Scalable Synthesis of Chiral Oxidized Aporphine Alkaloid Derivatives for Oncology Applications
Introduction to Patent CN103483256A and Chiral Aporphine Derivatives
The pharmaceutical industry is constantly seeking novel scaffolds with potent antitumor activity, and oxidized aporphine alkaloids have emerged as a promising class of candidates due to their broad-spectrum biological effects. Patent CN103483256A discloses a significant breakthrough in this field by providing a robust synthetic methodology for (-)-4-(2,3-dihydroxypropyl)-formamide-6-azabenzanthrone, herein referred to as (-)-FABA. Unlike traditional methods that rely on the difficult and low-yield extraction of these alkaloids from medicinal plants such as Magnoliaceae or Rutaceae, this patent outlines a fully synthetic route that guarantees the production of a single enantiomer with high optical purity. This is critical because, as established in pharmacological studies, the specific stereochemistry of a drug molecule often dictates its efficacy and safety profile, preventing the adverse effects associated with racemic mixtures. The disclosed compound exhibits remarkable proliferation inhibitory activity against various human tumor cell lines, particularly showing superior potency against BEL-7404 liver cancer cells compared to standard chemotherapeutic agents like cisplatin.
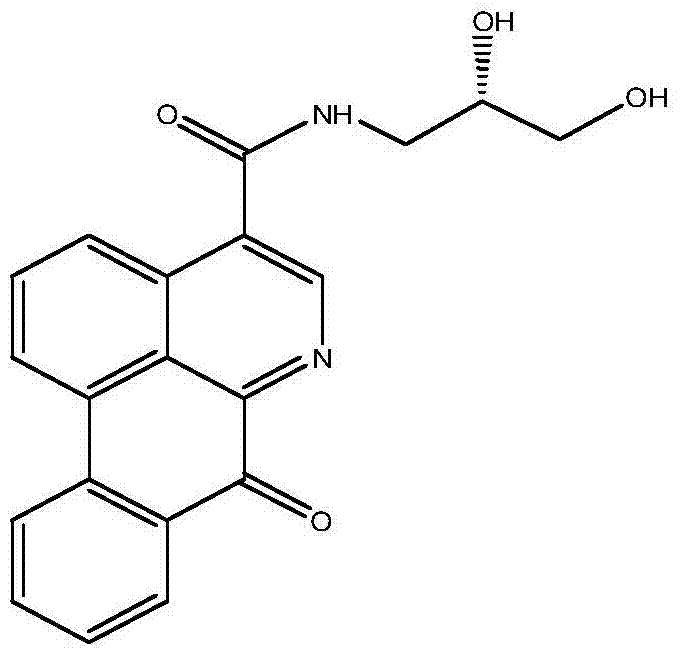
For R&D directors and procurement specialists, understanding the structural integrity and synthetic accessibility of (-)-FABA is paramount. The molecule features a rigid tetracyclic azabenzanthrone core linked via an amide bond to a chiral dihydroxypropyl side chain. This specific architecture is not merely a chemical curiosity but a functional necessity for interacting with biological targets involved in tumor growth. By securing a reliable pharmaceutical intermediate supplier capable of executing this synthesis, organizations can accelerate their oncology pipeline without the bottlenecks associated with natural product sourcing. The patent provides a clear roadmap from readily available starting materials to the high-value active pharmaceutical ingredient (API) precursor, marking a substantial step forward in cost reduction in API manufacturing for next-generation antitumor drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of oxidized aporphine alkaloids has been plagued by severe supply chain inconsistencies and technical hurdles inherent to natural product isolation. These compounds exist in trace amounts within their host plants, making large-scale extraction economically unviable and environmentally taxing. Furthermore, natural extracts are often complex mixtures containing numerous structurally similar analogs, necessitating arduous purification processes that drastically reduce overall yield. From a regulatory perspective, relying on botanical sources introduces variability in impurity profiles, which complicates the validation process for clinical trials. Additionally, conventional synthetic attempts often resulted in racemic mixtures, requiring expensive and inefficient chiral resolution steps to isolate the therapeutically active enantiomer. This lack of stereocontrol not only inflates production costs but also raises safety concerns regarding the inactive or potentially toxic mirror-image isomers, rendering such methods unsuitable for modern GMP-compliant commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
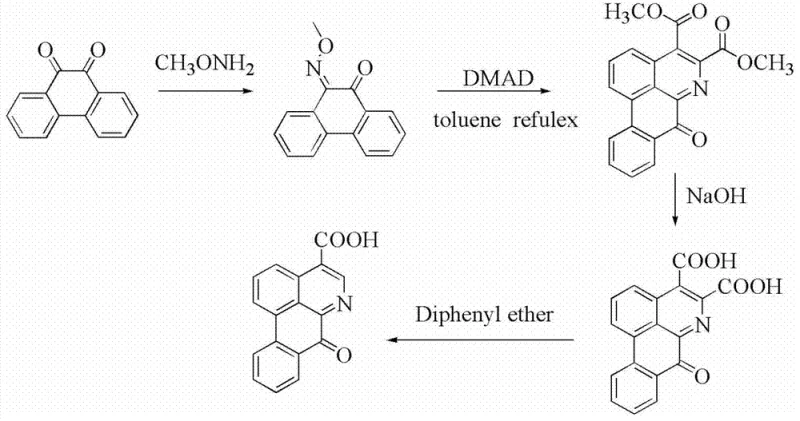
The methodology described in CN103483256A represents a paradigm shift by employing a convergent total synthesis strategy that bypasses the limitations of extraction. The process begins with the construction of the 4-carboxy-6-azabenzanthrone core from 9,10-phenanthrenequinone, a commercially abundant feedstock. This core is then coupled with a chiral amine building block, (-)-3-amino-1,2-propanediol, using advanced peptide coupling technology. This approach ensures that the chirality is built into the molecule from the outset, eliminating the need for post-synthesis resolution. The reaction conditions are remarkably mild, typically proceeding at temperatures between 20°C and 90°C, which minimizes thermal degradation and energy consumption. By utilizing a direct amidation pathway, the synthesis achieves high atom economy and simplifies the workup procedure. This novel route not only enhances the high-purity pharmaceutical intermediate output but also significantly streamlines the manufacturing workflow, making it an ideal candidate for industrial adoption.
Mechanistic Insights into PyBOP-Mediated Amidation
The cornerstone of this synthesis is the efficient formation of the amide bond between the sterically hindered carboxylic acid and the chiral amino alcohol. The patent specifies the use of PyBOP (benzotriazol-1-yl-oxytripyrrolidinyl hexafluorophosphate) as the coupling reagent in an N,N-dimethylacetamide (DMA) solvent system. Mechanistically, PyBOP activates the carboxyl group of the 4-carboxy-6-azabenzanthrone to form a highly reactive O-acylisourea intermediate, which is subsequently converted into an active ester species. This activation strategy is superior to traditional carbodiimide methods because it significantly suppresses racemization, a critical factor when handling chiral amines. The nucleophilic attack by the amino group of (-)-3-amino-1,2-propanediol proceeds smoothly to form the desired amide linkage. The choice of DMA as a solvent is strategic; it provides excellent solubility for both the polar reactants and the intermediate species, ensuring homogeneous reaction kinetics. Furthermore, the reaction can be monitored effectively via thin-layer chromatography, with completion typically achieved within 0.5 to 8 hours depending on the specific thermal profile employed.
Impurity control is another vital aspect addressed by this mechanistic design. The use of PyBOP generates soluble byproducts that can be easily removed during the aqueous workup phase, specifically through washing with saturated sodium carbonate solution. This step effectively quenches unreacted coupling reagents and removes phosphorous-containing residues. Subsequent purification via silica gel column chromatography using a chloroform-methanol gradient allows for the precise separation of the target (-)-FABA from any minor side products or unreacted starting materials. The patent data indicates that optimizing the molar ratios of reactants—specifically maintaining a balance between the acid, amine, and coupling agent—further minimizes the formation of N-acylurea byproducts. This rigorous control over the reaction environment ensures that the final product meets the stringent purity specifications required for preclinical and clinical evaluation, thereby reducing lead time for high-purity pharmaceutical intermediates entering the development pipeline.
How to Synthesize (-)-FABA Efficiently
The synthesis of (-)-4-(2,3-dihydroxypropyl)-formamide-6-azabenzanthrone requires precise adherence to the optimized parameters outlined in the patent to ensure maximum yield and stereochemical integrity. The process involves the preparation of the carboxylic acid precursor followed by the critical coupling step. Operators must ensure that all reagents, particularly the solvent DMA, are anhydrous to prevent hydrolysis of the activated intermediate. The reaction temperature should be carefully controlled, with a preference for the 40-60°C range to balance reaction rate and stability. Detailed standardized operating procedures for this synthesis are essential for reproducibility.
- Synthesize the key precursor 4-carboxy-6-azabenzanthrone from 9,10-phenanthrenequinone via oxime formation and DMAD cyclization followed by decarboxylation.
- Dissolve the carboxylic acid precursor and chiral amine (-)-3-amino-1,2-propanediol in anhydrous N,N-dimethylacetamide (DMA).
- Activate the carboxyl group using PyBOP coupling reagent at 40-60°C, followed by silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from extraction-based sourcing to this synthetic route offers profound strategic benefits. The primary advantage lies in the decoupling of production from agricultural variables; the synthesis relies on petrochemical-derived starting materials like 9,10-phenanthrenequinone and DMAD, which are available in bulk quantities with stable pricing. This shift eliminates the risks associated with crop failures, seasonal variations, and geopolitical instability often linked to botanical sourcing. Furthermore, the synthetic route is inherently scalable. The reaction conditions do not require exotic high-pressure equipment or cryogenic temperatures, allowing for implementation in standard multipurpose reactors found in most fine chemical manufacturing facilities. This compatibility with existing infrastructure translates to faster technology transfer and reduced capital expenditure for scaling operations.
- Cost Reduction in Manufacturing: The elimination of expensive chiral resolution steps is a major driver for cost efficiency. By using a chiral pool approach with (-)-3-amino-1,2-propanediol, the process avoids the 50% theoretical yield loss associated with resolving racemates. Additionally, the high efficiency of the PyBOP coupling reduces the need for excessive reagent usage and minimizes waste generation. The simplified purification protocol, which avoids complex recrystallization sequences, further lowers operational costs related to solvent consumption and labor. These factors collectively contribute to a more economical production model, enabling competitive pricing for the final API without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals ensures a robust and continuous supply chain. Unlike natural extracts, which may suffer from batch-to-batch variability in composition, this synthetic method produces a chemically defined substance with consistent impurity profiles. This consistency is crucial for regulatory filings and long-term supply agreements. The ability to produce the intermediate on demand reduces inventory holding costs and mitigates the risk of stockouts. Moreover, the synthetic route's flexibility allows for rapid adjustment of production volumes in response to market demand, providing a agile supply solution for pharmaceutical partners.
- Scalability and Environmental Compliance: The process is designed with environmental sustainability in mind. The solvents used, such as DMA and methanol, are recoverable and recyclable, aligning with green chemistry principles. The absence of heavy metal catalysts simplifies wastewater treatment and reduces the environmental footprint of the manufacturing process. The high yields reported in the patent examples indicate efficient resource utilization, minimizing the generation of hazardous waste. This environmental compliance not only reduces disposal costs but also enhances the corporate social responsibility profile of the manufacturing entity, making it a preferred partner for global pharmaceutical companies with strict sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of (-)-FABA. These answers are derived directly from the experimental data and claims presented in patent CN103483256A, providing clarity on the feasibility and advantages of this synthetic technology. Understanding these details is essential for stakeholders evaluating the integration of this intermediate into their drug development programs.
Q: What is the primary advantage of this synthetic route over natural extraction?
A: Natural extraction yields are extremely low and variable. This total synthesis method ensures a consistent supply of the single enantiomer with high purity, crucial for clinical development.
Q: How is chirality controlled in this process?
A: Chirality is introduced using the chiral building block (-)-3-amino-1,2-propanediol. The mild amidation conditions prevent racemization, ensuring the final product retains the specific (-) configuration required for biological activity.
Q: What are the key purification challenges?
A: The process utilizes standard silica gel column chromatography with a chloroform-methanol gradient. This avoids complex crystallization steps often required for similar alkaloids, simplifying downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (-)-FABA Supplier
The development of novel antitumor agents like (-)-FABA requires a manufacturing partner with deep technical expertise and a commitment to quality. NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of amidation reactions and chiral separations, ensuring that every batch meets stringent purity specifications. With rigorous QC labs and a dedicated team of process chemists, we are prepared to optimize this route for maximum efficiency and cost-effectiveness, delivering the high-quality intermediates necessary to advance your oncology pipeline.
We invite you to collaborate with us to unlock the full potential of this chiral aporphine derivative. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your regulatory submissions and clinical trial timelines. Let us be your trusted partner in bringing innovative cancer therapies to the market.
