Revolutionizing Tafamidis Derivative Production: A Green, Scalable Synthetic Route for Global Pharma Supply Chains
Revolutionizing Tafamidis Derivative Production: A Green, Scalable Synthetic Route for Global Pharma Supply Chains
The pharmaceutical industry is constantly seeking more efficient, safer, and environmentally sustainable pathways for synthesizing critical active pharmaceutical ingredients (APIs) and their intermediates. Patent CN113321627B introduces a groundbreaking methodology for the synthesis of novel Tafamidis derivatives, a class of compounds vital for treating transthyretin-mediated amyloidosis (ATTR-CM). Unlike traditional methods that rely on hazardous acylation and esterification reagents, this patented approach utilizes a green oxidative cyclization strategy involving catechol compounds, ammonium acetate, and aldehydes, mediated by sodium periodate. This innovation not only simplifies the synthetic route but also drastically reduces the environmental footprint by avoiding toxic diazo reagents and strong acidic or basic additives. For global supply chain leaders and R&D directors, this represents a significant opportunity to optimize production costs while adhering to stricter environmental, health, and safety (EHS) regulations.
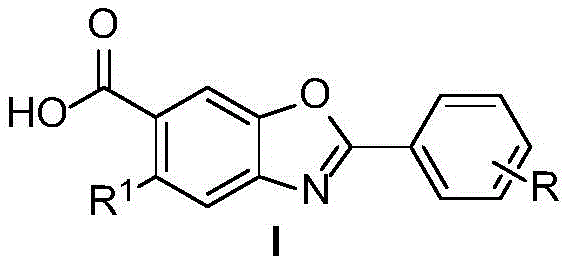
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Tafamidis and its analogues has been plagued by complex multi-step procedures that pose significant safety and cost challenges. The representative conventional method involves the acylation of aminohydroxybenzoic acid with dichlorobenzoyl chloride, followed by treatment with p-toluenesulfonic acid monohydrate. Crucially, this route necessitates the use of (trimethylsilyl)diazomethane for esterification and cyclization steps. Diazomethane derivatives are notoriously explosive and highly toxic, requiring specialized equipment and rigorous safety protocols that drive up operational expenditures. Furthermore, the reliance on strong acids and bases generates substantial hazardous waste, complicating disposal and increasing the overall cost of goods sold (COGS). These factors collectively limit the scalability of the process and hinder the rapid exploration of diverse derivatives needed for structure-activity relationship (SAR) studies.
The Novel Approach
In stark contrast, the novel synthetic route disclosed in the patent offers a streamlined, two-step process that bypasses these hazardous bottlenecks. By employing readily available catechol derivatives and substituted benzaldehydes as starting materials, the method constructs the benzoxazole core directly through an oxidative condensation reaction. The use of sodium periodate as the oxidant is particularly advantageous; it operates under mild conditions (80-100°C) and generates insoluble iodate byproducts that can be easily separated and recycled. This shift from a linear, hazard-heavy sequence to a convergent, green chemistry approach not only enhances the atom economy but also opens the door for the facile synthesis of a wide array of derivatives with different substituents on the benzoxazole skeleton, thereby accelerating drug discovery pipelines.
Mechanistic Insights into Sodium Periodate-Mediated Oxidative Cyclization
The core innovation of this technology lies in the efficient construction of the benzoxazole ring system via a sodium periodate-mediated oxidative coupling. In the first stage, a substituted catechol reacts with an aromatic aldehyde in the presence of ammonium acetate. The ammonium acetate serves as a nitrogen source, facilitating the formation of an imine intermediate which subsequently undergoes oxidative cyclization. Sodium periodate acts as a two-electron oxidant, driving the aromatization of the heterocyclic ring to form the stable benzoxazole structure. This mechanism is robust and tolerates a wide range of electronic environments, allowing for the incorporation of electron-withdrawing groups like nitro and trifluoromethyl, as well as electron-donating groups like methoxy and tert-butyl, without significant loss in yield.
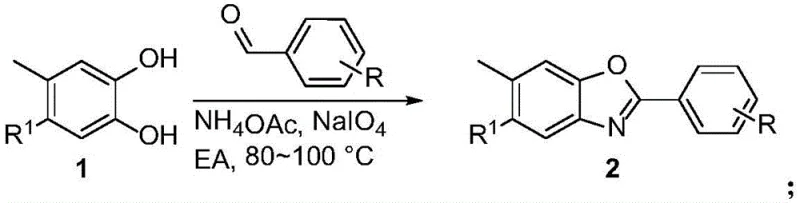
Following the formation of the benzoxazole core, the second critical transformation involves the oxidation of the methyl group at the 6-position to a carboxylic acid. This is achieved using potassium permanganate in a pyridine-water solvent system. The reaction proceeds through a radical mechanism where the benzylic hydrogen atoms are abstracted, eventually leading to the fully oxidized carboxyl functionality. The choice of pyridine as a co-solvent is crucial for solubilizing the organic intermediate while maintaining the reactivity of the permanganate ion. This step is highly selective, leaving the sensitive benzoxazole ring and other aromatic substituents intact, which is essential for maintaining the pharmacological integrity of the final Tafamidis derivative.
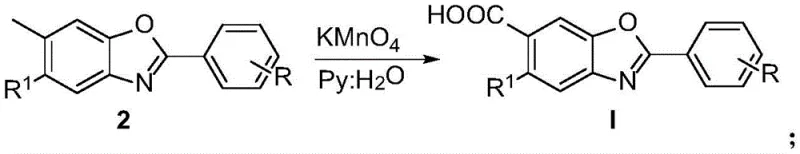
How to Synthesize Tafamidis Derivatives Efficiently
The implementation of this synthetic route requires precise control over reaction parameters to maximize yield and purity. The process begins with the condensation of specific catechol precursors, such as 4-tert-butyl-5-methylcatechol, with selected aldehydes in solvents like ethyl acetate or acetonitrile. Maintaining the reaction temperature between 80°C and 100°C under an inert atmosphere ensures optimal conversion while preventing side reactions. Following the isolation of the benzoxazole intermediate, the subsequent oxidation step demands careful monitoring of the stoichiometry of potassium permanganate to avoid over-oxidation or degradation of the product. Detailed standard operating procedures for scaling this chemistry from gram to kilogram scale are outlined below.
- Condense catechol compounds with aldehydes and ammonium acetate using sodium periodate as an oxidant in solvents like ethyl acetate at 80-100°C to form the benzoxazole core.
- Oxidize the methyl group on the benzoxazole intermediate to a carboxylic acid using potassium permanganate in a pyridine-water mixture at boiling temperature.
- Purify the final crude product via acidification, extraction, and column chromatography to obtain high-purity Tafamidis derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method translates into tangible strategic benefits beyond mere technical feasibility. The elimination of high-risk reagents like diazomethane removes the need for expensive blast-proof infrastructure and specialized handling training, directly lowering capital expenditure and insurance costs. Furthermore, the use of commodity chemicals such as catechols and benzaldehydes ensures a stable and diversified supply base, reducing the risk of raw material shortages that often plague proprietary reagent-dependent processes. The simplified work-up procedures, characterized by the precipitation of recyclable iodate salts, significantly reduce solvent consumption and waste treatment volumes, aligning perfectly with corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic impact of this route is driven primarily by the substitution of expensive and hazardous reagents with cost-effective alternatives. By avoiding dichlorobenzoyl chloride and trimethylsilyldiazomethane, manufacturers can eliminate the high costs associated with purchasing, storing, and disposing of these dangerous materials. Additionally, the ability to recycle the iodate byproduct further reduces the effective cost of the oxidant. The streamlined two-step process also reduces labor hours and utility consumption compared to the multi-step traditional route, resulting in substantial overall cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved due to the reliance on widely available bulk chemicals. Catechol derivatives and substituted benzaldehydes are produced by numerous global suppliers, preventing single-source bottlenecks. The robustness of the reaction conditions, which tolerate moisture and air better than sensitive organometallic or diazo chemistries, ensures consistent batch-to-batch quality even in large-scale production environments. This reliability allows for more accurate demand forecasting and inventory management, ensuring continuous availability of high-purity pharmaceutical intermediates for downstream API synthesis.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this green chemistry approach mitigates those concerns. The absence of strong acids and bases simplifies neutralization and wastewater treatment, making it easier to comply with increasingly stringent environmental regulations. The solid nature of the iodate byproduct facilitates easy filtration and potential reuse, minimizing the liquid waste stream. This environmental compatibility not only reduces regulatory compliance costs but also enhances the brand reputation of the manufacturer as a sustainable partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is critical for stakeholders evaluating its integration into existing production lines. The following questions address common concerns regarding reaction scope, safety profiles, and purification strategies. These insights are derived directly from the experimental data and claims within the patent documentation, providing a reliable foundation for technical decision-making.
Q: What are the key safety advantages of this new Tafamidis synthesis method?
A: This method eliminates the use of hazardous reagents such as dichlorobenzoyl chloride and (trimethylsilyl)diazomethane, which are required in traditional routes. It avoids strong acids, strong bases, and toxic diazo compounds, significantly reducing environmental hazards and operational risks.
Q: How does the use of sodium periodate improve atom economy?
A: Sodium periodate acts as a clean oxidant that facilitates the cyclization reaction efficiently. The byproduct, iodate, is insoluble in the reaction solution and can be easily recycled, minimizing waste generation and improving the overall atom economy of the process.
Q: Can this synthesis method tolerate diverse functional groups?
A: Yes, the method demonstrates excellent functional group tolerance. It successfully accommodates various substituents including halogens (F, Cl, Br, I), nitro groups, trifluoromethyl groups, and alkoxy groups on the aromatic rings, allowing for the creation of a diverse library of derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tafamidis Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this green synthetic route for the production of next-generation cardiovascular therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific solvent systems and oxidation chemistries required for this process, while our rigorous QC labs guarantee that every batch meets stringent purity specifications required for clinical and commercial applications.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this innovative technology for their pipeline projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and sustainability.
