Advanced Synthesis of Halogenated Dihydropyranopyrrolone Intermediates for Oncology Applications
Advanced Synthesis of Halogenated Dihydropyranopyrrolone Intermediates for Oncology Applications
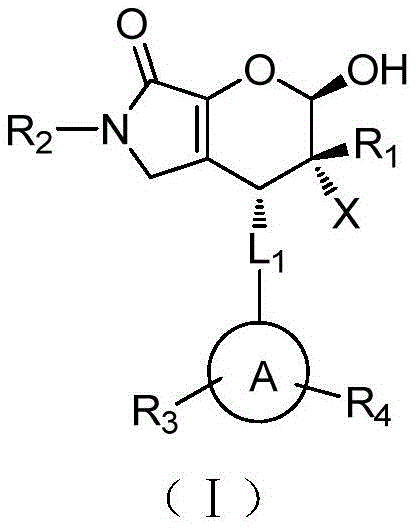
The pharmaceutical landscape is constantly evolving, driven by the urgent need for novel scaffolds that can effectively combat resistant tumor strains. Patent CN108084200B introduces a groundbreaking class of halogenated dihydropyranopyrrolone compounds, represented by Formula (I), which demonstrate potent antitumor properties. This technology addresses a critical gap in the synthesis of polysubstituted pyrano-ring skeletons, offering a robust pathway for generating high-value pharmaceutical intermediates. Unlike traditional methods that often struggle with stereocontrol and harsh reaction conditions, this invention leverages advanced organocatalysis to achieve exceptional purity and yield. For R&D directors and procurement specialists alike, understanding the nuances of this synthetic route is essential for securing a reliable supply chain for next-generation oncology therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of complex pyranopyrrolone frameworks has been fraught with challenges regarding regioselectivity and stereoselectivity. Prior art, such as the compounds disclosed in CN105541853A, focuses primarily on antibacterial activities and lacks the specific halogenated modifications required for optimized antitumor efficacy. Conventional synthetic routes often rely on transition metal catalysts, which introduce significant downstream processing burdens, including the costly and time-consuming removal of heavy metal residues to meet stringent regulatory standards for API intermediates. Furthermore, many existing methods suffer from low atom economy and require extreme temperatures or pressures, leading to inconsistent batch quality and increased operational expenditures. These limitations hinder the rapid scale-up necessary for clinical trials and commercial launch.
The Novel Approach
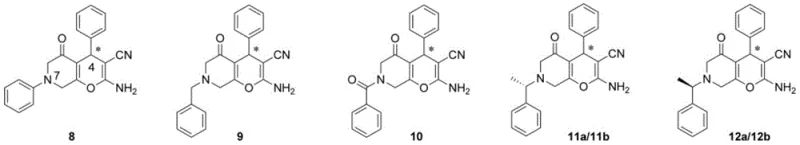
The methodology outlined in CN108084200B represents a paradigm shift towards greener and more efficient fine chemical manufacturing. By employing a modular three-step synthesis, the process allows for the precise installation of diverse functional groups—including halogens (F, Cl, Br, I) and various aryl substituents—at specific positions on the molecular scaffold. This flexibility is crucial for structure-activity relationship (SAR) studies, enabling medicinal chemists to rapidly iterate on lead compounds. The novel approach eliminates the need for precious metal catalysts, replacing them with an organocatalytic system that operates under mild conditions. This not only simplifies the purification workflow but also drastically reduces the environmental footprint of the production process, aligning with modern sustainability goals in the chemical industry.
Mechanistic Insights into Squaramide-Catalyzed Asymmetric Cyclization
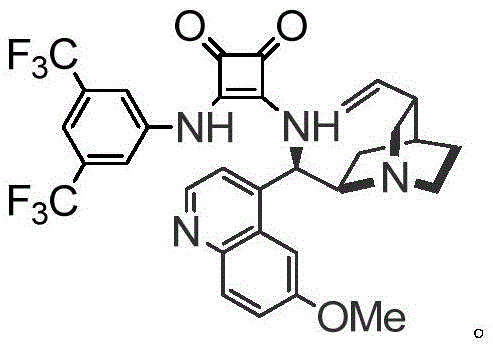
At the heart of this innovation lies a sophisticated organocatalytic cascade reaction mediated by a chiral squaramide catalyst, specifically derived from quinidine. This catalyst functions through a dual hydrogen-bonding activation mechanism, simultaneously activating both the electrophilic alkene intermediate (1) and the nucleophilic alpha-haloaldehyde (2). This precise spatial arrangement within the catalyst's chiral pocket ensures that the cyclization proceeds with high facial selectivity, resulting in the formation of multiple contiguous stereocenters with excellent control. The reaction typically proceeds at room temperature in chlorobenzene, avoiding the thermal degradation often seen in harsher acidic or basic conditions. This mechanistic elegance translates directly to product quality, delivering compounds with enantiomeric excess (ee) values reaching up to 99% and diastereomeric ratios (dr) as high as 96:4.
Impurity control is another critical aspect where this mechanism excels. The high specificity of the squaramide catalyst minimizes the formation of side products and regioisomers that typically plague non-catalyzed or metal-catalyzed cyclizations. By avoiding radical pathways or uncontrolled ionic reactions, the process ensures a clean impurity profile, which is vital for regulatory approval. The use of ethanol in the preliminary steps and chlorobenzene in the final cyclization allows for straightforward solvent recovery and recycling. For quality assurance teams, this means reduced analytical burden and higher confidence in batch-to-batch consistency, ensuring that every kilogram of high-purity pharmaceutical intermediate meets the rigorous specifications required for drug substance manufacturing.
How to Synthesize Halogenated Dihydropyranopyrrolone Efficiently
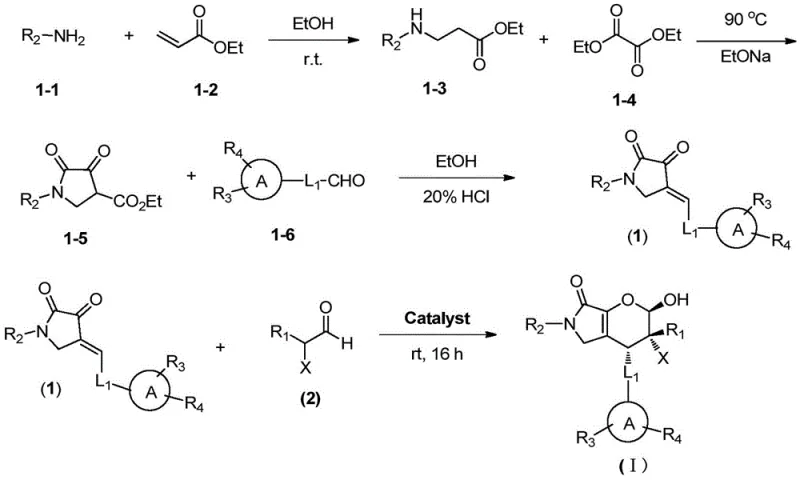
The synthesis protocol described in the patent is designed for operational simplicity without compromising on stereochemical outcomes. The process begins with the formation of a pyrrolone precursor via a Michael addition-cyclization sequence, followed by a Knoevenagel condensation to install the exocyclic double bond. The final and most critical step involves the organocatalytic hetero-Diels-Alder-type cyclization. Detailed standard operating procedures for each stage, including precise molar ratios and workup techniques, are essential for replicating the high yields reported in the examples. For a comprehensive guide on executing this synthesis in a GMP environment, please refer to the standardized protocol below.
- Preparation of pyrrolone precursor (1-5) via reaction of benzylamine, ethyl acrylate, and diethyl oxalate in ethanol followed by cyclization with sodium ethoxide.
- Condensation of precursor (1-5) with substituted benzaldehyde (1-6) in ethanol with HCl to form the alkene intermediate (1).
- Asymmetric cyclization of intermediate (1) with alpha-haloaldehyde (2) using a quinidine squaramide catalyst in chlorobenzene to yield the final compound (I).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial strategic advantages for procurement managers and supply chain heads. The elimination of expensive transition metal catalysts such as palladium or rhodium directly correlates to a significant reduction in raw material costs. Moreover, the absence of heavy metals removes the necessity for specialized scavenging resins and extensive purification steps, thereby streamlining the manufacturing timeline. This efficiency gain allows for faster turnaround times from order to delivery, enhancing the overall agility of the supply chain. The reliance on commodity chemicals like benzylamine, ethyl acrylate, and substituted benzaldehydes ensures a stable and resilient supply base, mitigating the risks associated with sourcing exotic or single-source reagents.
- Cost Reduction in Manufacturing: The organocatalytic nature of the final step significantly lowers the cost of goods sold (COGS) by removing the dependency on precious metals. Additionally, the high yields (often exceeding 90%) minimize waste generation and maximize the output per batch, leading to substantial economic efficiency. The mild reaction conditions also reduce energy consumption compared to high-temperature or high-pressure alternatives, contributing to lower utility costs over the lifecycle of the product.
- Enhanced Supply Chain Reliability: The synthetic route utilizes widely available starting materials that are produced by multiple global suppliers, reducing the risk of supply disruptions. The robustness of the reaction conditions means that the process is less sensitive to minor variations in input quality, ensuring consistent production even when sourcing from different vendors. This reliability is crucial for maintaining continuous manufacturing schedules and meeting the demanding timelines of pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram to multi-gram scales in the patent examples. The use of ethanol and chlorobenzene, while requiring proper handling, allows for established solvent recovery protocols that align with environmental regulations. The high atom economy and reduced waste generation support green chemistry initiatives, making it easier to obtain necessary environmental permits for large-scale production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these halogenated dihydropyranopyrrolone compounds. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals.
Q: What represents the key technological breakthrough in this synthesis method?
A: The use of a chiral squaramide organocatalyst enables high enantioselectivity (up to 99% ee) and diastereoselectivity under mild conditions, eliminating the need for toxic transition metals.
Q: What are the biological applications of these halogenated compounds?
A: The synthesized compounds exhibit significant antitumor activity against breast cancer cell lines (MB468, SKBR3, MB231) and melanoma (A375), making them valuable leads for oncology drug development.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes commercially available raw materials, operates at moderate temperatures (25-90°C), and achieves high yields (up to 99%), indicating excellent scalability for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halogenated Dihydropyranopyrrolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technology described in CN108084200B for the development of novel antitumor agents. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from preclinical research to market launch. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee the quality of every batch.
We invite you to collaborate with us to leverage this advanced synthetic methodology for your drug discovery programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the oncology therapeutic market.
