Scalable Synthesis of Isoquinoline Tau Imaging Agents for Commercial PET Applications
Introduction to Next-Generation Tau Imaging Technology
The escalating global burden of Alzheimer's Disease (AD) has necessitated the rapid development of precise diagnostic tools capable of detecting neurofibrillary tangles formed by hyperphosphorylated tau protein. Patent CN107163022B represents a significant technological leap in this domain by disclosing a novel class of isoquinoline derivatives designed specifically for Positron Emission Tomography (PET). Unlike earlier generations of imaging agents which often suffered from weak signals or limited dynamic range, these new compounds demonstrate exceptional binding capacity and selectivity. The structural versatility of the isoquinoline core allows for fine-tuning of pharmacokinetic properties, ensuring better blood-brain barrier penetration and reduced background noise in clinical settings. As a leading manufacturer, we recognize the critical importance of securing a reliable supply chain for these high-value pharmaceutical intermediates to support the growing demand for early AD diagnosis.
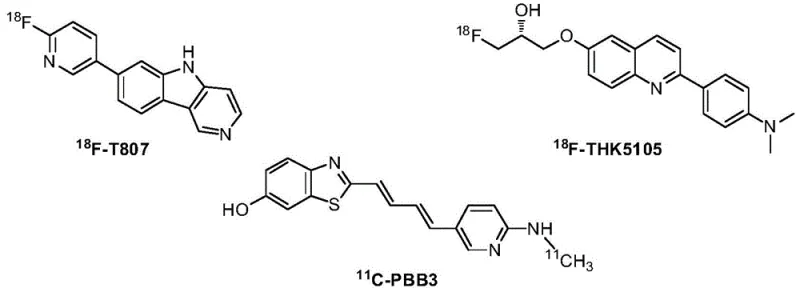
The strategic value of this technology lies not only in its diagnostic efficacy but also in its synthetic accessibility. The patent outlines robust pathways that transition smoothly from bench-scale discovery to commercial manufacturing. For procurement leaders and R&D directors alike, understanding the nuances of this synthesis is vital for mitigating supply risks associated with complex radiopharmaceuticals. By leveraging the modular design described in the intellectual property, manufacturers can produce a diverse library of analogues, facilitating structure-activity relationship (SAR) studies while maintaining strict control over impurity profiles. This report delves deep into the technical merits and commercial implications of adopting this advanced isoquinoline platform for your diagnostic pipeline.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tau-binding ligands has been plagued by linear, inefficient routes that rely heavily on harsh reaction conditions and difficult purification steps. Traditional methods often involve multiple protection and deprotection cycles, which drastically reduce overall yield and increase the generation of hazardous waste. Furthermore, many existing scaffolds lack the necessary chemical handles for efficient radiolabeling, forcing chemists to resort to low-yielding nucleophilic substitutions that compromise the specific activity of the final PET tracer. The reliance on expensive, scarce starting materials in older protocols creates significant bottlenecks in the supply chain, leading to extended lead times and volatile pricing structures. Additionally, the presence of residual heavy metal catalysts from cross-coupling reactions in conventional syntheses poses a severe regulatory hurdle, requiring extensive and costly downstream processing to meet stringent pharmaceutical purity standards.
The Novel Approach
In stark contrast, the methodology presented in CN107163022B utilizes a convergent synthetic strategy centered around the Suzuki-Miyaura cross-coupling reaction. This approach allows for the late-stage assembly of the complex bi-aryl architecture, significantly streamlining the production process. By employing a modular design where the isoquinoline core and the heterocyclic appendage are synthesized independently and then coupled, manufacturers gain unprecedented flexibility in generating diverse analogues without redesigning the entire synthetic route. The use of mild bases such as potassium carbonate and aqueous solvent systems minimizes environmental impact and simplifies workup procedures. Moreover, the inclusion of a dedicated nucleophilic fluorination pathway ensures that the introduction of the fluorine-18 isotope is both efficient and high-yielding, directly addressing the specific activity requirements for clinical PET imaging. This modern approach effectively decouples the complexity of the molecule from the difficulty of its synthesis.
Mechanistic Insights into Palladium-Catalyzed Cross-Coupling
The core of this innovative synthesis relies on the palladium-catalyzed Suzuki coupling between a brominated isoquinoline intermediate (Compound II) and a heteroaryl boronic ester (Compound III). Mechanistically, this transformation proceeds through a well-defined catalytic cycle involving oxidative addition, transmetallation, and reductive elimination. The use of zero-valent palladium complexes, specifically Pd(PPh3)4, facilitates the activation of the carbon-bromine bond on the isoquinoline ring under relatively mild thermal conditions (70-80°C). This step is critical as it generates the reactive organopalladium species necessary for bond formation. The subsequent transmetallation with the boronic ester, activated by the base, transfers the heterocyclic fragment to the palladium center. Finally, reductive elimination releases the coupled product and regenerates the active catalyst. This mechanism is highly tolerant of various functional groups, allowing for the incorporation of diverse substituents (R1-R5) without interfering with the reaction progress.
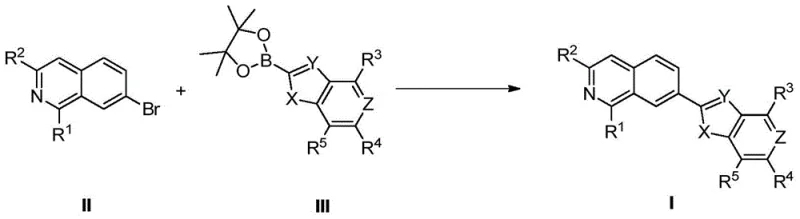
Impurity control is inherently built into this mechanistic framework. The high selectivity of the Suzuki reaction minimizes the formation of homocoupling byproducts, which are common pitfalls in other cross-coupling methodologies. Furthermore, the patent specifies the use of mixed solvent systems like DME and water, which enhance the solubility of inorganic bases and facilitate the separation of organic products from inorganic salts. The rigorous purification protocols described, including silica gel chromatography with specific mobile phase gradients, ensure that the final intermediates achieve purity levels exceeding 95%. For the radiolabeled variants, the mechanism shifts to nucleophilic aromatic substitution, where the electron-deficient nature of the nitro-substituted precursor activates the ring for attack by the fluoride ion. This electronic activation is key to achieving high radiochemical yields, ensuring that every atom of the expensive 18F isotope is utilized effectively in the final drug product.
How to Synthesize Isoquinoline Tau Imaging Agents Efficiently
The practical execution of this synthesis requires precise control over reaction parameters to maximize yield and purity. The process begins with the preparation of the key building blocks, followed by their convergence through palladium catalysis. Detailed operational guidelines regarding stoichiometry, temperature ramps, and quenching procedures are essential for reproducibility at scale. The following section outlines the standardized workflow derived from the patent examples, providing a clear roadmap for process chemists aiming to implement this technology in a GMP environment. Adhering to these optimized conditions ensures consistent quality and minimizes batch-to-batch variability.
- Prepare the bromo-isoquinoline intermediate (Compound II) and the boronic ester heterocycle (Compound III) via standard halogenation and borylation protocols.
- Perform a palladium-catalyzed Suzuki coupling reaction between Compound II and Compound III using Pd(PPh3)4 and K2CO3 in a DME/Water solvent system at 80°C.
- Execute nucleophilic fluorination on the resulting nitro-precursor using CsF or radioactive 18F- in DMF at elevated temperatures (120-150°C) to yield the final tracer.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this novel isoquinoline synthesis platform offers transformative benefits for procurement strategies and supply chain resilience. The shift from linear to convergent synthesis fundamentally alters the cost structure of producing these high-value diagnostics. By reducing the number of synthetic steps and utilizing robust, scalable reactions, manufacturers can significantly lower the cost of goods sold (COGS). This efficiency translates directly into more competitive pricing for the final imaging agents, making advanced Alzheimer's diagnostics more accessible to healthcare systems globally. Furthermore, the reliance on commercially available starting materials reduces dependency on custom synthesis vendors, thereby mitigating supply chain risks associated with raw material shortages.
- Cost Reduction in Manufacturing: The streamlined Suzuki coupling protocol eliminates the need for expensive protecting groups and harsh reagents, resulting in substantial savings on raw material costs. The ability to perform reactions in aqueous-organic mixtures reduces solvent consumption and waste disposal fees, contributing to a leaner manufacturing footprint. Additionally, the high yields reported in the patent examples mean less material is lost during production, maximizing the output from each batch of starting materials. This economic efficiency is crucial for maintaining profitability in the competitive radiopharmaceutical market while ensuring that cost does not become a barrier to patient access.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the stocking of key intermediates, enabling rapid response to fluctuating market demands. Since the coupling reaction is robust and tolerant of minor variations in conditions, the risk of batch failure is minimized, ensuring a consistent supply of critical intermediates. The use of stable boronic ester precursors further enhances shelf-life and logistics, as these materials can be stored and transported without special handling requirements. This reliability is paramount for PET centers that depend on just-in-time delivery of short-lived isotopes, as any delay in precursor availability can disrupt the entire imaging schedule.
- Scalability and Environmental Compliance: The reaction conditions described are inherently scalable, having been demonstrated to work efficiently from milligram to multi-gram scales without loss of performance. The avoidance of toxic heavy metals beyond the catalytic palladium, which is efficiently removed during workup, aligns with modern green chemistry principles and regulatory expectations. The simplified purification processes reduce the volume of organic waste generated, lowering the environmental burden and compliance costs associated with waste management. This sustainability profile not only meets corporate social responsibility goals but also future-proofs the manufacturing process against increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these isoquinoline compounds. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on feasibility and performance. Understanding these details is essential for stakeholders evaluating the integration of this technology into their existing portfolios.
Q: What is the primary advantage of this isoquinoline scaffold over existing Tau imaging agents?
A: The compounds disclosed in patent CN107163022B exhibit superior binding affinity (Ki < 10 nM) to hyperphosphorylated tau protein compared to prior art like T807, offering improved signal-to-noise ratios for PET imaging.
Q: Is the synthesis suitable for large-scale production of non-radioactive standards?
A: Yes, the modular Suzuki coupling strategy utilizes commercially available reagents and mild conditions (80°C), allowing for the efficient production of multi-gram quantities of reference standards required for QC and clinical trials.
Q: How is the radioactive 18F label introduced into the molecule?
A: The patent describes a late-stage nucleophilic fluorination step where a nitro-precursor is treated with no-carrier-added 18F- fluoride, ensuring high specific activity essential for in vivo diagnostic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoquinoline Compound Supplier
The development of effective Tau imaging agents represents a frontier in neurological diagnostics, and NINGBO INNO PHARMCHEM is positioned at the forefront of this innovation. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of isoquinoline intermediate meets the exacting standards required for radiopharmaceutical applications. Our commitment to quality ensures that the final PET tracers derived from our materials deliver the high signal-to-noise ratios necessary for accurate diagnosis.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with us, you gain access to specific COA data and route feasibility assessments that validate the industrial viability of this synthesis. Contact us today to discuss how we can support your Alzheimer's research and diagnostic programs with reliable, high-quality intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
