Advanced Manufacturing of Brivaracetam Intermediates via Novel Cyclization and Resolution
Advanced Manufacturing of Brivaracetam Intermediates via Novel Cyclization and Resolution
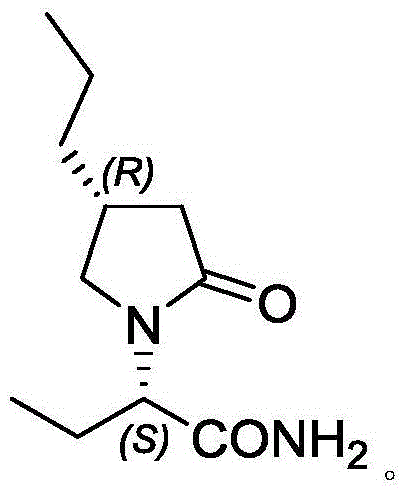
The pharmaceutical industry is constantly seeking more efficient pathways for the synthesis of complex antiepileptic agents, and the recent disclosure in patent CN112939900A offers a transformative approach to producing Brivaracetam intermediates. This intellectual property details a robust preparation method that bypasses the traditional reliance on costly chiral chromatographic separation, instead employing a sophisticated sequence of chemical resolution and cyclization reactions. The core innovation lies in the conversion of a specific amino acid derivative into a chiral butyrolactone structure, which serves as a critical scaffold for the final API. By leveraging a novel nitrite-mediated cyclization strategy combined with a classical Hoffmann rearrangement, this process achieves high stereoselectivity and yield without the need for ultra-low temperature operations or hazardous reagents often associated with prior art. For global supply chain leaders, this represents a significant opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with improved economic efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Brivaracetam and its precursors has been plagued by significant bottlenecks related to chiral purification and operational complexity. Many existing patents, such as CN1882535A, disclose methods that necessitate the use of preparative chiral chromatography columns, specifically utilizing stationary phases like CHIRALPAK AD with hexane/ethanol eluents. While effective for laboratory-scale purification, this technique is notoriously difficult to scale commercially due to the exorbitant cost of chiral stationary phases, the large volumes of solvent required, and the limited throughput of chromatographic systems. Furthermore, other prior art routes involve the use of highly reactive and dangerous reagents such as epichlorohydrin or require cryogenic conditions (ultra-low temperatures) that demand specialized refrigeration infrastructure and increase energy consumption drastically. These factors collectively inflate the cost of goods sold (COGS) and introduce substantial supply chain risks, making it challenging for procurement managers to secure consistent, cost-effective supplies of high-purity intermediates.
The Novel Approach
In stark contrast to these legacy methods, the methodology outlined in CN112939900A introduces a streamlined synthetic route that prioritizes chemical resolution over physical separation. The process initiates with the reaction of a dicarboxylic acid derivative with urea to form a lactam, which is subsequently hydrolyzed under mild alkaline conditions. A pivotal advantage of this new approach is the chiral resolution step, which employs readily available resolving agents such as S-(-)-alpha-phenylethylamine to separate enantiomers chemically rather than chromatographically. This shift not only eliminates the need for expensive chiral columns but also simplifies the downstream processing, as the resolved intermediate can be easily isolated via crystallization and filtration. The subsequent transformation involves a Hofmann rearrangement to shorten the carbon chain, followed by a unique nitrite-mediated cyclization to close the lactone ring. This sequence is designed to operate under moderate temperature conditions, avoiding the extreme thermal requirements of previous methods and thereby enhancing the overall safety and feasibility of industrial production.
Mechanistic Insights into Nitrite-Mediated Cyclization and Rearrangement
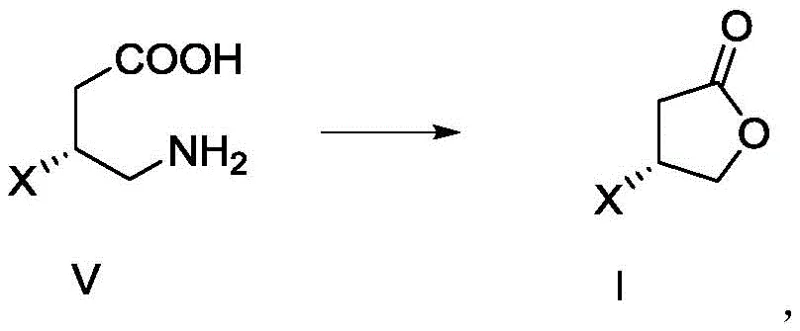
The chemical elegance of this patent lies in its final cyclization step, where Compound V is converted into the target lactone Compound I through a diazotization-mediated mechanism. In this critical transformation, the primary amine group of Compound V reacts with sodium nitrite in an acidic aqueous environment, typically utilizing hydrochloric acid with a mass fraction of 36% to 38%. This reaction generates a diazonium intermediate in situ, which is inherently unstable and rapidly undergoes intramolecular nucleophilic attack by the neighboring carboxylic acid group. The result is the formation of the five-membered lactone ring with the concomitant release of nitrogen gas. The patent specifies that maintaining the reaction temperature between -10°C and 50°C is crucial for optimizing the yield and minimizing side reactions. Following the initial acid reaction, the addition of an organic solvent such as dichloromethane or ethyl acetate facilitates the extraction and completion of the reaction, ensuring that the product partitions efficiently into the organic phase for easy isolation. This mechanistic pathway is particularly advantageous because it avoids the use of transition metal catalysts, thereby eliminating the risk of heavy metal contamination in the final product.
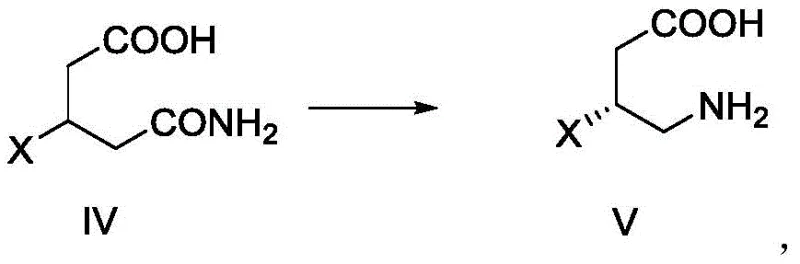
Preceding this cyclization is the Hoffmann rearrangement step, which converts the chiral amide (Compound IV) into the corresponding amine (Compound V) with one fewer carbon atom. This reaction is executed using sodium hypochlorite in the presence of sodium hydroxide, a classic reagent combination that generates the reactive N-chloroamide species necessary for the rearrangement. The patent highlights the importance of controlling the pH and temperature during this exothermic process, recommending a reaction range of -5°C to 40°C to preserve the stereochemical integrity of the chiral center. The use of a 10% available chlorine sodium hypochlorite solution ensures a steady supply of the oxidizing agent without the hazards associated with handling pure chlorine gas. The subsequent workup involves precise pH adjustments to precipitate the product, demonstrating a high level of control over impurity profiles. By integrating these two powerful transformations, the process achieves excellent stereoselectivity, as evidenced by the high ee values reported in the examples, ensuring that the final intermediate meets the rigorous purity standards required for pharmaceutical applications.
How to Synthesize Brivaracetam Intermediate Efficiently
The synthesis of this high-value intermediate is structured around four distinct chemical transformations that can be seamlessly integrated into a continuous or batch manufacturing workflow. The process begins with the condensation of a substituted glutaric acid with urea, followed by hydrolysis to generate the racemic amide-acid. The critical chiral resolution step then isolates the desired enantiomer using S-(-)-alpha-phenylethylamine in a mixed solvent system of ethanol and dichloromethane. Once the optical purity is established, the material undergoes the Hoffmann degradation to yield the chiral amino acid, which is finally cyclized using sodium nitrite to form the target lactone. Each step has been optimized for yield and purity, with detailed protocols provided for temperature control, reagent stoichiometry, and workup procedures. For process chemists looking to implement this route, the detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and compliance with GMP standards.
- React compound II with urea at 60-150°C, followed by alkaline hydrolysis to obtain compound III.
- Perform chiral resolution on compound III using S-(-)-alpha-phenylethylamine to isolate the optically active compound IV.
- Convert compound IV to compound V via Hoffmann rearrangement using sodium hypochlorite and sodium hydroxide.
- Cyclize compound V with sodium nitrite in acidic solution to form the final lactone intermediate Compound I.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound benefits for procurement managers and supply chain directors tasked with optimizing the cost structure of antiepileptic drug manufacturing. The most significant advantage is the drastic reduction in capital expenditure and operating costs associated with the elimination of chiral chromatography. By replacing expensive resin columns and large solvent volumes with standard chemical resolution techniques, manufacturers can achieve substantial cost savings in raw materials and waste disposal. Furthermore, the reagents utilized in this process, such as urea, sodium hypochlorite, and sodium nitrite, are commodity chemicals that are widely available in the global market, reducing the risk of supply disruptions caused by specialty reagent shortages. The simplicity of the operation, which avoids ultra-low temperature reactors and high-pressure hydrogenation equipment, also translates to lower maintenance costs and higher equipment utilization rates. These factors combine to create a more resilient and cost-effective supply chain, allowing pharmaceutical companies to negotiate better pricing and secure long-term supply agreements with greater confidence.
- Cost Reduction in Manufacturing: The elimination of chiral chromatographic purification represents a major breakthrough in cost engineering for this intermediate. Traditional methods require the continuous purchase of expensive chiral stationary phases and vast quantities of HPLC-grade solvents, which constitute a significant portion of the variable costs. By shifting to a chemical resolution strategy using inexpensive amines and standard solvents, the process significantly lowers the direct material costs. Additionally, the avoidance of transition metal catalysts removes the need for costly metal scavenging steps and extensive analytical testing for residual metals, further streamlining the production budget. This leaner cost structure enables manufacturers to offer more competitive pricing to their downstream clients while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals rather than specialized proprietary reagents greatly enhances the robustness of the supply chain. Reagents like sodium hypochlorite and hydrochloric acid are produced globally in massive quantities, ensuring that production schedules are not held hostage by the lead times of niche suppliers. Moreover, the process operates under mild conditions that do not require specialized cryogenic infrastructure, meaning that production can be easily transferred between different manufacturing sites without significant retrofitting. This flexibility is crucial for mitigating geopolitical risks and ensuring business continuity in the face of regional disruptions. The simplified logistics of sourcing raw materials allow for larger inventory buffers and more predictable delivery timelines for the final intermediate.
- Scalability and Environmental Compliance: The process design inherently supports large-scale commercial production due to its straightforward unit operations and manageable exotherms. The reaction steps, such as the nitrite cyclization and Hoffmann rearrangement, are well-understood chemical transformations that can be safely scaled from pilot plants to multi-ton reactors. From an environmental standpoint, the method generates less hazardous waste compared to routes using epichlorohydrin or heavy metals, facilitating easier compliance with increasingly stringent environmental regulations. The ability to recycle solvents like dichloromethane and ethanol further reduces the environmental footprint, aligning with the sustainability goals of modern pharmaceutical companies. This scalability ensures that the supply can grow in tandem with market demand for Brivaracetam, preventing bottlenecks as the drug gains wider clinical adoption.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and claims presented in patent CN112939900A, providing a factual basis for evaluating the technology's viability. Understanding these details is essential for R&D teams assessing the technical risk and procurement teams evaluating the cost-benefit analysis. The clarity provided here aims to facilitate informed decision-making regarding the adoption of this advanced manufacturing process.
Q: How does this patent improve upon conventional Brivaracetam synthesis?
A: Unlike prior art relying on expensive chiral chromatography columns (e.g., CHIRALPAK AD), this method utilizes chemical chiral resolution with affordable reagents like S-(-)-alpha-phenylethylamine, significantly lowering equipment costs and improving scalability.
Q: What are the critical reaction conditions for the final cyclization step?
A: The conversion of Compound V to Compound I requires reacting with nitrite in an acid solution (preferably 36-38% HCl) at temperatures between -10°C and 50°C, followed by extraction and distillation to ensure high purity and yield.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method avoids hazardous reagents like epichlorohydrin and ultra-low temperature conditions found in other patents, utilizing common solvents and standard temperature ranges suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brivaracetam Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing a stable and high-quality supply of critical pharmaceutical intermediates like the Brivaracetam precursor described in CN112939900A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet the most stringent purity specifications. Our state-of-the-art facilities are equipped with rigorous QC labs capable of performing advanced chiral analysis and impurity profiling, guaranteeing that every batch delivered adheres to the highest industry standards. We understand that the transition to a new synthetic route requires a partner who can navigate the complexities of process validation and regulatory compliance with precision and expertise.
We invite you to collaborate with us to leverage this innovative technology for your supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your COGS. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply of high-purity intermediates that will strengthen your position in the competitive antiepileptic drug market.
