Revolutionizing Pamidronate Disodium Production: A Green One-Pot Synthesis Strategy for Global Markets
Introduction to Advanced Pamidronate Disodium Manufacturing
The pharmaceutical landscape for osteoporosis treatment has long relied on bisphosphonates, with Pamidronate Disodium standing out as a potent second-generation inhibitor of bone resorption. As detailed in the recent patent CN116253759A, a significant technological leap has been achieved through the development of a novel one-pot synthesis method. This innovation addresses critical bottlenecks in traditional manufacturing, specifically targeting the elimination of hazardous solvents and the simplification of purification steps. The new process utilizes a solvent-free phosphorylation strategy followed by direct hydrolysis and salt formation, fundamentally altering the production economics and environmental footprint of this key pharmaceutical intermediate. By integrating gradient heating and controlled crystallization, manufacturers can now achieve superior quality metrics while adhering to stricter green chemistry principles.
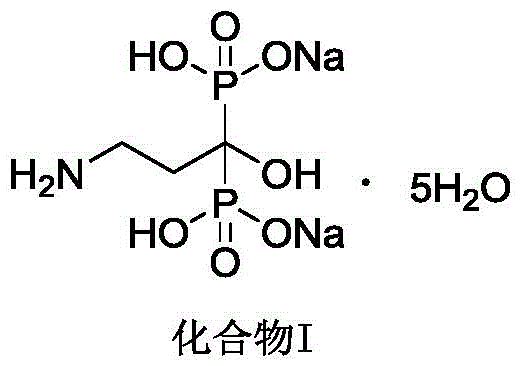
This structural integrity is paramount for biological activity, as the molecule mimics endogenous pyrophosphate but resists enzymatic degradation. The ability to produce this complex molecule with high fidelity and minimal impurity burden is a primary concern for R&D directors overseeing API synthesis. The patent highlights a shift away from legacy methods that struggled with viscous reaction masses and toxic solvent residues. Instead, this modern approach leverages precise stoichiometric control of phosphorous acid and phosphorus trichloride to drive the reaction to completion without the need for intermediate isolation. This not only streamlines the workflow but also significantly reduces the potential for cross-contamination and material loss during transfer operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Pamidronate Disodium has been plagued by reliance on hazardous organic solvents such as chlorobenzene and methanesulfonic acid. These solvents were necessary to manage the viscosity of the reaction mixture during phosphorylation, yet they introduced severe safety and environmental liabilities. Chlorobenzene, being highly toxic and difficult to recover completely, poses significant risks regarding residual solvent limits in the final drug substance. Furthermore, traditional processes often required the isolation of free pamidronic acid using alcohol solvents like methanol or acetone. This step is particularly problematic because methanol can react with phosphonic acid groups to form genotoxic phosphate ester impurities, while acetone may react with the amino group to create Schiff base derivatives. These side reactions compromise product purity and necessitate complex, costly purification protocols to ensure patient safety.
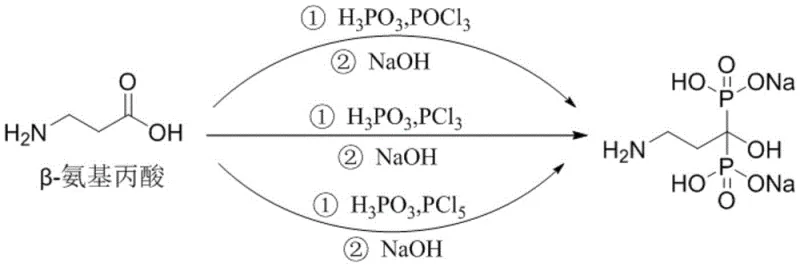
In addition to impurity concerns, the conventional multi-step approach suffers from low operational efficiency. The need to isolate, dry, and then re-dissolve intermediates increases the overall processing time and equipment occupancy. The use of volatile organic compounds (VOCs) also escalates the cost of waste treatment and requires specialized explosion-proof infrastructure. For supply chain managers, these factors translate into longer lead times and higher vulnerability to regulatory changes regarding solvent emissions. The accumulation of viscous byproducts in reactors further complicates heat transfer and mixing, creating hot spots that can lead to runaway reactions or inconsistent batch quality. Consequently, scaling these legacy processes to multi-ton production levels has always been a challenging endeavor fraught with technical and economic inefficiencies.
The Novel Approach
The breakthrough described in the patent introduces a streamlined one-pot methodology that effectively bypasses these historical hurdles. By conducting the phosphorylation of beta-alanine under solvent-free conditions, the process eliminates the need for chlorobenzene entirely. The reaction mixture is managed through a sophisticated gradient heating profile, starting from room temperature and carefully ramping up to 75°C. This thermal control prevents the violent exotherms often associated with phosphorus trichloride additions, ensuring a safe and homogeneous reaction environment. Following phosphorylation, the mixture undergoes direct hydrolysis with hydrochloric acid, and subsequently, the pH is adjusted with sodium hydroxide to precipitate the disodium salt directly. This telescoping of steps removes the isolation of the free acid, thereby cutting down processing time and minimizing material handling losses.
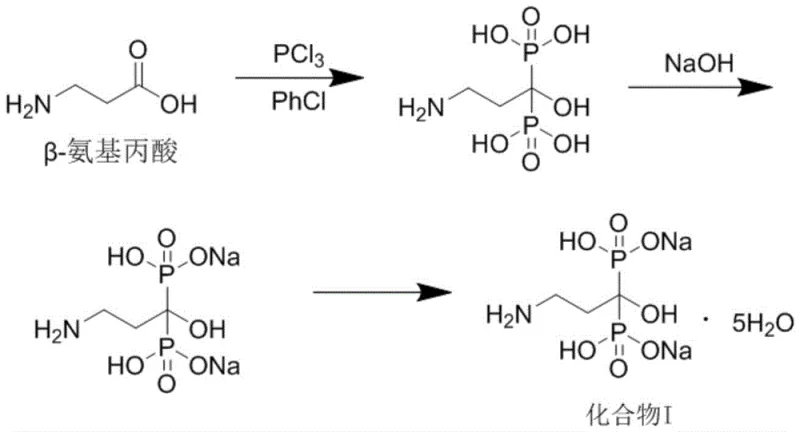
Furthermore, the novel approach replaces organic crystallization solvents with purified water, utilizing a gradient cooling technique to optimize crystal growth. This shift not only eradicates the risk of genotoxic phosphate ester formation but also simplifies the downstream processing significantly. Water is non-toxic, inexpensive, and easy to remove, aligning perfectly with modern sustainability goals. The elimination of organic solvents also means that the final product is free from the complex solvent residue profiles that often delay regulatory approval. For procurement teams, this translates to a more robust supply chain less dependent on fluctuating organic solvent markets. The overall result is a manufacturing process that is inherently safer, cleaner, and more cost-effective, providing a distinct competitive advantage in the global market for high-purity pharmaceutical intermediates.
Mechanistic Insights into Solvent-Free Phosphorylation and Crystallization
The core of this technological advancement lies in the mechanistic control of the phosphorylation reaction without solvent mediation. In traditional chemistry, solvents act as heat sinks and diluents to manage the reactivity of phosphorus trichloride. In this solvent-free system, the stoichiometry of phosphorous acid and phosphorus trichloride relative to beta-alanine is critically tuned, typically in a molar ratio ranging from 1.5:1.5:1 to 2.5:2.5:1. The gradient heating protocol—ramping slowly to 45°C, holding, then to 60°C, and finally to 75°C—allows the reaction mass to transition through phase changes smoothly. This prevents the formation of intractable solids that would otherwise halt mixing. The phosphorus trichloride activates the phosphorous acid in situ, generating the reactive phosphorylating species that attacks the carboxylic acid group of beta-alanine. This precise thermal management ensures complete conversion while minimizing the degradation of the sensitive amino group.
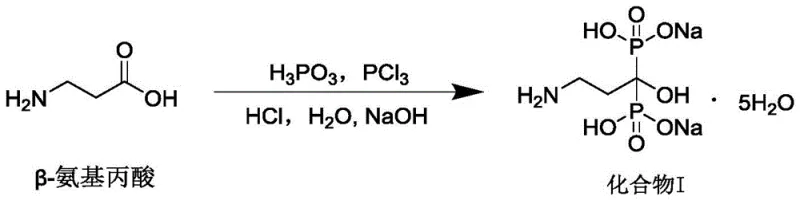
Following the reaction, the hydrolysis step converts the intermediate chlorides into the desired phosphonic acid groups. The subsequent salt formation is driven by pH control, where adjusting to a range of 7.0 to 8.0 ensures the formation of the disodium salt while keeping impurities in solution. The most critical mechanistic feature for purity enhancement is the gradient cooling crystallization. By cooling the aqueous solution first to 25°C and then to 5°C at controlled rates, the supersaturation level is managed to favor the growth of large, pure crystals over rapid nucleation. Rapid cooling often traps mother liquor and impurities within the crystal lattice, a phenomenon known as inclusion. The slow, staged cooling allows impurities to remain in the supernatant, resulting in a product with purity exceeding 99.96%. This level of control is essential for meeting the stringent impurity specifications required for injectable formulations used in oncology and osteoporosis therapy.
How to Synthesize Pamidronate Disodium Efficiently
Implementing this one-pot synthesis requires strict adherence to the thermal profiles and stoichiometric ratios defined in the patent to ensure reproducibility and safety. The process begins with the careful charging of beta-alanine, phosphorous acid, and phosphorus trichloride into a reactor capable of handling corrosive materials and exothermic reactions. The gradient heating must be automated or closely monitored to prevent thermal runaway during the initial phosphorylation phase. Once the reaction solidifies, the addition of hydrochloric acid for hydrolysis must be done cautiously to manage gas evolution. The subsequent neutralization with sodium hydroxide requires precise pH monitoring to avoid overshooting, which could affect the crystallization yield. Finally, the recrystallization step demands patience; the gradient cooling cannot be rushed if the target purity of >99.96% is to be achieved. Detailed standard operating procedures (SOPs) based on these parameters are essential for technology transfer.
- Phosphorylation: React beta-alanine with phosphorous acid and phosphorus trichloride under solvent-free conditions with gradient heating to 75°C.
- Hydrolysis and Salt Formation: Hydrolyze the mixture with hydrochloric acid, then directly adjust pH with sodium hydroxide to form the disodium salt without isolating the free acid.
- Purification: Recrystallize the crude product using purified water and activated carbon, employing a controlled gradient cooling protocol to maximize purity and yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solvent-free one-pot technology offers transformative benefits that extend beyond simple yield improvements. The most immediate impact is seen in the drastic reduction of raw material costs associated with organic solvents. By eliminating chlorobenzene, methanesulfonic acid, methanol, and acetone from the process, manufacturers remove the need for purchasing, storing, and recovering these expensive and regulated chemicals. This simplification of the bill of materials leads to significant cost reduction in pharmaceutical intermediate manufacturing. Furthermore, the removal of solvent recovery units reduces capital expenditure (CAPEX) for new facilities and lowers the operational expenditure (OPEX) related to energy consumption for distillation. The process becomes leaner, with fewer unit operations required to move from raw materials to the final packaged product.
- Cost Reduction in Manufacturing: The elimination of toxic organic solvents removes the substantial costs associated with solvent procurement, recycling infrastructure, and hazardous waste disposal. Without the need for complex solvent recovery systems, the energy load on the manufacturing plant is drastically lowered, leading to substantial operational savings. Additionally, the one-pot nature of the synthesis reduces labor hours and equipment occupancy time, allowing for higher throughput without expanding facility footprint. The avoidance of genotoxic impurity formation also reduces the cost of quality control testing and potential batch rejections, ensuring a more predictable cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: Relying on water as the primary crystallization medium decouples the production process from the volatility of the petrochemical solvent market. Organic solvents are subject to price fluctuations and supply disruptions based on oil prices and environmental regulations. By shifting to an aqueous system, the supply chain becomes more resilient and stable. Moreover, the simplified process flow reduces the number of potential failure points, such as solvent drying columns or recovery distillation towers, which are common bottlenecks in traditional synthesis. This reliability ensures consistent delivery schedules for downstream API manufacturers, reducing the risk of stockouts in the final drug supply.
- Scalability and Environmental Compliance: The solvent-free phosphorylation step significantly reduces the volume of hazardous waste generated per kilogram of product. This aligns with increasingly strict global environmental regulations regarding VOC emissions and wastewater treatment. Easier compliance translates to fewer regulatory hurdles and lower fines or remediation costs. From a scalability perspective, the absence of viscous solvent mixtures improves heat transfer efficiency in large-scale reactors, making the transition from pilot plant to commercial production smoother and safer. The process is inherently greener, enhancing the corporate sustainability profile of the manufacturer and appealing to environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this technology for their supply chains. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: How does the solvent-free method improve the safety profile of Pamidronate Disodium?
A: By eliminating toxic organic solvents like chlorobenzene and methanesulfonic acid, the process removes the risk of residual solvent toxicity and avoids the formation of genotoxic phosphate ester impurities often associated with alcohol-based crystallization.
Q: What are the yield and purity benchmarks achieved with this new one-pot technology?
A: The optimized one-pot process consistently achieves a total yield exceeding 81.5% and a final product purity greater than 99.96%, significantly outperforming traditional multi-step solvent methods.
Q: Why is gradient cooling crystallization critical in this synthesis route?
A: Gradient cooling prevents the rapid entrapment of impurities within the crystal lattice, ensuring higher chemical purity while maintaining a robust yield, which is essential for meeting stringent pharmacopeial standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pamidronate Disodium Supplier
As the global demand for effective osteoporosis and oncology treatments continues to rise, the need for high-quality, cost-effective intermediates like Pamidronate Disodium has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic methodologies to deliver superior products. Our expertise extends beyond simple manufacturing; we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art rigorous QC labs that enforce stringent purity specifications, ensuring that every batch meets or exceeds international pharmacopeial standards. We understand that consistency is key in the pharmaceutical supply chain, and our robust quality management systems are designed to guarantee batch-to-batch reproducibility.
We invite potential partners to engage with our technical team to explore how this innovative one-pot synthesis can optimize your supply chain. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Whether you are looking to secure a long-term supply agreement or need support in scaling a new formulation, NINGBO INNO PHARMCHEM is committed to being your strategic partner in delivering life-saving medications to patients worldwide efficiently and sustainably.
