Scalable Synthesis of 5-Bromo Oxoisoaporphine: A Strategic Route for Oncology Drug Development
Scalable Synthesis of 5-Bromo Oxoisoaporphine: A Strategic Route for Oncology Drug Development
The pharmaceutical landscape is continuously evolving with the demand for novel scaffolds that exhibit potent biological activity against resistant tumor lines. Patent CN103044326A introduces a groundbreaking total synthesis method for 5-bromo oxoisoaporphine, a derivative of the oxoisoaporphine alkaloid family known for its significant antitumor properties. This technical insight report analyzes the proprietary synthetic pathway disclosed in the patent, highlighting its feasibility for commercial manufacturing and its potential as a critical building block in oncology drug discovery. The compound, chemically defined as 1-aza-5-bromo benzanthrone, represents a strategic entry point for developing next-generation anticancer agents, offering a robust alternative to natural extraction methods which are often plagued by low yields and supply chain inconsistencies.
The core innovation lies in the efficient construction of the tetracyclic skeleton through a sequence of well-defined organic transformations. Starting from readily available 4-bromophenylacetonitrile, the process navigates through reduction, condensation, and dual cyclization steps to achieve the target molecule. This approach not only secures a reliable supply of high-purity material but also opens avenues for structural diversification around the bromo-substituted core. For R&D directors and procurement specialists, understanding the nuances of this synthesis is vital for evaluating its integration into existing pipelines for API intermediate sourcing and cost reduction in pharmaceutical manufacturing.
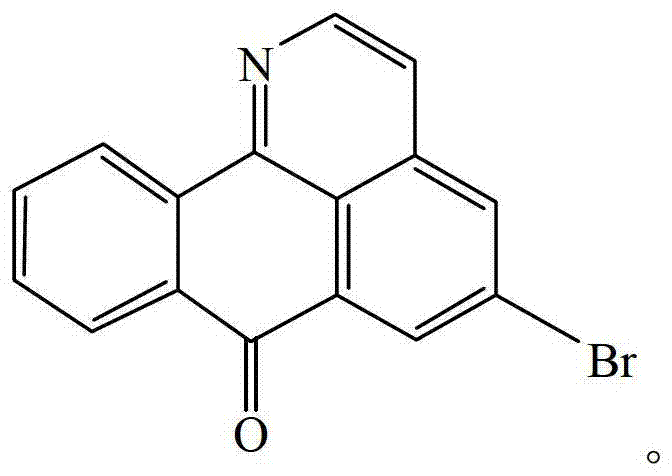
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, oxoisoaporphine alkaloids such as menisporphine were primarily obtained through extraction from Menispermaceous plants, a process fraught with logistical and economic challenges. The reliance on botanical sources introduces significant variability in yield due to seasonal changes, geographical location, and the inherent low concentration of these alkaloids within the plant matrix. Furthermore, the isolation and purification of specific derivatives from a complex natural extract require extensive chromatographic separation, leading to substantial solvent consumption and increased operational costs. Previous synthetic attempts, such as those reported by King in 1953 or Iwashima in 1984, often involved multi-step sequences with harsh conditions or limited scope for introducing specific substituents like the bromo group at the 5-position, thereby restricting their utility in modern medicinal chemistry programs focused on structure-activity relationship (SAR) studies.
The Novel Approach
The methodology outlined in CN103044326A presents a paradigm shift by employing a concise four-step total synthesis that bypasses the limitations of natural extraction. This novel approach utilizes 4-bromophenylacetonitrile as a cost-effective starting material, leveraging its inherent bromo functionality to direct subsequent cyclization reactions. The strategy involves a clever combination of phthalimide formation followed by two distinct Friedel-Crafts acylation events to construct the rigid tetracyclic framework. By controlling the reaction conditions, specifically the temperature and the nature of the Lewis acid media, the process achieves high regioselectivity and conversion rates. This synthetic route is not only shorter but also more amenable to scale-up, providing a consistent source of material that meets the stringent quality requirements of the pharmaceutical industry.
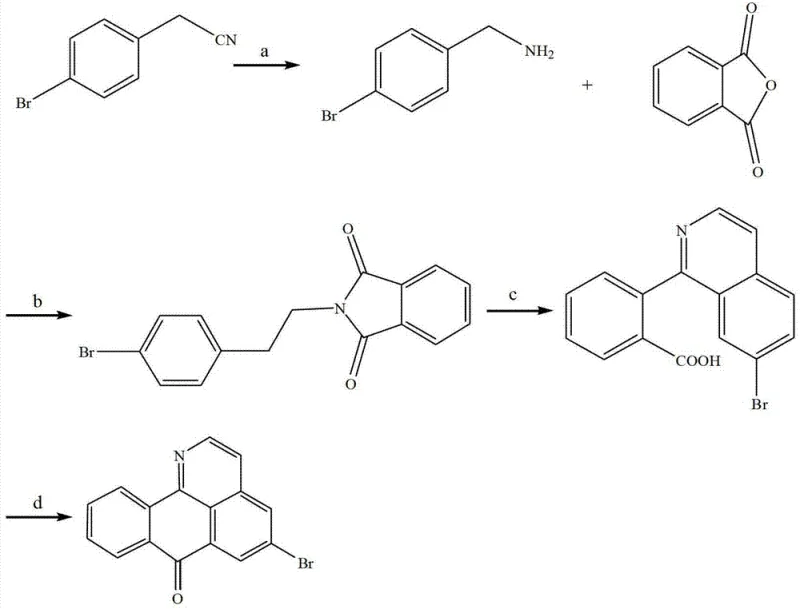
Mechanistic Insights into Dual Friedel-Crafts Cyclization Strategy
The success of this synthesis hinges on the precise execution of two sequential cyclization reactions that forge the carbon-carbon bonds necessary to close the rings. The first critical transformation involves the reaction of the phthalimide intermediate with anhydrous aluminum chloride in a molten sodium chloride medium. In this step, the aluminum chloride acts as a potent Lewis acid, activating the carbonyl group of the imide towards electrophilic aromatic substitution. The high temperature range of 180 to 220°C provides the necessary thermal energy to overcome the activation barrier for this intramolecular acylation, effectively closing the first ring to form the tricyclic intermediate. The use of a molten salt system is particularly ingenious as it serves as both a solvent and a heat transfer medium, ensuring uniform heating and preventing localized decomposition of the sensitive organic substrate.
Following the initial cyclization, the second ring closure is achieved through treatment with hot concentrated sulfuric acid. This step involves the protonation of the remaining carbonyl or imine functionality, generating a highly reactive electrophile that attacks the adjacent aromatic ring. The harsh acidic environment facilitates the dehydration and aromatization processes required to establish the fully conjugated oxoisoaporphine system. The mechanism likely proceeds through an acylium ion intermediate, which undergoes electrophilic attack on the electron-rich aromatic system ortho to the nitrogen atom. This dual cyclization strategy demonstrates exceptional control over the molecular architecture, ensuring that the bromo substituent remains intact throughout the vigorous reaction conditions, ultimately yielding the target 5-bromo oxoisoaporphine with high structural fidelity.
How to Synthesize 5-Bromo Oxoisoaporphine Efficiently
The synthesis of this valuable intermediate requires careful attention to reaction parameters, particularly temperature control and reagent stoichiometry, to maximize yield and purity. The process begins with the reduction of the nitrile group, followed by condensation and the aforementioned cyclization steps. Each stage has been optimized in the patent examples to balance reaction kinetics with product stability. For instance, the reduction step utilizes Lithium Aluminium Hydride, requiring strict anhydrous conditions to prevent premature quenching of the reagent. Similarly, the final purification via silica gel chromatography is critical for removing trace impurities and colored byproducts formed during the high-temperature acid treatments. Detailed operational guidelines for executing this synthesis are provided below to assist process chemists in replicating these results.
- Reduction of 4-bromophenylacetonitrile to 4-bromophenethylamine using Lithium Aluminium Hydride in anhydrous diethyl ether.
- Condensation with phthalic anhydride in ethanol under reflux to form the phthalimide intermediate.
- First cyclization via Friedel-Crafts acylation using molten Aluminum Chloride and Sodium Chloride at 180-220°C.
- Final ring closure in hot concentrated sulfuric acid at 200-240°C followed by purification via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers compelling advantages for procurement managers and supply chain heads looking to secure reliable sources of complex pharmaceutical intermediates. The primary benefit stems from the use of commodity chemicals as starting materials, which significantly mitigates the risk of supply disruptions associated with specialty reagents or natural extracts. 4-bromophenylacetonitrile and phthalic anhydride are produced on a massive industrial scale, ensuring their availability and price stability. This foundational security allows for long-term planning and contract negotiation without the fear of sudden raw material shortages that often plague niche botanical supply chains.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts, such as palladium or rhodium, which are common in cross-coupling reactions, results in substantial cost savings. Instead, the process relies on abundant and inexpensive Lewis acids like aluminum chloride and sulfuric acid. Furthermore, the avoidance of precious metals simplifies the downstream processing, as there is no need for costly metal scavenging steps or rigorous testing for residual heavy metals in the final API. This streamlined workflow reduces both the direct material costs and the operational overhead associated with waste management and quality control.
- Enhanced Supply Chain Reliability: The robustness of the chemical steps ensures high reproducibility, which is crucial for maintaining a steady supply of intermediates. The reactions described, such as reflux in ethanol or heating in molten salts, are standard unit operations that can be easily performed in multipurpose chemical reactors found in most cGMP facilities. This compatibility with existing infrastructure reduces the need for specialized equipment investment, thereby shortening the lead time for technology transfer and commercial scale-up of complex pharmaceutical intermediates.
- Scalability and Environmental Compliance: While the process involves strong acids, the waste streams are well-characterized and can be managed using standard neutralization and treatment protocols established in the fine chemical industry. The high atom economy of the cyclization steps, where most of the starting material ends up in the product, minimizes the generation of organic waste. Additionally, the ability to recycle solvents like chloroform and ethyl acetate during the purification phase further enhances the environmental profile of the manufacturing process, aligning with modern green chemistry initiatives and regulatory expectations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 5-bromo oxoisoaporphine. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these aspects is essential for stakeholders evaluating the feasibility of incorporating this intermediate into their drug development portfolios.
Q: What is the primary advantage of this synthetic route over plant extraction?
A: This total synthesis route eliminates the dependency on scarce natural sources like Menispermaceous plants, ensuring a consistent, high-purity supply chain independent of seasonal or geographical variations.
Q: How is the purity of the final 5-bromo oxoisoaporphine controlled?
A: The process utilizes a rigorous purification protocol involving chloroform extraction and silica gel column chromatography with a petroleum ether/ethyl acetate gradient, achieving purity levels exceeding 99.5%.
Q: Is this intermediate suitable for large-scale manufacturing?
A: Yes, the methodology relies on standard industrial reagents like Aluminum Chloride and Sulfuric Acid, avoiding expensive transition metal catalysts, which facilitates straightforward scale-up from kilogram to tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Bromo Oxoisoaporphine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in accelerating the drug discovery timeline. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We are committed to delivering 5-bromo oxoisoaporphine with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and assay. Our state-of-the-art facilities are equipped to handle the specific reaction conditions required for this synthesis, including high-temperature melt reactions and corrosive acid handling, guaranteeing a safe and efficient manufacturing environment.
We invite you to collaborate with us to leverage this innovative synthetic route for your oncology programs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can support your journey from bench-scale research to commercial launch. Let us be your trusted partner in bringing life-saving therapies to market faster and more efficiently.
