Advanced Enzymatic Synthesis of Natural Alpha-Tocopheryl Succinate for Commercial Scale-Up
Introduction to Green Biocatalytic Synthesis
The pharmaceutical and nutritional industries are increasingly demanding sustainable manufacturing pathways that align with stringent safety regulations while maintaining high product integrity. Patent CN101475968B introduces a groundbreaking methodology for synthesizing natural alpha-tocopheryl succinate monoester, a critical derivative of Vitamin E, utilizing lipase as a biocatalyst. This technology represents a significant paradigm shift from traditional chemical esterification, addressing long-standing issues regarding toxicity and energy consumption. By leveraging the specificity of enzymes like Candida lipase or Rhizopus lipase, this process operates under mild conditions, typically between 20°C and 45°C, thereby preserving the delicate stereochemistry of natural alpha-tocopherol. For R&D directors and procurement specialists, this patent offers a viable route to produce high-purity intermediates without the baggage of heavy metal residues or toxic amine catalysts, positioning it as a superior choice for modern supply chains focused on quality and compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of alpha-tocopheryl succinate has relied heavily on chemical catalysis involving harsh reagents and extreme conditions. Traditional protocols often utilize highly toxic tertiary amines, pyridine derivatives, or alkali metal salts as catalysts, necessitating reaction temperatures as high as 120°C to 140°C. These aggressive conditions pose severe risks to the stability of the tocopherol molecule, which is prone to oxidation when exposed to heat, light, and air, often requiring complex nitrogen blanketing systems to prevent degradation. Furthermore, the lack of specificity in chemical catalysts leads to a multitude of side reactions, resulting in dark-colored crude products that are difficult to refine. The subsequent purification steps required to remove toxic catalyst residues and by-products not only inflate production costs but also introduce potential safety hazards for end-users in the food and pharmaceutical sectors, making the conventional route increasingly untenable for premium applications.
The Novel Approach
In stark contrast, the novel enzymatic approach detailed in the patent utilizes lipase to catalyze the esterification of natural alpha-tocopherol with succinic anhydride in a non-aqueous system. This method operates at near-ambient temperatures, drastically reducing energy consumption and eliminating the thermal stress that causes vitamin E degradation. The use of polar organic solvents, such as dimethyl sulfoxide (DMSO) or its mixtures with tert-amyl alcohol, enhances the solubility of the hydrophobic tocopherol substrate while maintaining a low water activity environment favorable for lipase activity. This specificity ensures that the succinyl group is introduced selectively at the 6-position of the phenolic ring, minimizing by-product formation and yielding a product with superior color and stability. By removing the need for toxic catalysts and nitrogen protection, this process simplifies the workflow significantly, offering a cleaner, greener, and more economically viable pathway for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Lipase-Catalyzed Esterification
The core of this technological advancement lies in the unique catalytic mechanism of lipases within non-aqueous media. Unlike chemical catalysts that rely on brute force thermal energy to overcome activation barriers, lipases function through a precise lock-and-key interaction with the substrate. In this specific reaction, the enzyme's active site facilitates the nucleophilic attack of the phenolic hydroxyl group of alpha-tocopherol on the carbonyl carbon of succinic anhydride. The choice of solvent is critical; polar solvents like DMSO are employed not merely as a medium but as a strategic tool to dissolve the bulky tocopherol molecule without denaturing the enzyme's protein structure. This balance allows the reaction to proceed with high regioselectivity, ensuring that the esterification occurs exclusively at the desired position without affecting other functional groups on the chroman ring or the phytyl tail. The result is a highly controlled synthesis that mirrors biological precision, a feat difficult to achieve with traditional inorganic or organic chemical catalysts.
Furthermore, the impurity profile of the final product is inherently superior due to the enzyme's specificity. Chemical methods often generate poly-esters or oxidized degradation products due to the non-selective nature of high-temperature catalysis. In the enzymatic route, the mild conditions prevent the formation of free radicals that lead to oxidation, thereby preserving the antioxidant capacity of the vitamin E backbone. The immobilization of the lipase on carriers such as diatomaceous earth or resin further enhances the process by allowing the catalyst to be easily filtered and reused, which not only reduces waste but also prevents enzyme contamination in the final product. This mechanistic elegance translates directly into a simpler downstream processing workflow, where the absence of toxic catalyst residues means fewer washing and neutralization steps are required, ultimately leading to a higher overall yield and a cleaner final API or ingredient.
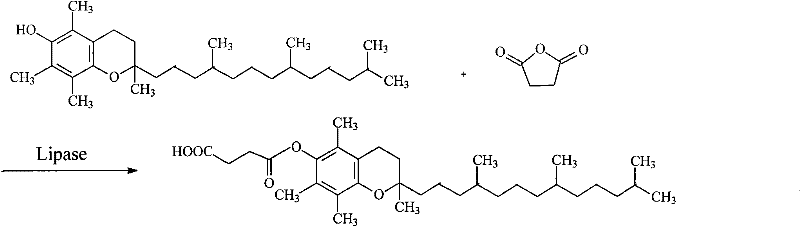
How to Synthesize Natural Alpha-Tocopheryl Succinate Efficiently
Implementing this enzymatic synthesis requires careful attention to solvent selection and reaction parameters to maximize the conversion efficiency of the valuable natural alpha-tocopherol starting material. The patent outlines a robust protocol where the molar ratio of substrates can be adjusted between 1:1 and 1:8 to drive the equilibrium towards the product, with reaction times varying from 20 to 72 hours depending on the desired throughput. Detailed standardized synthetic steps, including specific enzyme loading rates and agitation speeds, are essential for reproducibility at scale. For process engineers looking to adopt this technology, understanding the interplay between solvent polarity and enzyme stability is key to optimizing the batch cycle time. The detailed standardized synthesis steps are provided in the guide below to ensure consistent quality and yield.
- Prepare the reaction mixture by adding natural alpha-tocopherol and succinic anhydride into an organic solvent such as dimethyl sulfoxide (DMSO) or its mixtures with tert-amyl alcohol, maintaining a molar ratio between 1: 1 and 1:8.
- Introduce the biocatalyst, specifically free Candida lipase, Rhizopus lipase, or their immobilized forms, into the reaction vessel to initiate the esterification process.
- Maintain the reaction temperature between 20°C and 45°C with continuous shaking or stirring for a duration of 20 to 72 hours to achieve high conversion rates up to 98.39%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enzymatic process offers profound strategic benefits beyond mere technical superiority. The elimination of hazardous chemical catalysts fundamentally alters the cost structure of manufacturing by removing the need for expensive waste disposal and complex purification infrastructure. Traditional methods require extensive downstream processing to meet safety standards for food and pharmaceutical grades, whereas this green chemistry approach produces a cleaner crude product that requires less refinement. This simplification of the workflow directly correlates to reduced operational expenditures and a lower environmental footprint, aligning with global sustainability goals. Additionally, the mild reaction conditions reduce the energy load on manufacturing facilities, contributing to significant long-term cost savings in utility consumption without compromising on output quality or volume.
- Cost Reduction in Manufacturing: The replacement of toxic tertiary amines and pyridines with reusable immobilized lipase eliminates the costly steps associated with catalyst removal and neutralization. Since the enzyme can be filtered and reused multiple times, the recurring cost of catalysts is drastically minimized compared to single-use chemical reagents. Furthermore, the avoidance of high-temperature heating and nitrogen blanketing systems reduces energy consumption and equipment complexity, leading to substantial operational cost reductions. The simplified purification process also means higher recovery rates of the valuable active ingredient, maximizing the return on raw material investment.
- Enhanced Supply Chain Reliability: Sourcing high-purity natural alpha-tocopherol derivatives is often bottlenecked by the limited number of suppliers capable of meeting strict safety standards. This enzymatic method lowers the barrier to entry for high-quality production by utilizing readily available, non-toxic solvents like DMSO and common biocatalysts. The robustness of the reaction conditions, which do not require inert gas protection, simplifies reactor requirements and reduces the risk of batch failures due to oxidation. This reliability ensures a more consistent supply of critical intermediates, mitigating the risk of shortages that can disrupt downstream formulation schedules for pharmaceutical and nutraceutical clients.
- Scalability and Environmental Compliance: Scaling up enzymatic reactions is often perceived as challenging, but this specific protocol demonstrates high conversion rates even in mixed solvent systems, indicating strong potential for large-scale industrial application. The use of low-toxicity solvents and biodegradable enzymes aligns perfectly with increasingly stringent environmental regulations regarding volatile organic compounds (VOCs) and hazardous waste. By adopting this greener pathway, manufacturers can future-proof their operations against regulatory tightening while appealing to eco-conscious consumers and partners. The ability to operate at atmospheric pressure and moderate temperatures also enhances safety profiles, reducing insurance and liability costs associated with high-pressure or high-temperature chemical plants.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this lipase-catalyzed synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these nuances is crucial for stakeholders evaluating the feasibility of integrating this technology into their existing production lines. The answers provided reflect the specific advantages of the enzymatic method over traditional chemical synthesis, focusing on yield, safety, and operational simplicity.
Q: What are the primary advantages of using lipase over chemical catalysts for Vitamin E succinate synthesis?
A: The enzymatic approach eliminates the need for highly toxic tertiary amines or pyridines and avoids high-temperature conditions that cause oxidation. This results in a safer product profile suitable for food and pharmaceutical applications, with simplified downstream purification.
Q: Which solvents are compatible with this lipase-catalyzed esterification process?
A: The patent specifies the use of polar organic solvents, primarily dimethyl sulfoxide (DMSO), either alone or in mixed systems with acetone, tert-amyl alcohol, or tert-butanol. These solvents enhance substrate solubility while maintaining enzyme stability.
Q: What conversion efficiency can be expected from this biological synthesis route?
A: Under optimized conditions ranging from 20°C to 45°C over a period of 20 to 72 hours, the conversion rate of natural alpha-tocopherol reaches between 80% and 98.39%, demonstrating high reaction efficiency without the need for nitrogen protection.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Natural Alpha-Tocopheryl Succinate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of enzymatic catalysis in producing high-value fine chemicals and pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN101475968B are successfully translated into robust industrial realities. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards. We understand that consistency is key for our clients, and our advanced manufacturing capabilities allow us to deliver natural alpha-tocopheryl succinate with the reliability and purity required for sensitive pharmaceutical and nutritional applications.
We invite forward-thinking organizations to collaborate with us to leverage this green synthesis technology for their supply chains. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how switching to this enzymatic route can optimize your bottom line. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a secure, sustainable, and cost-effective source of premium vitamin E derivatives, empowering your R&D and production teams to focus on innovation while we handle the complexities of high-quality manufacturing.
