Advanced One-Pot Synthesis of Trifloxystrobin Intermediates for Commercial Scale-Up
Advanced One-Pot Synthesis of Trifloxystrobin Intermediates for Commercial Scale-Up
The global demand for high-efficiency fungicides continues to drive innovation in the synthesis of key agrochemical building blocks. Patent CN111807990A introduces a groundbreaking synthetic methodology for producing the critical intermediate (E)-2-(methoxyimino)-o-methyl phenylacetic acid methyl ester, which serves as the foundational precursor for trifloxystrobin. This technical breakthrough addresses long-standing challenges in the industry by offering a route that combines operational simplicity with exceptional stereochemical control. By leveraging a novel one-pot hydrolysis and methylation strategy, manufacturers can now achieve product purity levels exceeding 99% while significantly streamlining the production workflow. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, this patent represents a pivotal shift towards more sustainable and economically viable manufacturing paradigms that align with modern regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of trifloxystrobin intermediates has been plagued by inefficient multi-step sequences that impose heavy burdens on both cost and environmental compliance. Traditional routes often rely on the bromination of precursor fragments or lengthy six-step reaction cascades involving repeated acidification and extraction processes. As illustrated in prior art documentation, these conventional methods frequently consume excessive equivalents of strong bases and mineral acids, leading to substantial generation of saline wastewater and solid waste. Furthermore, the necessity for multiple purification steps, such as vacuum distillation and recrystallization, often results in cumulative yield losses, with some reported total yields stagnating around 75%. These inefficiencies not only escalate the cost reduction in agrochemical manufacturing but also introduce significant supply chain risks due to complex handling requirements and lower overall throughput capacity.
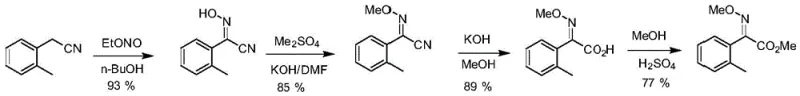
The Novel Approach
In stark contrast to these legacy processes, the methodology disclosed in CN111807990A utilizes a streamlined approach that consolidates hydrolysis and methylation into a cohesive, high-efficiency workflow. By starting from 2-oxime-o-tolylacetonitrile, the process enables the direct formation of acid or salt intermediates which are then subjected to a controlled double methylation reaction without the need for intermediate isolation in specific embodiments. This strategic simplification eliminates the need for harsh bromination reagents and reduces the reliance on toxic cyanides often found in older pathways. The result is a robust synthetic route that maintains a single trans-configuration throughout the reaction, ensuring that the final product meets the stringent purity specifications required for high-performance fungicide formulations without extensive downstream processing.
Mechanistic Insights into One-Pot Hydrolysis and Methylation
The core of this technological advancement lies in the precise control of reaction conditions during the alkaline hydrolysis and subsequent methylation phases. The process begins with the heating of 2-oxime-o-tolylacetonitrile in an alkaline aqueous solution, where temperature regulation up to 106°C facilitates complete conversion to the corresponding carboxylic acid or its sodium/potassium salts. This step is critical for establishing the correct stereochemistry, as the conditions favor the formation of the thermodynamically stable (E)-isomer. Following hydrolysis, the reaction mixture is either acidified to retrieve the free acid or maintained as a salt solution, which is then dissolved in polar aprotic solvents like DMF or DMSO. The addition of methylating agents such as dimethyl sulfate or methyl chloride at controlled temperatures ranging from -10°C to 45°C ensures selective methylation of both the oxime and carboxyl groups, driving the reaction to completion with minimal byproduct formation.
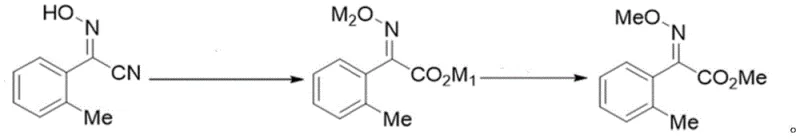
Impurity control is inherently built into this mechanistic design through the avoidance of intermediate isolation and the use of specific molar ratios of reagents. By optimizing the ratio of base to methylating agent, the process suppresses side reactions that typically lead to cis-isomer contamination or over-alkylation. The patent data indicates that liquid phase monitoring consistently shows raw material content dropping below 1% before the final workup, confirming high conversion efficiency. Moreover, the ability to use the crude salt directly in the methylation step without purification prevents the loss of material that occurs during filtration and drying in traditional methods. This mechanistic elegance translates directly into higher overall yields, with experimental examples demonstrating isolated yields reaching up to 86% and HPLC purity consistently above 99%, providing a level of quality assurance that is essential for downstream API synthesis.
How to Synthesize (E)-2-(Methoxyimino)-o-methyl Phenylacetic Acid Methyl Ester Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and temperature profiling to maximize the benefits of the one-pot design. The process is designed to be flexible, allowing manufacturers to choose between free acid or salt pathways depending on their specific equipment capabilities and waste treatment infrastructure. Detailed standard operating procedures for scaling this reaction from laboratory to pilot plant involve precise stoichiometry control and safety measures for handling methylating agents. The following guide outlines the critical operational parameters derived from the patent examples to ensure reproducible high-quality outcomes.
- Hydrolyze 2-oxime-o-tolylacetonitrile in alkaline aqueous solution at reflux temperatures around 106°C to form the acid or salt intermediate.
- Adjust pH if necessary to isolate free acid or proceed directly with the salt solution depending on the desired methylation pathway.
- React the intermediate with a methylating agent like dimethyl sulfate in organic solvent at controlled temperatures between -10°C to 45°C to finalize the ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical advantages that extend beyond simple yield improvements. The reduction in unit operations directly correlates to lower capital expenditure on equipment and reduced labor hours per batch, creating a leaner manufacturing profile. By eliminating the need for multiple extraction and neutralization steps, the process significantly lowers the consumption of organic solvents and inorganic reagents, which are major cost drivers in fine chemical production. This efficiency gain allows for more competitive pricing structures while maintaining healthy margins, making it an attractive option for long-term supply contracts in the volatile agrochemical market.
- Cost Reduction in Manufacturing: The streamlined nature of the one-pot process drastically reduces the consumption of acids, bases, and solvents compared to multi-step alternatives. By avoiding repeated pH adjustments and extractions, the operational expenditure associated with raw material procurement and waste disposal is substantially lowered. This efficiency means that manufacturers can achieve significant cost savings without compromising on the quality or purity of the final intermediate, providing a stronger value proposition for buyers seeking budget-friendly yet high-specification inputs.
- Enhanced Supply Chain Reliability: Simplifying the synthesis route reduces the number of potential failure points in the production schedule, thereby enhancing overall supply continuity. The use of readily available starting materials like 2-oxime-o-tolylacetonitrile ensures that raw material sourcing remains stable even during market fluctuations. Furthermore, the robustness of the reaction conditions minimizes the risk of batch failures due to sensitive parameters, allowing suppliers to maintain consistent delivery timelines and reduce lead time for high-purity agrochemical intermediates.
- Scalability and Environmental Compliance: The reduction in three-waste generation aligns perfectly with increasingly strict environmental regulations globally, reducing the liability and cost associated with waste treatment. The process is inherently safer and easier to scale from 100 kgs to 100 MT annual commercial production because it avoids hazardous bromination steps and toxic cyanide usage. This environmental compatibility ensures long-term operational viability and reduces the risk of regulatory shutdowns, securing the supply chain against compliance-related disruptions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this technology resolves specific industry pain points. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their broader manufacturing strategies.
Q: How does this new synthesis method improve impurity control compared to traditional routes?
A: The novel one-pot process minimizes intermediate isolation steps, which significantly reduces the introduction of external contaminants and degradation products often seen in multi-step batch processes, ensuring single trans-configuration purity exceeding 99%.
Q: What are the environmental benefits of using the salt-based methylation pathway?
A: By utilizing monosodium or disodium salts directly for methylation, the process eliminates the need for repeated acidification and neutralization cycles, drastically reducing the consumption of inorganic acids and bases and lowering the overall three-waste discharge burden.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the method employs readily available raw materials and mild reaction conditions without requiring expensive metal catalysts or harsh bromination steps, making it highly adaptable for commercial scale-up of complex agrochemical intermediates with consistent quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E)-2-(Methoxyimino)-o-methyl Phenylacetic Acid Methyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global agrochemical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN111807990A are fully realized in practical manufacturing environments. We are committed to delivering products with stringent purity specifications and rigorous QC labs testing every batch to guarantee consistency. Our infrastructure is designed to handle complex chemistries safely, providing our partners with a secure and reliable source for essential fungicide intermediates.
We invite you to collaborate with us to leverage these technical advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your production goals efficiently and sustainably.
