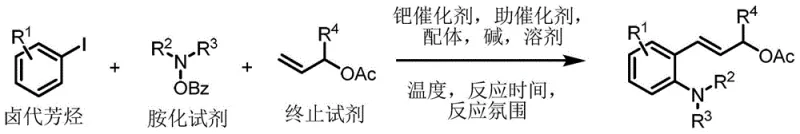
Advanced Palladium-Catalyzed Synthesis of 2-Aminocinnamyl Alcohol Esters for Pharmaceutical Intermediates
The pharmaceutical and agrochemical industries constantly demand efficient routes to polysubstituted aromatic hydrocarbons, which serve as critical scaffolds for bioactive molecules. A recent technological breakthrough documented in patent CN111943912A introduces a robust preparation method for 2-aminocinnamyl alcohol ester derivatives, addressing long-standing challenges in regioselectivity and functional group tolerance. This innovation leverages a palladium-catalyzed Catellani-type reaction, enabling the simultaneous introduction of multiple substituents onto an aromatic ring in a single operational sequence. By utilizing a cooperative catalytic system involving palladium and norbornene, this methodology bypasses the limitations of traditional stepwise functionalization, offering a streamlined pathway to complex intermediates essential for drug discovery and development.
For R&D directors and process chemists, the ability to access diverse chemical space rapidly is paramount. This patent outlines a versatile protocol that accommodates a wide range of halogenated aromatic hydrocarbons, including those bearing alkyl, halogen, cyano, acyl, ester, nitro, or trifluoroalkoxy groups. The reaction proceeds under relatively mild thermal conditions, specifically at 80°C, which helps preserve sensitive functional groups that might degrade under harsher regimes. Furthermore, the process demonstrates excellent stereoselectivity, exclusively yielding the thermodynamically stable E-configuration of the 2-aminocinnamyl alcohol ester product, a crucial attribute for ensuring consistent biological activity in downstream applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted arenes has relied heavily on sequential cross-coupling reactions or nucleophilic aromatic substitution (SNAr). These traditional approaches suffer from inherent inefficiencies, primarily because they can typically introduce only one substituent at a time. The position of the new functional group is rigidly dictated by the pre-existing location of the halogen atom on the starting material, severely restricting molecular design flexibility. Moreover, achieving ortho-functionalization often requires directing groups or harsh conditions that are incompatible with complex molecule synthesis. In the context of cinnamyl derivatives, controlling stereochemistry to obtain pure E-isomers often necessitates additional purification steps or specific reagents that drive up costs and extend lead times.
The Novel Approach
The methodology described in CN111943912A represents a paradigm shift by employing a Catellani reaction strategy, which integrates Heck reaction chemistry with norbornene mediation. This approach allows for the ortho-C-H activation of the aryl halide followed by ipso-substitution, effectively installing two different groups in a single pot. Crucially, this method overcomes the challenge of using allyl acetates as terminating reagents, which typically compete with other mechanisms in palladium catalysis. By optimizing the ligand and cocatalyst system, the invention ensures that the allyl acetate participates efficiently in the termination step, delivering the desired cinnamyl ester structure with high fidelity. This eliminates the need for silver salts, which are frequently used in alternative methods to promote reactivity but add significant expense and environmental burden to the process.
Mechanistic Insights into Pd/Norbornene Cooperative Catalysis
The core of this synthetic advancement lies in the intricate interplay between the palladium catalyst and the norbornene cocatalyst. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-halogen bond of the aryl iodide. Subsequently, the norbornene molecule inserts into the Pd-Aryl bond, forming a key palladacycle intermediate. This transient species enables the activation of the ortho-C-H bond on the aromatic ring, a step that is otherwise kinetically inaccessible. The amination reagent then intercepts this activated complex, installing the amine functionality at the ortho position relative to the original halogen site.
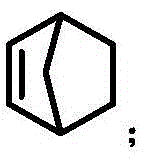
Following the ortho-amination, the norbornene moiety undergoes beta-carbon elimination, restoring the aromaticity and regenerating a Pd-Aryl species at the ipso position. This sets the stage for the final Heck-type coupling with the allyl acetate terminating reagent. The specific choice of ligands, such as tris(2-furyl)phosphine, plays a vital role in stabilizing the palladium center throughout this complex cycle and ensuring the exclusive formation of the E-alkene geometry during the termination step. This mechanistic precision minimizes the formation of Z-isomers or other regioisomeric byproducts, simplifying the impurity profile and facilitating easier downstream purification. Understanding this cycle is critical for scaling the process, as maintaining the correct stoichiometry of norbornene and base is essential to prevent catalyst deactivation.
How to Synthesize 2-Aminocinnamyl Alcohol Ester Efficiently
Implementing this synthesis requires careful attention to the reaction environment and reagent quality to maximize yield and reproducibility. The protocol is designed to be operationally simple, suitable for both laboratory-scale optimization and potential commercial scale-up. The process begins with the rigorous exclusion of oxygen, as the palladium catalyst is sensitive to oxidation. A standard procedure involves charging a reaction vessel with the catalyst system, substrates, and solvent under a nitrogen blanket. The reaction is then heated to a moderate temperature, allowing the catalytic cycle to turnover efficiently over a 24-hour period. Detailed standardized synthesis steps see the guide below.
- Charge a nitrogen-purged reaction vessel with palladium catalyst, ligand, norbornene cocatalyst, base, amination reagent, halogenated aromatic hydrocarbon, terminating reagent, and organic solvent.
- Heat the reaction mixture to 80°C and maintain stirring for 24 hours under a nitrogen atmosphere to ensure complete conversion and stereoselectivity.
- Cool the reaction mixture to room temperature, filter off solids, concentrate the filtrate, and purify the crude product via thin-layer or column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented method offers tangible benefits that translate directly into cost efficiency and supply reliability. The elimination of silver salts is a major economic driver, as silver reagents are not only expensive but also create heavy metal waste streams that require costly disposal procedures. By removing this requirement, the overall material cost per kilogram of the active intermediate is significantly reduced. Additionally, the use of common solvents like toluene and commercially available bases like cesium carbonate ensures that raw material sourcing remains stable and unaffected by niche supply chain disruptions.
- Cost Reduction in Manufacturing: The process achieves high yields without the need for precious metal additives beyond the catalytic amount of palladium, and specifically avoids stoichiometric silver salts. This reduction in reagent complexity lowers the bill of materials substantially. Furthermore, the mild reaction temperature of 80°C reduces energy consumption compared to high-temperature protocols, contributing to lower utility costs in large-scale production facilities. The simplified workup, involving basic filtration and concentration, minimizes labor hours and solvent usage during isolation.
- Enhanced Supply Chain Reliability: The substrates required for this reaction, such as substituted iodobenzenes and morpholine derivatives, are commodity chemicals with robust global supply chains. This reduces the risk of production delays caused by the scarcity of exotic starting materials. The robustness of the catalytic system against various functional groups means that the same process platform can be adapted for multiple analogues without extensive re-optimization, allowing for agile responses to changing market demands for different API intermediates.
- Scalability and Environmental Compliance: The reaction generates minimal hazardous waste, aligning with modern green chemistry principles and stringent environmental regulations. The absence of silver waste simplifies effluent treatment, making it easier to obtain necessary environmental permits for manufacturing sites. The straightforward purification via chromatography or crystallization ensures that the process can be scaled from grams to tons while maintaining high purity specifications, ensuring a consistent supply of high-quality intermediates for downstream drug substance manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy for process development teams evaluating this route for their own pipelines.
Q: What is the primary advantage of this Catellani reaction method over traditional cross-coupling?
A: Unlike traditional cross-coupling or nucleophilic substitution which typically introduce only one substituent at a time dependent on halogen position, this method utilizes a norbornene-mediated Pd catalytic cycle to achieve multi-substitution on the arene ring simultaneously with high stereoselectivity for the E-configuration.
Q: Does this synthesis require expensive silver salts?
A: No, a significant benefit of this patented protocol is that it achieves high yields and stereoselectivity without the need for additional silver salts, which are often required in other methods to facilitate halide abstraction, thereby reducing material costs and waste.
Q: What represents the optimal reaction conditions for this transformation?
A: The preferred embodiment utilizes palladium chloride as the catalyst, tris(2-furyl)phosphine as the ligand, cesium carbonate as the base, and toluene as the solvent, reacting at 80°C for 24 hours under nitrogen protection.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminocinnamyl Alcohol Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced catalytic technologies like the Pd-catalyzed Catellani reaction in accelerating drug development timelines. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-aminocinnamyl alcohol ester derivatives meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with our technical team to evaluate the feasibility of this route for your specific project needs. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in your supply chain.
