Scalable Synthesis of Dihydropyrano-thiazole Derivatives for Oncology Drug Discovery
The landscape of oncology drug discovery is constantly evolving, driven by the need for novel heterocyclic scaffolds that offer both potent biological activity and synthetic accessibility. A significant advancement in this domain is detailed in Chinese Patent CN107759619B, which discloses a series of dihydropyrano-thiazole ring compounds represented by formula (I). These structures are not merely academic curiosities; they represent a robust class of intermediates with demonstrated antitumor properties against breast cancer and melanoma cell lines. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the synthetic versatility of these compounds is crucial. The patent outlines a methodology that bypasses traditional harsh conditions, utilizing a sophisticated organocatalytic cascade to achieve high stereocontrol.
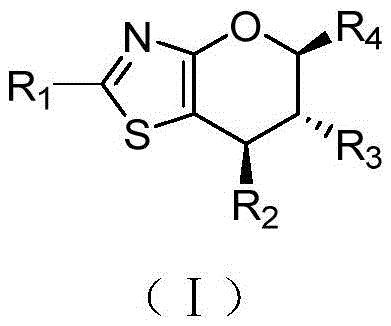
The core innovation lies in the modular nature of the synthesis, allowing for extensive derivatization at the R1, R2, and R3 positions. This flexibility is paramount for medicinal chemists aiming to optimize structure-activity relationships (SAR) during lead optimization phases. Furthermore, the compounds can exist in two distinct forms: the hydroxyl-substituted variant (I-1) and the deoxygenated variant (I-2), providing diverse chemical handles for further functionalization. This dual-pathway capability significantly enhances the utility of the scaffold in building complex libraries for high-throughput screening.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of fused thiazole-pyran systems has been fraught with challenges that hinder efficient cost reduction in pharmaceutical intermediate manufacturing. Traditional routes often rely on transition metal catalysis, which introduces the risk of heavy metal contamination—a critical failure point for GMP-grade API production. Moreover, many classical cyclization strategies require elevated temperatures, strong acids, or stoichiometric amounts of hazardous reagents, leading to poor atom economy and difficult waste stream management. These factors collectively increase the environmental footprint and operational costs, making such processes less attractive for large-scale commercial adoption.
The Novel Approach
In stark contrast, the methodology presented in CN107759619B employs a metal-free organocatalytic strategy that operates under remarkably mild conditions. The synthesis begins with the formation of a thiazole substrate, which then undergoes an asymmetric reaction catalyzed by a chiral secondary amine, specifically a diarylprolinol silyl ether. This approach eliminates the need for expensive and toxic transition metals, thereby simplifying the purification process and ensuring a cleaner impurity profile. The reaction proceeds efficiently at room temperature (25 ± 3°C), drastically reducing energy consumption compared to refluxing protocols. This shift towards greener chemistry aligns perfectly with modern sustainability goals while maintaining high yields and exceptional enantiomeric excess.
Mechanistic Insights into Organocatalytic Asymmetric Cyclization
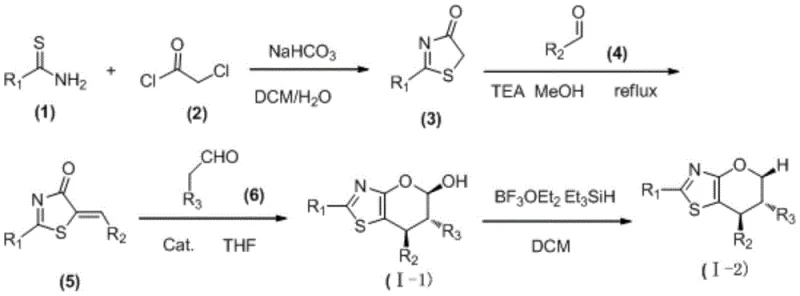
The heart of this synthetic breakthrough is the chiral amine-catalyzed cascade reaction. Mechanistically, the chiral catalyst activates the aliphatic aldehyde via enamine or iminium ion formation, facilitating a highly stereoselective attack on the electrophilic thiazole substrate. This step is critical for establishing the chiral centers at the C3 and C4 positions of the pyran ring. The use of tetrahydrofuran (THF) as a solvent, often with a small amount of water or benzoic acid as an additive, creates an optimal environment for this transformation. The result is the formation of the hydroxyl-bearing intermediate (I-1) with enantiomeric excess values frequently exceeding 90%, and in many cases reaching up to 99% ee, as evidenced by the experimental data for compounds 017 through 035.
For the synthesis of the deoxygenated analogues (I-2), the patent describes a subsequent reduction step using boron trifluoride diethyl etherate and triethylsilane. This Lewis acid-mediated hydride reduction selectively removes the hydroxyl group without affecting the sensitive thiazole ring or other functional groups on the aromatic substituents. This two-stage flexibility allows manufacturers to access a broader chemical space from a common set of precursors. The ability to toggle between the alcohol and the reduced alkane forms provides significant strategic advantages in drug design, enabling the fine-tuning of metabolic stability and binding affinity without redesigning the entire synthetic route.
How to Synthesize Dihydropyrano-thiazole Efficiently
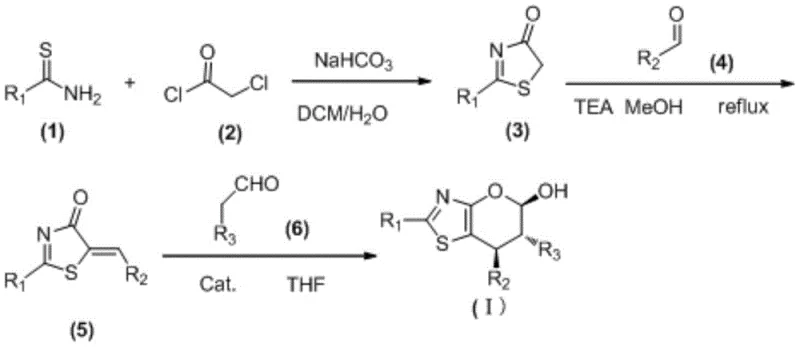
The practical execution of this synthesis is straightforward and amenable to standard laboratory and plant equipment. The process initiates with the reaction of a thioamide with chloroacetyl chloride in a biphasic system of dichloromethane and water, buffered by sodium bicarbonate. This generates the key thiazolone intermediate, which is then condensed with an aromatic aldehyde to form the reactive thiazole substrate. The final cyclization step involves mixing this substrate with an aliphatic aldehyde and the chiral catalyst in THF. Detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are essential for ensuring reproducibility and safety.
- React thiobenzamide with chloroacetyl chloride and sodium bicarbonate to form the thiazolone intermediate, followed by condensation with an aldehyde to generate the thiazole substrate.
- Perform an asymmetric organocatalytic reaction between the thiazole substrate and an aliphatic aldehyde using a chiral diarylprolinol silyl ether catalyst in THF.
- For deoxygenated derivatives, treat the hydroxyl-containing intermediate with boron trifluoride diethyl etherate and triethylsilane in dichloromethane.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The reliance on commodity chemicals such as chloroacetyl chloride, various substituted benzaldehydes, and simple aliphatic aldehydes ensures a stable and cost-effective raw material base. Unlike processes dependent on scarce noble metals or proprietary ligands, the catalysts used here are increasingly accessible, mitigating the risk of supply bottlenecks. This stability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical partners.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for expensive scavenging resins and complex metal removal validation steps, which are significant cost drivers in API manufacturing. Furthermore, the high yields reported (often above 90%) minimize raw material waste and maximize throughput per batch. The mild reaction conditions also translate to lower energy costs, as there is no need for prolonged heating or cryogenic cooling, resulting in substantial operational expenditure savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions—specifically the tolerance to ambient temperature and standard solvents—makes the process highly scalable. This reduces the technical risk associated with technology transfer from lab to pilot plant to commercial scale. Suppliers can confidently commit to long-term contracts knowing that the process is not susceptible to minor fluctuations in utility parameters, thereby ensuring a consistent supply of high-purity intermediates for downstream drug development.
- Scalability and Environmental Compliance: The aqueous workup in the initial step and the use of relatively benign solvents like THF and DCM (which can be recycled) simplify waste treatment protocols. By avoiding heavy metals, the process inherently meets stricter environmental regulations regarding effluent discharge. This compliance reduces the administrative burden and potential fines associated with hazardous waste disposal, making the overall manufacturing footprint lighter and more sustainable.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating these intermediates into their pipelines, we have compiled answers to common inquiries based on the patent specifications. These insights address critical aspects of purity, scalability, and application, providing a clear picture of what to expect when sourcing these materials.
Q: What is the primary advantage of the organocatalytic method described in CN107759619B?
A: The primary advantage is the ability to construct complex chiral dihydropyrano-thiazole scaffolds under mild conditions (25°C) with high enantioselectivity (up to 99% ee) and excellent yields, avoiding toxic heavy metal catalysts.
Q: Can this synthesis route be scaled for commercial production?
A: Yes, the process utilizes commercially available starting materials like thioamides and chloroacetyl chloride, and operates at ambient temperatures, making it highly suitable for scale-up from kilogram to multi-ton production without specialized high-pressure equipment.
Q: What biological activities do these compounds exhibit?
A: The synthesized dihydropyrano-thiazole derivatives demonstrate significant antitumor activity against various cell lines, including human breast cancer (MB468, SKBR3, MB231) and melanoma (A375), indicating their potential as oncology drug candidates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydropyrano-thiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that high-quality heterocyclic intermediates play in accelerating oncology research. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of dihydropyrano-thiazole derivative meets the highest international standards, ready for immediate use in preclinical and clinical studies.
We invite you to collaborate with us to leverage this advanced synthetic technology for your drug discovery programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to support your regulatory filings and development timelines.
