Revolutionizing Substituted Taurine Production: A Salt-Free Route for Commercial Scale-Up
Revolutionizing Substituted Taurine Production: A Salt-Free Route for Commercial Scale-Up
The landscape of fine chemical manufacturing is constantly evolving, driven by the urgent need for greener, more efficient synthetic pathways that minimize waste and maximize yield. A pivotal advancement in this domain is detailed in patent CN101851182A, which introduces a groundbreaking salt-free preparation method for substituted taurine. This technology represents a significant departure from traditional methodologies that rely heavily on inorganic sulfites or bisulfites, often resulting in complex purification challenges. By utilizing a strategic sequence of addition, oxidation, and reduction reactions starting from nitroolefins and thiocarboxylic acids, this novel approach eliminates the generation of inorganic salts entirely. For global pharmaceutical and agrochemical enterprises, this innovation offers a robust pathway to access high-purity intermediates essential for drug development and specialty chemical applications. The implications for supply chain stability and cost efficiency are profound, as the removal of desalination steps streamlines the entire production lifecycle.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of taurine and its substituted derivatives has been plagued by inherent inefficiencies associated with salt-based reagents. Traditional routes frequently employ sodium bisulfite or sulfite salts to introduce the sulfonic acid moiety, a process that inevitably generates stoichiometric amounts of inorganic salt byproducts. These salts create a significant burden on downstream processing, necessitating rigorous and often yield-diminishing desalination procedures such as extensive washing, ion exchange, or complex crystallization techniques. Furthermore, these conventional methods often suffer from limited substrate scope, struggling to accommodate diverse steric and electronic environments required for complex disubstituted or trisubstituted structures. The presence of residual inorganic ions can also compromise the purity profile of the final active pharmaceutical ingredient (API), posing risks for regulatory compliance in sensitive therapeutic applications. Consequently, manufacturers face elevated operational costs and extended lead times due to the laborious purification protocols required to meet stringent quality standards.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN101851182A leverages a purely organic synthetic strategy that circumvents the formation of inorganic salts altogether. By reacting readily available nitroolefins with thiocarboxylic acids, the process initiates a clean addition reaction to form a carboxylic acid nitro mercaptan ester. This intermediate is subsequently subjected to oxidation and reduction steps to yield the target substituted taurine. The absence of inorganic salt generation means that the cumbersome desalination purification processes are rendered obsolete, allowing for a much more direct isolation of the product. This streamlined workflow not only enhances the overall atom economy of the reaction but also significantly reduces the volume of wastewater generated, aligning with modern environmental sustainability goals. Moreover, this approach demonstrates exceptional versatility, successfully facilitating the preparation of cis and trans disubstituted and trisubstituted taurines, thereby expanding the chemical space accessible to medicinal chemists and process engineers alike.
Mechanistic Insights into the Three-Step Cascade Synthesis
The core of this innovative synthesis lies in a meticulously orchestrated three-step cascade that transforms simple starting materials into complex amino sulfonic acids. The journey begins with a Michael-type addition where the sulfur atom of the thiocarboxylic acid acts as a nucleophile, attacking the electron-deficient beta-carbon of the nitroolefin. This step is crucial as it establishes the carbon-sulfur bond while preserving the nitro group for subsequent transformation. Following the addition, the sulfide moiety undergoes a powerful oxidation event, typically mediated by organic peracids or amine N-oxides, which elevates the sulfur oxidation state to that of a sulfonic acid. This transformation is critical for imparting the characteristic acidity and solubility properties of taurine derivatives. Finally, the nitro group is selectively reduced to a primary amine, often via catalytic hydrogenation, completing the assembly of the substituted taurine scaffold. This sequential logic ensures that each functional group is installed with high precision, minimizing side reactions and maximizing the fidelity of the molecular architecture.
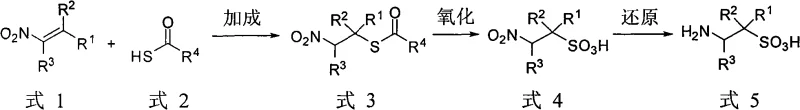
Beyond the primary reaction pathway, the mechanism offers sophisticated control over stereochemistry, a feature often lacking in bulk chemical synthesis. When the starting nitroolefin possesses substituents that allow for geometric isomerism, the resulting carboxylic acid nitro mercaptan ester intermediate exists as distinct cis and trans isomers. The patent highlights that these isomers can be effectively separated through standard techniques such as recrystallization or column chromatography prior to the final reduction step. This capability is invaluable for the production of chiral or stereodefined intermediates required for specific biological activities, such as enzyme inhibition or receptor binding. By isolating the stereoisomers early in the sequence, manufacturers can ensure that the final substituted taurine product possesses the exact spatial configuration needed for its intended application, whether it be as a nutrient, a surfactant, or a precursor for sulfonyl peptides. This level of control underscores the method's suitability for producing high-value fine chemicals where purity and structural integrity are paramount.
How to Synthesize Substituted Taurine Efficiently
Implementing this salt-free synthesis route requires a clear understanding of the operational parameters that drive high yields and purity. The process is designed to be operationally simple, utilizing commercially available raw materials that do not require exotic handling conditions. The initial addition reaction can be conducted in a variety of common organic solvents, including ethers, esters, or hydrocarbons, providing flexibility for process optimization. Following the formation of the intermediate, the oxidation and reduction steps are performed under controlled conditions to ensure complete conversion without degrading the sensitive amino acid backbone. For detailed technical specifications regarding reagent ratios, temperature profiles, and workup procedures, please refer to the standardized synthesis guide below which outlines the critical process parameters derived from the patent data.
- Perform a Michael addition reaction between a nitroolefin and a thiocarboxylic acid to form a carboxylic acid nitro mercaptan ester intermediate.
- Oxidize the sulfide intermediate using an organic peracid or amine N-oxide to convert the sulfur moiety into a sulfonic acid group.
- Reduce the nitro group of the oxidized intermediate using catalytic hydrogenation to yield the final substituted taurine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this salt-free technology translates directly into tangible operational improvements and risk mitigation. The most significant impact is observed in the simplification of the purification train; by eliminating the need for desalination, the number of unit operations is drastically reduced. This reduction not only shortens the overall batch cycle time but also decreases the consumption of utilities such as water and energy, which are major cost drivers in large-scale chemical manufacturing. Furthermore, the avoidance of inorganic salts minimizes the generation of saline wastewater, thereby lowering the burden on effluent treatment plants and reducing environmental compliance costs. These factors collectively contribute to a more resilient and cost-effective supply chain, enabling manufacturers to respond more agilely to market demands while maintaining competitive pricing structures for their clients.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive and time-consuming desalination steps. Traditional methods often incur significant losses of product during washing and filtration stages required to remove inorganic salts; by removing this requirement, the new method preserves yield and reduces raw material consumption per kilogram of finished product. Additionally, the use of simple, commercially available solvents and reagents avoids the premium costs associated with specialized catalysts or hard-to-source precursors. This streamlined approach allows for a substantial optimization of the cost of goods sold (COGS), making the production of substituted taurine derivatives financially viable even at smaller scales or for niche applications.
- Enhanced Supply Chain Reliability: From a sourcing perspective, the reliance on nitroolefins and thiocarboxylic acids offers a distinct advantage due to the broad availability of these feedstocks in the global chemical market. Unlike proprietary reagents that may be subject to single-source bottlenecks, these starting materials are produced by multiple suppliers, ensuring a stable and continuous flow of inputs. This diversity in the supply base mitigates the risk of production stoppages caused by raw material shortages. Moreover, the robustness of the reaction conditions means that the process is less susceptible to minor variations in reagent quality, further enhancing the reliability of supply for downstream customers who depend on consistent delivery schedules for their own manufacturing timelines.
- Scalability and Environmental Compliance: The design of this synthetic route is inherently scalable, making it ideally suited for transition from laboratory benchtop to multi-ton commercial production. The absence of solid inorganic byproducts prevents reactor fouling and piping blockages, which are common issues when scaling up salt-generating processes. This smooth scalability ensures that capacity can be ramped up quickly to meet surging demand without requiring major capital investment in new equipment. Environmentally, the process aligns with green chemistry principles by reducing waste generation and avoiding the discharge of high-salinity effluents. This compliance with increasingly strict environmental regulations future-proofs the manufacturing asset, reducing the risk of regulatory fines or shutdowns and enhancing the corporate sustainability profile of the organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this salt-free synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is critical for R&D teams evaluating the feasibility of this route for their specific product portfolios, as well as for procurement professionals assessing the long-term viability of the supply source. The answers below clarify the scope of the technology, its limitations, and its potential impact on product quality and regulatory filings.
Q: What is the primary advantage of the salt-free preparation method for substituted taurine?
A: The primary advantage is the elimination of inorganic salt byproducts, which removes the need for cumbersome and yield-reducing desalination purification steps, significantly simplifying the workflow for large-scale industrial production.
Q: Can this synthetic route produce stereoisomers of substituted taurine?
A: Yes, the method is highly versatile and capable of producing both cis and trans disubstituted and trisubstituted taurines. The cis-trans isomers of the intermediate carboxylic acid nitro mercaptan ester can be separated via recrystallization or chromatography before the final reduction step.
Q: What are the typical applications for the substituted taurine produced via this method?
A: The resulting compounds serve as critical building blocks for nutrients, pharmaceutical agents, enzyme inhibitors, antibacterial agents, surfactants, plant growth regulators, and precursors for synthesizing sulfonyl peptides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Taurine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the salt-free synthesis route described in patent CN101851182A for the production of high-value amino sulfonic acids. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory methodology into a robust, commercial-scale reality. Our facilities are equipped to handle complex organic syntheses, and we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to ensure that every batch of substituted taurine adheres to the highest international standards. Whether you require custom synthesis for drug discovery or bulk manufacturing for industrial applications, our team is ready to deploy this advanced chemistry to secure your supply chain.
We invite you to engage with our technical procurement team to discuss how this salt-free technology can be tailored to your specific project requirements. By leveraging our process development capabilities, we can provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener synthetic route for your operations. We encourage you to contact us today to request specific COA data for our existing taurine derivatives or to initiate a dialogue regarding route feasibility assessments for your novel targets. Let us partner with you to drive efficiency, sustainability, and innovation in your chemical supply chain.
