Revolutionizing Cilastatin Acid Production: Low-Temperature Isomerization for Commercial Scale-Up
Revolutionizing Cilastatin Acid Production: Low-Temperature Isomerization for Commercial Scale-Up
The pharmaceutical industry constantly seeks robust manufacturing pathways for critical enzyme inhibitors like cilastatin, a key component in combination therapies to protect renal function. A pivotal advancement in this domain is detailed in Chinese Patent CN101386588B, which discloses a novel preparation method for cilastatin acid that fundamentally alters the approach to stereochemical control. Unlike legacy processes that struggle with isomeric mixtures and thermal degradation, this technology employs a strategic low-temperature isomerization of the ester precursor. By directly converting the (E)-isomer mixture into the pure (Z)-configuration under strong acidic conditions at temperatures between -5°C and -1°C, the process achieves superior yield and purity profiles. This technical breakthrough addresses long-standing challenges in the synthesis of (Z)-7-chloro-((S)-2,2-dimethylcyclopropanecarboxamido)-2-heptenoic acid derivatives, offering a viable route for reliable pharmaceutical intermediates supplier networks aiming to enhance product quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cilastatin acid intermediates has been plagued by inefficiencies related to isomer separation and thermal stability. Prior art, such as United States Patent No. 5147868, describes methods where mixtures of E and Z isomers are heated to approximately 90°C under acidic conditions to induce isomerization. However, this high-thermal stress frequently triggers the formation of unknown impurities and leads to the degradation of the sensitive (S)-2,2-dimethylcyclopropane carboxamide moiety. Furthermore, alternative approaches disclosed in documents like WO2006/022511 rely on selective alkaline hydrolysis, which often suffers from poor selectivity, resulting in the concurrent hydrolysis of both E and Z esters. This lack of specificity necessitates complex purification steps, such as repeated washing with organic solvents or the use of macroporous resins, which drastically increase energy consumption and solvent waste. Another method, CN101200434A, attempts selective acid hydrolysis but results in significant yield loss of the desired Z-ester during the removal of the E-isomer, making it economically unfavorable for large-scale manufacturing.
The Novel Approach
The methodology outlined in CN101386588B circumvents these thermal and selectivity pitfalls by shifting the isomerization step to the ester phase under controlled cryogenic conditions. Instead of heating the mixture, the process utilizes a strong acid catalyst to drive the equilibrium from the (E)-7-chloro-((S)-2,2-dimethylcyclopropaneamido)-2-heptenoic acid ethyl ester to the desired (Z)-isomer at temperatures ranging from -5°C to -1°C. This mild condition prevents the thermal decomposition observed in older patents, ensuring the cyclopropane ring and amide bond remain intact. Following isomerization, the reaction solution contains predominantly the pure Z-ester, which is then subjected to direct alkaline hydrolysis. This sequence eliminates the need for separating isomers after hydrolysis, as the conversion happens beforehand. The result is a streamlined workflow that avoids the energy-intensive concentration of large aqueous volumes and removes the dependency on macroporous resin adsorption, thereby facilitating easier industrialization and cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Acid-Catalyzed Isomerization
The core chemical innovation lies in the precise manipulation of the double bond geometry within the heptenoic acid ester chain. The starting material typically exists as a mixture of geometric isomers, where the (E)-configuration is thermodynamically stable but pharmacologically inactive for cilastatin synthesis. Under the influence of a strong protonic acid—such as hydrochloric acid, sulfuric acid, or p-toluenesulfonic acid—the pi-electron density of the alkene is perturbed, allowing for rotation around the carbon-carbon double bond. By maintaining the reaction environment at sub-zero temperatures (-5°C to -1°C), the kinetic pathway favors the formation of the (Z)-isomer while suppressing side reactions like amide hydrolysis or cyclopropane ring opening. This temperature control is critical; higher temperatures would provide enough activation energy for degradation pathways, whereas the cryogenic window locks in the desired stereochemistry. The use of non-polar solvents like toluene further aids in managing the solubility of the organic esters while allowing the acid catalyst to function effectively at the interface or within the solution phase.
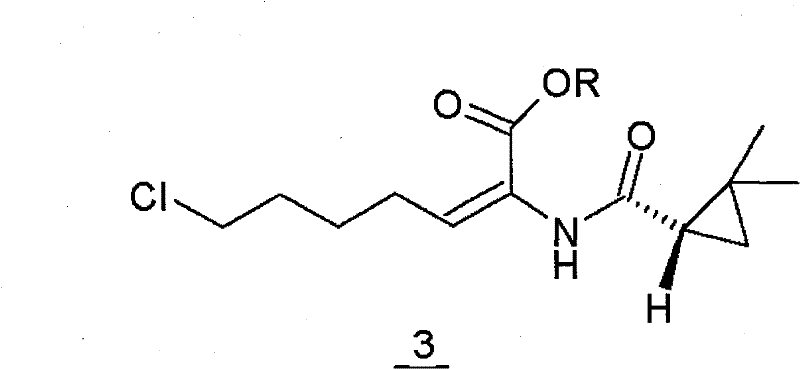
Following the successful isomerization, the mechanism proceeds to alkaline hydrolysis, where the ester functionality is cleaved to reveal the free carboxylic acid, immediately neutralized to form the sodium salt. The structural integrity of the molecule, particularly the chiral center on the cyclopropane ring and the chlorine tail, is preserved throughout this sequence. The avoidance of harsh thermal conditions during the isomerization step means that the impurity profile is significantly cleaner, with minimal formation of the difficult-to-remove (S)-2,2-dimethylcyclopropane carboxamide byproduct. This mechanistic precision ensures that the final high-purity pharmaceutical intermediates meet stringent regulatory specifications without requiring extensive chromatographic purification. The ability to drive the E-to-Z conversion to completion before hydrolysis essentially acts as a 'chemical filter,' ensuring that only the correct geometric isomer enters the final salt formation stage.
How to Synthesize (Z)-7-chloro-((S)-2,2-dimethylcyclopropanecarboxamido)-2-heptenoic Acid Efficiently
The synthesis of this critical cilastatin precursor requires strict adherence to temperature controls and stoichiometric ratios to maximize the E-to-Z conversion efficiency. The process begins with the preparation of the ester mixture, followed by the critical low-temperature isomerization step using a strong acid catalyst in a solvent like toluene. Once the isomerization is complete, typically requiring 3 to 24 hours depending on the specific acid strength and temperature, the reaction mixture is directly treated with a strong base such as sodium hydroxide. Detailed standardized synthetic steps see the guide below.
- Prepare a mixed solution containing (Z)- and (E)-7-chloro-((S)-2,2-dimethylcyclopropaneamido)-2-heptenoic acid ethyl esters in a suitable organic solvent like toluene.
- Conduct direct isomerization under strong acidic conditions at a controlled low temperature range of -5°C to -1°C to convert the E-isomer into the desired Z-isomer.
- Perform direct alkaline hydrolysis on the isomerized reaction solution to obtain the pure sodium salt, followed by acidification and salt formation to isolate the final intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented isomerization technology translates into tangible operational efficiencies and risk mitigation. Traditional methods that rely on high-temperature isomerization or non-selective hydrolysis often result in batch failures or inconsistent purity, leading to supply disruptions. By implementing a process that operates at low temperatures and avoids thermal degradation, manufacturers can achieve much higher consistency in batch-to-batch quality. This reliability is paramount for maintaining continuous supply lines for downstream API production. Furthermore, the elimination of macroporous resin and the reduction in solvent usage for purification directly correlate with a simplified waste management profile, reducing the environmental compliance burden and associated disposal costs.
- Cost Reduction in Manufacturing: The new method significantly lowers production costs by removing the need for expensive macroporous resin columns and the associated solvents required for their regeneration. Additionally, by avoiding the concentration of large volumes of aqueous solutions—a common requirement in older alkaline hydrolysis methods—the process drastically reduces energy consumption related to evaporation and drying. The higher yield of the desired Z-isomer from the starting mixture means that less raw material is wasted, optimizing the overall material cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: The robustness of the low-temperature isomerization process ensures that production timelines are not extended by complex purification loops or re-processing of off-spec batches. Since the method effectively converts the unwanted E-isomer into the useful Z-isomer, the reliance on highly enriched starting materials is reduced, allowing for greater flexibility in sourcing raw esters. This flexibility enhances supply chain resilience, as manufacturers are less vulnerable to shortages of specific high-purity isomers and can utilize broader grade feedstocks.
- Scalability and Environmental Compliance: Scaling this process is straightforward because it utilizes standard unit operations such as cooled stirred-tank reactors and liquid-liquid extraction, rather than specialized chromatographic equipment. The reduction in solvent waste and the avoidance of hazardous high-temperature acidic conditions contribute to a safer and more environmentally friendly manufacturing footprint. This alignment with green chemistry principles facilitates easier regulatory approval and supports long-term sustainability goals for chemical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this isomerization technology for cilastatin acid production. These insights are derived directly from the comparative data and process descriptions found in the patent literature, highlighting the specific advantages over prior art methods.
Q: Why is low-temperature isomerization preferred over traditional heating methods for cilastatin intermediates?
A: Traditional methods involving heating to 90°C often lead to the generation of unknown impurities and the degradation of the sensitive cyclopropane amide structure. Low-temperature isomerization (-5°C to -1°C) preserves the structural integrity of the molecule while effectively converting the unwanted E-isomer into the pharmacologically active Z-configuration.
Q: How does this new method improve the purity profile compared to alkaline hydrolysis routes?
A: Conventional alkaline hydrolysis often lacks selectivity, hydrolyzing both E and Z esters simultaneously, which complicates purification. By isomerizing the ester mixture to pure Z-ester *before* hydrolysis, this method ensures that the subsequent hydrolysis step yields a highly pure Z-acid sodium salt, significantly reducing the burden of downstream purification.
Q: What are the scalability advantages of avoiding macroporous resin in this process?
A: Eliminating the need for macroporous resin adsorption simplifies the equipment requirements and reduces solvent consumption associated with resin regeneration. This makes the process more robust for industrial scale-up, as it relies on standard liquid-liquid extraction and crystallization techniques rather than specialized chromatographic columns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cilastatin Acid Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of stereochemical purity and process robustness in the manufacture of renal dehydropeptidase inhibitors. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless. We are committed to delivering high-purity pharmaceutical intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation to verify isomeric ratios and impurity profiles.
We invite global partners to collaborate with us to leverage this advanced isomerization technology for their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your product quality while reducing overall procurement costs.
