Advanced Continuous Esterification Technology for High-Purity Reduced Coenzyme Q10 Acetate Manufacturing
Advanced Continuous Esterification Technology for High-Purity Reduced Coenzyme Q10 Acetate Manufacturing
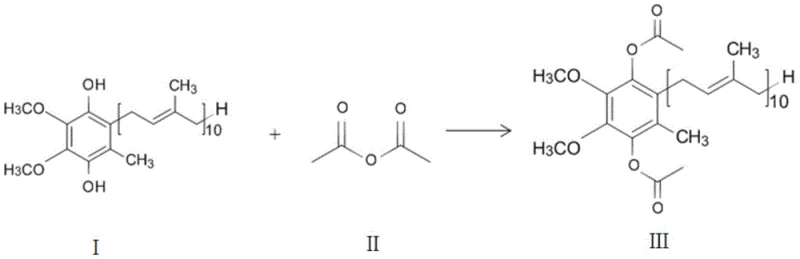
The pharmaceutical and nutraceutical industries have long grappled with the inherent instability of reduced coenzyme Q10, also known as ubiquinol, which limits its application despite superior bioavailability compared to its oxidized counterpart. Patent CN112047841B introduces a transformative methodology for the continuous production of reduced coenzyme Q10 acetate, addressing critical stability issues through a novel esterification protocol. This technology leverages a specialized supported catalyst system combined with continuous vacuum rectification to drive reaction equilibrium towards the desired product without the need for traditional acid-binding agents. By integrating catalyst regeneration loops and efficient byproduct separation, this process achieves conversion rates exceeding 97% and product purity levels above 99%, setting a new benchmark for the reliable reduced coenzyme q10 acetate supplier market. The implications for large-scale manufacturing are profound, offering a pathway to consistent, high-quality antioxidant intermediates that meet stringent global regulatory standards for dietary supplements and therapeutic formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acetylation of reduced coenzyme Q10 has been plagued by inefficiencies associated with batch processing and the reliance on soluble acid scavengers such as pyridine or triethylamine. As disclosed in prior art like GB925581a and CN104903289a, these conventional routes require stoichiometric quantities of base to neutralize the acetic acid byproduct, leading to significant challenges in downstream purification and waste management. The presence of residual acid-binding agents often necessitates multiple washing and extraction steps, which not only increases solvent consumption but also exposes the sensitive ubiquinol backbone to potential oxidative degradation during prolonged processing times. Furthermore, batch operations inherently suffer from variability in heat and mass transfer, resulting in inconsistent conversion rates and purity profiles that complicate quality control for high-purity OLED material or pharmaceutical grade applications. The inability to continuously remove the acidic byproduct also limits the reaction equilibrium, capping the theoretical yield and forcing manufacturers to deal with substantial amounts of unreacted starting material that are difficult to separate from the thermally labile product.
The Novel Approach
In stark contrast, the methodology outlined in CN112047841B revolutionizes this landscape by implementing a continuous flow system that utilizes a heterogeneous supported catalyst to facilitate esterification without soluble bases. This approach fundamentally alters the reaction dynamics by coupling the chemical transformation with simultaneous physical separation; specifically, the process employs reduced pressure rectification to continuously strip acetic acid from the reaction mixture as it forms. This application of Le Chatelier's principle drives the equilibrium overwhelmingly towards the formation of reduced coenzyme Q10 acetate, achieving conversion efficiencies that batch methods cannot match. The use of a supported catalyst, such as DMAP immobilized on mesoporous silica or polyamide resin, ensures that the catalytic activity remains confined to the solid phase, allowing for simple filtration and direct recycling of the catalyst bed. This eliminates the contamination risks associated with homogeneous catalysis and removes the burden of treating nitrogen-rich wastewater generated by amine scavengers, thereby offering a clear route for cost reduction in pharmaceutical intermediates manufacturing while enhancing the environmental profile of the production facility.
Mechanistic Insights into Supported Catalyst Esterification
The core of this technological advancement lies in the sophisticated design of the supported catalyst and its interaction with the reactants under dynamic vacuum conditions. The catalyst is engineered through a multi-step grafting process where a carrier material, such as mesoporous silica gel or molecular sieves, is first functionalized with a coupling agent like gamma-chloropropyl triethoxysilane. This creates a robust anchor for the active catalytic species, which can be selected from a group including 4-Dimethylaminopyridine (DMAP), dicyclohexylcarbodiimide (DCC), or benzyltriethylammonium chloride (TEBAC). When molten reduced coenzyme Q10 and acetic anhydride are introduced to this solid surface, the nucleophilic catalysis proceeds efficiently at temperatures between 60°C and 100°C. Crucially, the reaction vessel is maintained under a vacuum degree of -0.06 to -0.09 MPa, which lowers the boiling point of the generated acetic acid, allowing it to vaporize and exit the reaction zone immediately. This continuous removal prevents the accumulation of acid which could otherwise catalyze side reactions or reverse the esterification, ensuring that the reaction kinetics remain favorable throughout the residence time of 1 to 6 hours.
Beyond the primary reaction mechanics, the system incorporates a closed-loop regeneration strategy that preserves the integrity of the catalytic surface over extended operational cycles. As the supported catalyst undergoes repeated use, minor deactivation may occur due to fouling or leaching of the active component; however, the patent details a specific regeneration protocol where the spent catalyst is treated again with the coupling agent and active species to restore its loading capacity. This capability is vital for maintaining the commercial scale-up of complex polymer additives or fine chemical intermediates where catalyst cost is a significant factor. Furthermore, the purification stage utilizes a controlled crystallization process involving ethanol, where the crude product is dissolved at 50-60°C and slowly cooled to 0-10°C. This thermal gradient promotes the selective precipitation of the target acetate ester while leaving impurities and any trace unreacted ubiquinol in the mother liquor, resulting in a refined product with exceptional stability and purity suitable for sensitive biological applications.
How to Synthesize Reduced Coenzyme Q10 Acetate Efficiently
Implementing this continuous synthesis route requires precise control over feed rates, temperature gradients, and vacuum levels to maximize the efficiency of the supported catalyst system. The process begins with the preparation of the heterogeneous catalyst, followed by the continuous introduction of molten ubiquinol and acetic anhydride into the esterification reactor equipped with a rectification column. Detailed operational parameters, including specific reflux ratios and catalyst loading amounts, are critical to achieving the reported >97% conversion rates. For process engineers and technical teams looking to adopt this methodology, the following standardized synthesis steps outline the critical unit operations required to transition from laboratory scale to industrial production while maintaining product quality and safety standards.
- Prepare a supported catalyst by coupling a carrier like mesoporous silica with an active component such as DMAP using a silane coupling agent.
- Continuously feed molten reduced coenzyme Q10 and acetic anhydride mixed with the catalyst into a reactor under inert atmosphere at 60-100°C.
- Perform vacuum rectification during reaction to remove acetic acid, followed by distillation to recover excess anhydride and crystallization for purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from batch to continuous processing represented by this patent offers tangible strategic advantages beyond mere technical specifications. The elimination of stoichiometric acid-binding agents fundamentally restructures the cost basis of production by removing the expense of purchasing large volumes of amines and the subsequent costs associated with their disposal and wastewater treatment. This simplification of the bill of materials directly contributes to substantial cost savings, as the process relies primarily on the recyclable supported catalyst and recoverable acetic anhydride. Moreover, the continuous nature of the reaction ensures a steady output stream, mitigating the bottlenecks and inventory fluctuations typical of batch manufacturing. This reliability is essential for reducing lead time for high-purity nutraceutical ingredients, allowing downstream formulators to plan their production schedules with greater confidence and reduced safety stock requirements.
- Cost Reduction in Manufacturing: The economic model of this process is heavily favored by the ability to recycle both the catalyst and the excess acetic anhydride. In traditional methods, the acid scavenger is consumed and becomes waste, representing a sunk cost; here, the solid catalyst is filtered and reused, and the volatile anhydride is distilled and returned to the feed tank. This circular material flow drastically reduces raw material consumption per kilogram of product. Additionally, the sale of recovered acetic acid as a byproduct creates a potential revenue stream that further offsets production costs. By avoiding the complex extraction and washing steps needed to remove soluble bases, the process also reduces solvent usage and energy consumption related to solvent recovery, leading to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: Continuous processing inherently provides a more consistent product quality profile compared to batch-to-batch variations, which is critical for maintaining regulatory compliance in the pharmaceutical sector. The robustness of the supported catalyst system means that production can run for extended periods without interruption for catalyst changeovers, ensuring a continuous supply of material to meet market demand. This stability is particularly valuable for global supply chains where delays can have cascading effects on finished goods availability. The ability to regenerate the catalyst on-site further insulates the supply chain from external disruptions in catalyst sourcing, granting manufacturers greater autonomy and resilience against raw material price volatility or logistics constraints.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this technology offers a significantly cleaner production profile. The absence of nitrogen-containing waste streams simplifies effluent treatment, reducing the load on wastewater treatment plants and lowering compliance risks. The closed-system design minimizes operator exposure to reactive chemicals and volatile solvents, enhancing workplace safety. Scalability is straightforward because the continuous flow parameters can be adjusted by increasing the throughput of the reactor train rather than simply building larger vessels, which often introduces heat transfer challenges. This modularity allows for flexible capacity expansion to meet growing market demand for antioxidant supplements without requiring massive capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this continuous esterification technology. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing clarity on how this method compares to existing industry standards. Understanding these nuances is essential for technical evaluators assessing the feasibility of integrating this process into existing manufacturing lines or for procurement specialists validating the quality claims of suppliers utilizing this proprietary method.
Q: Why is reduced coenzyme Q10 acetate preferred over raw ubiquinol?
A: Raw reduced coenzyme Q10 (ubiquinol) is highly unstable and prone to oxidation in air. The acetate form significantly enhances chemical stability and shelf-life while maintaining bioavailability, making it superior for commercial formulation.
Q: How does the supported catalyst improve the esterification process?
A: The supported catalyst eliminates the need for soluble acid-binding agents like pyridine, simplifying downstream purification. It allows for easy filtration and regeneration, enabling continuous operation and reducing heavy metal or organic solvent residues.
Q: What represents the main cost driver in traditional ubiquinol acetylation?
A: Traditional methods rely on stoichiometric amounts of acid scavengers which generate significant wastewater and require complex separation steps. The new continuous method recycles both the catalyst and excess acetic anhydride, drastically lowering operational expenditures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Reduced Coenzyme Q10 Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced continuous manufacturing processes is key to securing a competitive edge in the global nutraceutical and pharmaceutical markets. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN112047841B are fully realized in practical, large-scale operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of reduced coenzyme Q10 acetate meets the highest standards of stability and potency required by top-tier international clients. Our commitment to technological excellence allows us to deliver products that not only meet but exceed the performance metrics of traditional batch-produced intermediates.
We invite you to collaborate with us to leverage these cutting-edge production capabilities for your specific product portfolios. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to our continuously produced materials. Please contact us to request specific COA data and route feasibility assessments tailored to your volume requirements, and let us demonstrate how our optimized supply chain can support your long-term growth and innovation goals in the health and wellness sector.
