Optimizing Acequinocyl Intermediate Production via Novel Fries Rearrangement Technology
Introduction to Advanced Acylation Technologies
The global demand for high-efficiency acaricides continues to drive innovation in the synthesis of key intermediates like 2-lauroyl-1-naphthol, a critical precursor for the commercial miticide Acequinocyl. Patent CN114956971A introduces a transformative two-stage synthetic methodology that replaces hazardous traditional Lewis acid catalysts with a safer, more efficient Brønsted acid-catalyzed esterification followed by thermal Fries rearrangement. This technological shift addresses long-standing industry pain points regarding equipment corrosion, heavy metal contamination, and complex waste streams associated with conventional Friedel-Crafts acylation. By integrating esterification and rearrangement into a streamlined sequence, the process achieves reported yields exceeding 90% with HPLC purity reaching 99%, setting a new benchmark for reliable agrochemical intermediate supplier standards. The strategic implementation of this route allows manufacturers to bypass the severe safety risks of handling boron trifluoride etherate while maintaining exceptional regioselectivity for the ortho-acylated product.
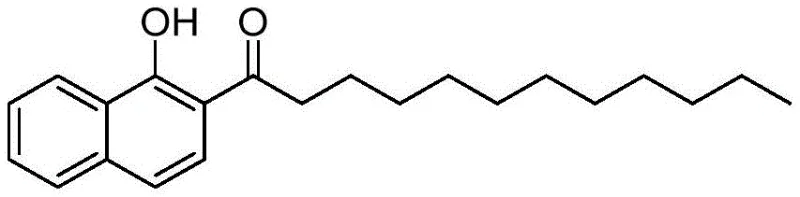
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-lauroyl-1-naphthol has relied heavily on direct Friedel-Crafts acylation using 1-naphthol and either lauric acid or lauroyl chloride in the presence of aggressive Lewis acids. As illustrated in prior art documents, methods utilizing anhydrous zinc chloride require massive excesses of catalyst and generate substantial volumes of heavy metal salt wastewater upon quenching with dilute hydrochloric acid. Alternatively, processes employing boron trifluoride diethyl etherate pose significant operational hazards, including the generation of corrosive hydrogen fluoride gas and the formation of explosive diethyl ether during aqueous quenching. These traditional pathways not only inflict severe corrosion on reactor vessels, necessitating expensive Hastelloy or glass-lined equipment, but also create a burdensome environmental liability due to the difficulty in treating boron-containing and zinc-laden effluent. Furthermore, the use of rare earth catalysts like Yb(OTf)3, while effective in small-scale laboratory settings, is economically prohibitive for industrial scale-up due to the exorbitant cost of the catalyst itself.
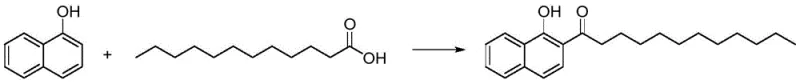
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a tandem esterification-Fries rearrangement strategy that fundamentally alters the reaction landscape. Instead of direct acylation, 1-naphthol is first converted into a naphthol ester intermediate using a catalytic amount of p-toluenesulfonic acid in a toluene solvent system. This initial stage operates under mild dehydration conditions, effectively removing water to drive the equilibrium forward without generating acidic wastewater. The subsequent thermal rearrangement step proceeds at elevated temperatures (160-180°C) without the need for additional catalysts, leveraging the intrinsic reactivity of the ester bond to migrate the acyl group to the ortho-position. This methodology completely eliminates the requirement for stoichiometric Lewis acids, thereby removing the source of heavy metal contamination and significantly simplifying the downstream purification process to a straightforward alcohol crystallization.
Mechanistic Insights into Tandem Esterification-Fries Rearrangement
The core of this innovative synthesis lies in the precise control of the two distinct reaction stages, which together ensure high regioselectivity and minimal byproduct formation. In the first stage, the Brønsted acid catalyst protonates the carbonyl oxygen of the long-chain fatty acid, facilitating nucleophilic attack by the hydroxyl group of 1-naphthol. The continuous removal of the reaction solvent (toluene) via azeotropic distillation ensures that the water produced during esterification is efficiently separated, pushing the reaction conversion to near completion before the temperature is raised. This careful management of reaction conditions prevents the premature degradation of the sensitive naphthol ring and ensures that the ester intermediate is formed in high yield before the rearrangement begins.
Upon increasing the temperature to the 160-180°C range, the reaction transitions into the Fries rearrangement phase, a concerted intramolecular migration of the acyl group from the oxygen atom to the adjacent carbon atom on the aromatic ring. This thermal activation overcomes the energy barrier for the [1,3]-sigmatropic shift-like process, favored by the coordination of the carbonyl oxygen with the phenolic proton or residual acid species. The mechanism inherently favors the ortho-position due to the proximity effect and the stability of the resulting chelated intermediate, which explains the high selectivity for the 2-acyl isomer over the 4-acyl isomer. Following the reaction, the addition of an alcohol solvent like ethanol induces crystallization, leveraging the solubility differences between the desired product and any unreacted starting materials or minor isomers to achieve the reported 99% purity without the need for column chromatography.
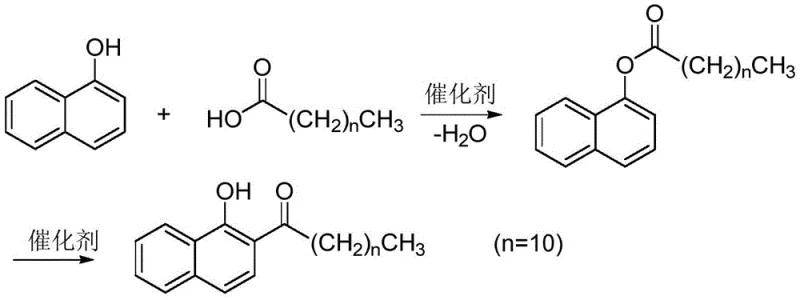
How to Synthesize 2-Lauroyl-1-Naphthol Efficiently
The operational protocol for this synthesis is designed for seamless integration into existing fine chemical manufacturing infrastructure, requiring only standard glass-lined or stainless steel reactors equipped with Dean-Stark traps for water removal. The process begins by charging 1-naphthol and lauric acid into the reactor with toluene and a catalytic load of p-toluenesulfonic acid, followed by heating to reflux to initiate esterification. Once water evolution ceases, the temperature is ramped up to induce rearrangement, after which the solvent is stripped under vacuum and the crude product is crystallized directly from ethanol. This streamlined workflow minimizes unit operations and solvent swaps, reducing both processing time and energy consumption compared to multi-step workups involving aqueous washes and extractions. For detailed standardized operating procedures and specific parameter optimization, please refer to the technical guide below.
- Stage 1: Esterification. React 1-naphthol with lauric acid (molar ratio 1:1.0-1.2) in toluene solvent using p-toluenesulfonic acid catalyst (0.01-0.10 eq) at 110-130°C for 3-4 hours with continuous water removal.
- Stage 2: Fries Rearrangement. Increase temperature to 160-180°C to induce thermal rearrangement of the naphthol ester intermediate for 2-3 hours without additional catalysts.
- Purification: Remove solvent under vacuum, cool to 80°C, add ethanol for crystallization, filter, and dry to obtain product with >99% HPLC purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this esterification-Fries rearrangement technology offers profound strategic benefits that extend beyond simple yield improvements. The elimination of expensive and supply-constrained Lewis acid catalysts, such as rare earth triflates or anhydrous zinc chloride, decouples production costs from the volatility of the specialty metals market. By relying on commodity chemicals like p-toluenesulfonic acid and toluene, manufacturers can secure a more stable and predictable cost structure, ensuring cost reduction in agrochemical intermediate manufacturing through raw material optimization rather than speculative trading. Furthermore, the simplified workup procedure reduces the consumption of auxiliary chemicals like hydrochloric acid and sodium bicarbonate, which are traditionally used in large quantities for quenching and neutralization in older processes.
- Cost Reduction in Manufacturing: The transition away from stoichiometric Lewis acids removes the significant expense associated with purchasing, handling, and disposing of these hazardous reagents. Since the new catalyst is used in merely catalytic quantities (0.01 to 0.10 molar ratio) and is inexpensive, the overall reagent cost per kilogram of product is drastically lowered. Additionally, the ability to recycle the crystallization mother liquor (ethanol) further enhances the economic efficiency of the process by minimizing solvent loss and waste disposal fees.
- Enhanced Supply Chain Reliability: Sourcing high-purity anhydrous zinc chloride or boron trifluoride complexes can often be a bottleneck due to their classification as hazardous materials and limited supplier base. In contrast, p-toluenesulfonic acid and long-chain fatty acids are widely available bulk commodities with robust global supply chains. This shift ensures that production schedules are not disrupted by raw material shortages, providing a reliable foundation for long-term supply contracts with downstream agrochemical formulators.
- Scalability and Environmental Compliance: The reduction in hazardous waste generation is a critical factor for scaling operations in regions with strict environmental regulations. By avoiding the creation of heavy metal wastewater and corrosive off-gases, facilities can operate with lower environmental compliance costs and reduced risk of regulatory shutdowns. The process is inherently safer, eliminating the risk of diethyl ether formation and explosion, which facilitates easier permitting for capacity expansion and commercial scale-up of complex agrochemical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the comparative data and experimental examples provided in the patent documentation, highlighting the practical implications for industrial adoption. Understanding these nuances is essential for technical teams evaluating the feasibility of transitioning from legacy Friedel-Crafts processes to this greener alternative.
Q: What are the primary advantages of the Fries rearrangement route over traditional Friedel-Crafts acylation for 2-lauroyl-1-naphthol?
A: The Fries rearrangement route eliminates the need for stoichiometric amounts of corrosive Lewis acids like anhydrous zinc chloride or boron trifluoride etherate. This significantly reduces heavy metal wastewater generation, minimizes equipment corrosion risks, and simplifies the post-treatment workup by avoiding complex hydrolysis and neutralization steps required in traditional methods.
Q: Can this synthesis method be adapted for analogues other than the lauroyl derivative?
A: Yes, the patent demonstrates high versatility for synthesizing analogues where the alkyl chain length varies. By substituting lauric acid with other long-chain n-alkyl carboxylic acids (where n=4 to 16), such as caprylic acid or stearic acid, the process yields corresponding 2-acyl-1-naphthol derivatives with similarly high regioselectivity and purity profiles suitable for diverse agrochemical applications.
Q: How does the new catalytic system impact environmental compliance and waste management?
A: By utilizing organic sulfonic acids like p-toluenesulfonic acid instead of heavy metal salts, the process drastically lowers the toxicity of the waste stream. The absence of boron or zinc residues means wastewater treatment is less costly and complex, aligning better with stringent modern environmental regulations for fine chemical manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Lauroyl-1-Naphthol Supplier
As the agrochemical sector increasingly prioritizes sustainable and cost-effective manufacturing pathways, NINGBO INNO PHARMCHEM stands ready to leverage this advanced Fries rearrangement technology for your supply needs. Our R&D team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity and yield metrics observed in the lab are faithfully reproduced at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-lauroyl-1-naphthol meets the exacting standards required for the synthesis of high-value miticides like Acequinocyl.
We invite you to engage with our technical procurement team to discuss how this optimized route can enhance your supply chain resilience and reduce your overall cost of goods. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. Please contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines.
