reaction parameters to maximize efficiency.
Following the borylation, the subsequent hydrolysis step is equally critical for ensuring the purity and stability of the final boronic acid. The patent specifies the direct addition of acid, such as hydrochloric acid, to the reaction mixture to cleave the pinacol ester group. This acidic environment not only drives the hydrolysis equilibrium towards the free boronic acid but also helps in protonating any basic impurities or residual amine bases, keeping them in the aqueous phase during workup. The careful control of pH during the final neutralization with sodium bicarbonate ensures the precipitation of the target product while leaving soluble impurities in the mother liquor. This dual-functionality of the acid treatment—acting as both a hydrolysis agent and a purification aid—exemplifies the elegance of the process design, delivering a solid product with a simplified impurity profile that meets stringent pharmaceutical standards.
How to Synthesize 2,6-Dimethylpyridine-3-Boric Acid Efficiently
The execution of this synthesis requires precise attention to stoichiometry and thermal management to replicate the high yields reported in the patent examples. Operators must ensure the complete dissolution of the palladium catalyst and base prior to heating to prevent localized hot spots that could degrade the catalyst. The reaction progress should be monitored via HPLC or TLC to confirm the complete consumption of the starting bromide before proceeding to the hydrolysis stage, ensuring maximum conversion. Detailed standardized operating procedures regarding the addition rate of acid and the temperature ramping during hydrolysis are essential for consistent batch-to-batch reproducibility. For a comprehensive guide on the exact molar ratios and specific workup protocols, please refer to the technical instructions below.
- Combine 2,6-dimethyl-3-bromopyridine, pinacol diboride, a palladium catalyst, and a base in an organic solvent such as dioxane or toluene.
- Heat the mixture to 80-120°C for 6-12 hours to form the intermediate boronic acid pinacol ester without isolation.
- Add mineral acid directly to the reaction system and maintain heating to hydrolyze the ester, followed by neutralization and filtration to isolate the pure solid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this one-pot synthesis route offers substantial strategic benefits that extend beyond simple yield improvements. The elimination of cryogenic cooling requirements translates directly into reduced utility costs and a lower dependency on specialized, high-maintenance equipment, thereby enhancing the overall asset utilization rate of the manufacturing facility. Furthermore, the use of commercially available and stable reagents like pinacol diboride and common palladium catalysts mitigates supply chain risks associated with hazardous or hard-to-source chemicals. This robustness ensures a more predictable production schedule, allowing supply chain managers to commit to tighter delivery windows with greater confidence. The simplified workup procedure, which avoids multiple extraction and drying steps typical of two-pot processes, significantly reduces solvent consumption and waste generation, aligning with increasingly strict environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The economic impact of switching to this methodology is profound, primarily driven by the removal of energy-intensive cooling steps and the reduction in processing time. By consolidating two distinct reaction steps into a single vessel operation, manufacturers save significantly on labor costs, solvent volumes, and reactor occupancy time. The avoidance of intermediate isolation means there is no yield loss associated with filtration, drying, and re-dissolution of the boronic ester, effectively boosting the overall mass balance of the process. Additionally, the ability to use less expensive bases like potassium carbonate or sodium acetate instead of costly organometallic reagents further drives down the raw material cost per kilogram, creating a highly competitive cost structure for the final product.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the inherent stability of the reagents and the forgiving nature of the reaction conditions. Unlike lithiation chemistry which demands absolute exclusion of moisture and air, this palladium-catalyzed route is more tolerant, reducing the risk of batch failures due to minor environmental fluctuations. The use of standard solvents such as dioxane, toluene, or DMF ensures that raw material sourcing remains flexible and resilient against market volatility. This reliability allows procurement teams to negotiate better terms with vendors and maintain leaner inventory levels without compromising production output, ultimately leading to a more agile and responsive supply network capable of meeting sudden spikes in demand.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial production is straightforward due to the absence of exothermic hazards associated with organolithium additions. The thermal profile of the reaction is manageable with standard jacketed reactors, facilitating a smooth technology transfer to multi-ton scale facilities. From an environmental standpoint, the one-pot nature drastically cuts down on the volume of organic waste generated, as fewer washes and separations are required. The simplified effluent stream makes wastewater treatment more efficient and cost-effective, ensuring compliance with global environmental standards and reducing the carbon footprint of the manufacturing operation, which is increasingly a key criterion for partnership with major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the operational realities. Stakeholders are encouraged to review these points to assess the feasibility of integrating this technology into their existing supply chains. For further clarification on specific impurity profiles or custom scaling requirements, direct consultation with our technical team is recommended.
Q: What are the primary advantages of the one-pot method over traditional lithiation routes?
A: The one-pot method eliminates the need for cryogenic conditions (-78°C) and hazardous reagents like butyl lithium, significantly improving operational safety and reducing energy consumption while maintaining high yields.
Q: Which palladium catalysts are most effective for this transformation?
A: The patent demonstrates efficacy with various catalysts including palladium acetate, tetrakis(triphenylphosphine)palladium, and PdCl2(dppf), allowing flexibility based on cost and availability.
Q: How does this process impact impurity profiles in the final API intermediate?
A: By avoiding the harsh quenching steps associated with organolithium chemistry and utilizing a controlled hydrolysis step, the process minimizes side reactions and simplifies purification, leading to a cleaner impurity profile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Dimethylpyridine-3-Boric Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify identity and assay. Our dedication to process excellence means that we can reliably supply 2,6-dimethylpyridine-3-boric acid with the consistency and quality required for GMP environments.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to understand how our optimized manufacturing capabilities can enhance your supply chain resilience and drive down your overall cost of goods.
