Advanced Manufacturing of Fluorinated Biphenyl Liquid Crystals via Heterogeneous Catalysis
Advanced Manufacturing of Fluorinated Biphenyl Liquid Crystals via Heterogeneous Catalysis
The rapid evolution of the display technology sector demands liquid crystal materials with increasingly precise electro-optical properties, driving the need for robust and scalable synthetic methodologies. Patent CN102093901B introduces a groundbreaking preparation method for 4-[2-(trans-4-alkylcyclohexyl)ethyl]-4'-but-3-enylfluoro-1,1'-biphenyl, a critical intermediate in the formulation of high-performance liquid crystal mixtures. This technology addresses the longstanding challenges associated with synthesizing fluorinated biphenyl derivatives containing ethylene bridges and terminal olefins, which are essential for achieving low melting points and wide nematic phase ranges. By shifting from traditional homogeneous catalysis to a heterogeneous system, this patent outlines a pathway that not only enhances product purity but also drastically simplifies the post-reaction workup, positioning it as a vital asset for any reliable electronic chemical supplier aiming to optimize their manufacturing portfolio.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted biphenyl compounds has relied heavily on palladium-catalyzed Suzuki coupling reactions utilizing homogeneous catalysts such as tetrakis(triphenylphosphine)palladium(0) or palladium chloride. While these traditional methods often deliver acceptable reaction yields in a laboratory setting, they present severe bottlenecks when translated to industrial manufacturing environments. The primary drawback lies in the inability to recover and reuse the expensive palladium species, leading to inflated raw material costs that erode profit margins. Furthermore, homogeneous catalysts are prone to oxidation, complicating storage and handling protocols, and their removal from the final product requires rigorous and costly purification steps to meet the stringent metal residue specifications demanded by the optoelectronics industry. These factors collectively hinder the cost reduction in electronic chemical manufacturing and limit the scalability of the production process.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by employing a heterogeneous 5% Pd/C catalyst for the key coupling step, fundamentally altering the economic and operational dynamics of the synthesis. This approach allows the catalyst to be easily separated from the reaction mixture via simple filtration, enabling its recovery and repeated use for multiple cycles without significant loss of activity. The elimination of complex metal scavenging procedures not only streamlines the workflow but also ensures a cleaner impurity profile, which is critical for high-purity OLED material and liquid crystal applications. Additionally, the subsequent steps involving acid hydrolysis and Wittig olefination are optimized for mild conditions, further reducing energy consumption and operational hazards, thereby offering a comprehensive solution for the commercial scale-up of complex polymer additives and fine chemical intermediates.
Mechanistic Insights into Heterogeneous Suzuki Coupling and Wittig Olefination
The core of this synthetic strategy relies on the efficient formation of the biaryl backbone through a heterogeneous Suzuki-Miyaura coupling mechanism. In this process, the aryl boronic acid and the halogenated aromatic precursor interact on the surface of the carbon-supported palladium particles. The heterogeneous nature of the catalyst facilitates a unique reaction environment where the active palladium sites are accessible to the substrates in the liquid phase while remaining immobilized on the solid support. This spatial separation is crucial for preventing the leaching of palladium into the solution, which is a common issue with other supported catalysts. The reaction proceeds through the standard oxidative addition, transmetallation, and reductive elimination cycle, but the solid support acts as a sink for the catalyst, allowing it to be physically removed once the conversion is complete. This mechanism ensures that the resulting intermediate retains minimal metal contamination, a key quality metric for any reliable agrochemical intermediate or pharmaceutical supplier expanding into electronic materials.
Following the coupling, the synthesis proceeds through a deprotection and functionalization sequence designed to install the terminal alkene moiety with high stereochemical control. The acetal protecting group is hydrolyzed under acidic conditions to reveal the aldehyde, which then serves as the electrophile in a Wittig reaction. 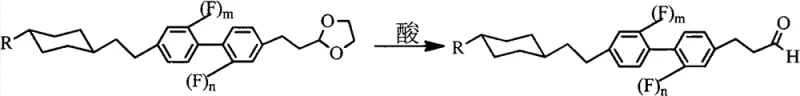 The Wittig olefination involves the generation of a phosphorus ylide from a phosphonium salt and a strong base, which subsequently attacks the carbonyl carbon of the aldehyde.
The Wittig olefination involves the generation of a phosphorus ylide from a phosphonium salt and a strong base, which subsequently attacks the carbonyl carbon of the aldehyde. 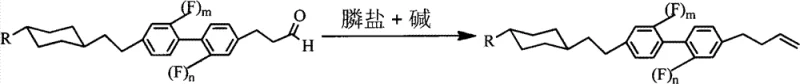 This step is meticulously controlled at temperatures ranging from -20°C to 25°C to favor the formation of the desired terminal alkene isomer while suppressing side reactions. The combination of these mechanistic steps results in a final product with a well-defined molecular structure, exhibiting the low viscosity and high voltage holding ratio required for advanced display applications.
This step is meticulously controlled at temperatures ranging from -20°C to 25°C to favor the formation of the desired terminal alkene isomer while suppressing side reactions. The combination of these mechanistic steps results in a final product with a well-defined molecular structure, exhibiting the low viscosity and high voltage holding ratio required for advanced display applications.
How to Synthesize Fluorinated Biphenyl Liquid Crystal Efficiently
The synthesis protocol described in the patent provides a clear roadmap for producing this valuable intermediate with high efficiency and reproducibility. The process begins with the coupling of specific aryl boronic acids with halogenated acetals in a mixed solvent system, followed by a straightforward acidic workup to generate the aldehyde precursor. The final transformation utilizes standard Wittig reagents to introduce the unsaturated tail, completing the molecular architecture. This route is specifically designed to minimize waste and maximize throughput, making it an ideal candidate for facilities looking to enhance their capabilities in reducing lead time for high-purity liquid crystal intermediates. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Perform Suzuki coupling using 5% Pd/C catalyst at 75-110°C to form the protected biphenyl acetal intermediate.
- Conduct acid hydrolysis at 50°C to convert the ethylene glycol acetal group into the corresponding aldehyde.
- Execute Wittig reaction at -20 to 25°C using phosphonium salts to generate the terminal alkene functionality.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented methodology offers substantial strategic benefits that extend beyond mere technical feasibility. The transition to a heterogeneous catalyst system directly addresses the volatility of precious metal markets by decoupling production costs from the continuous consumption of fresh palladium complexes. This stability allows for more accurate long-term budgeting and reduces the risk exposure associated with fluctuating commodity prices. Moreover, the simplified purification train reduces the dependency on specialized scavenging resins and extensive chromatography, which are often bottlenecks in large-scale manufacturing. These improvements collectively contribute to a more resilient supply chain capable of meeting the rigorous delivery schedules of global display manufacturers.
- Cost Reduction in Manufacturing: The implementation of a recyclable 5% Pd/C catalyst eliminates the recurring expense of purchasing stoichiometric or near-stoichiometric amounts of expensive homogeneous palladium catalysts for every batch. By enabling the catalyst to be filtered, washed, and reused multiple times, the overall consumption of precious metals is drastically reduced, leading to significant cost savings in the raw material bill. Furthermore, the removal of the catalyst via filtration obviates the need for costly downstream processing steps dedicated to removing trace metal residues, thereby lowering utility and consumable costs associated with purification.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain reliability by minimizing the number of critical reagents that require specialized handling or have long lead times. The use of common solvents like toluene, THF, and DMF, along with commercially available phosphonium salts, ensures that raw material sourcing remains flexible and less susceptible to single-supplier disruptions. The ability to recycle the catalyst also reduces the frequency of orders for high-value palladium species, smoothing out procurement cycles and ensuring a steady flow of production without waiting for external catalyst deliveries.
- Scalability and Environmental Compliance: This process is inherently scalable due to its reliance on heterogeneous catalysis and standard unit operations such as filtration and crystallization, which are easily adapted from pilot plant to full commercial production scales. The reduction in hazardous waste generation, particularly regarding heavy metal contamination in wastewater streams, aligns with increasingly strict environmental regulations. This compliance advantage mitigates the risk of regulatory fines and shutdowns, ensuring continuous operation and long-term viability for the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the operational advantages and quality standards associated with this technology. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this intermediate into their existing supply chains.
Q: What is the primary advantage of using Pd/C over homogeneous palladium catalysts in this synthesis?
A: The use of heterogeneous 5% Pd/C allows for simple filtration and catalyst recycling (over 5 times), eliminating the need for expensive and complex heavy metal scavenging processes required by homogeneous catalysts like Pd(PPh3)4.
Q: How does this method ensure high purity for electronic grade applications?
A: The process utilizes recrystallization steps with ethyl acetate and ethanol after each major transformation, combined with the physical removal of the solid catalyst, ensuring minimal metal contamination and high chemical purity suitable for display materials.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the protocol avoids sensitive homogeneous catalysts and uses standard solvents like toluene and THF, making it highly adaptable for commercial scale-up from 100 kgs to 100 MT annual production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorinated Biphenyl Liquid Crystal Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance intermediates play in the advancement of next-generation display technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of fluorinated biphenyl liquid crystal meets the exacting standards required by the global optoelectronics market.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific application needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and efficiency in your supply chain.
