Revolutionizing S-Ibuprofen Production: Advanced Biocatalytic Resolution for Global Pharma Supply Chains
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for producing chiral active pharmaceutical ingredients (APIs), and the technology disclosed in patent CN101104861B represents a significant leap forward in the biosynthesis of S-ibuprofen. This patent details a novel catalytic synthesis method that utilizes extracellular lipase derived from Yarrowia lipolytica to resolve racemic ibuprofen into high-optical-purity S-ibuprofen and its corresponding esters. Unlike traditional chemical resolution methods that rely on expensive chiral resolving agents or complex asymmetric synthesis requiring precious metal catalysts, this biocatalytic approach leverages the inherent stereoselectivity of microbial enzymes. The process involves the esterification of racemic ibuprofen with various alcohols, followed by the separation of the desired S-enantiomer ester and subsequent hydrolysis. For R&D directors and procurement specialists, this technology offers a compelling alternative that promises to streamline manufacturing workflows while adhering to stricter environmental and safety standards prevalent in modern pharmaceutical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
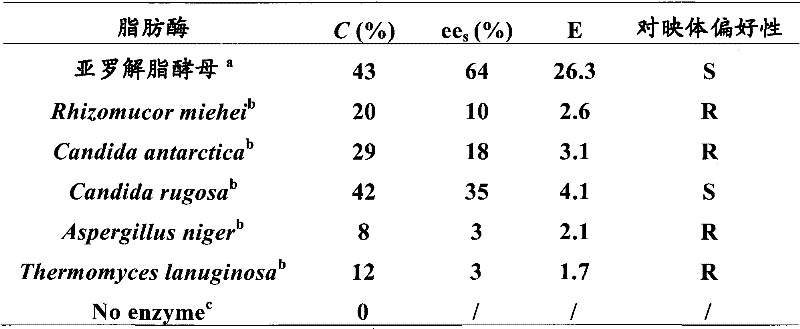
Historically, the production of single-enantiomer NSAIDs like S-ibuprofen has been plagued by significant technical and economic hurdles associated with conventional resolution techniques. Traditional chemical resolution often necessitates the use of stoichiometric amounts of chiral acids or bases, which not only drives up raw material costs but also generates substantial quantities of salt waste that require complex disposal procedures. Furthermore, asymmetric chemical synthesis, while elegant in theory, frequently depends on transition metal catalysts that pose risks of heavy metal contamination in the final drug product, necessitating rigorous and costly purification steps to meet regulatory limits. Existing biotechnological methods using lipases from sources such as Rhizomucor miehei or Candida rugosa have also shown limitations, often exhibiting low catalytic conversion rates and poor stereoselectivity, resulting in suboptimal yields of the desired S-isomer. These inefficiencies create bottlenecks in the supply chain, leading to longer lead times and higher overall production costs for manufacturers relying on outdated technologies.
The Novel Approach
The innovative method presented in the patent data overcomes these historical barriers by employing extracellular lipase from Yarrowia lipolytica, a yeast strain recognized for its safety and robustness in industrial fermentation. This novel approach achieves remarkably high stereoselectivity, with experimental data indicating enantiomeric excess (ee) values and E-values that far surpass those of competing enzymatic systems. By optimizing reaction parameters such as solvent choice and temperature, the process ensures that the S-enantiomer is preferentially esterified while the R-enantiomer remains largely unreacted, facilitating a clean separation. The use of immobilized enzyme forms further enhances the economic viability of the process by allowing for catalyst recovery and reuse, thereby reducing the per-batch cost of the biocatalyst. This shift towards a highly selective, bio-based resolution strategy not only simplifies the downstream purification process but also aligns with the growing industry demand for green chemistry solutions that minimize environmental impact.
Mechanistic Insights into Lipase-Catalyzed Kinetic Resolution
The core of this technological advancement lies in the specific interaction between the Yarrowia lipolytica lipase and the racemic ibuprofen substrate within a non-aqueous organic medium. The enzyme functions through a kinetic resolution mechanism where it selectively recognizes and catalyzes the esterification of the S-enantiomer of ibuprofen with short-chain or long-chain alcohols. The active site of the lipase possesses a chiral environment that sterically hinders the binding of the R-enantiomer, effectively discriminating between the two mirror-image molecules. This discrimination is quantified by the enantiomeric ratio (E), where higher values indicate greater selectivity; the patent data highlights that under optimized conditions, particularly with the addition of crown ether additives like 18-crown-6, the E-value can reach exceptional levels, ensuring that the resulting ester pool is enriched almost exclusively with the pharmacologically active S-form. Understanding this mechanistic specificity is crucial for process chemists aiming to replicate these results, as it dictates the strict control needed over reaction variables to maintain high optical purity.

Temperature control plays a pivotal role in modulating the activity and selectivity of the biocatalyst, as illustrated by the relationship between thermal conditions and reaction performance. The data indicates that while higher temperatures generally increase reaction kinetics, there is an optimal window, typically around 40°C, where the enzyme maintains its structural integrity and stereoselective capability without denaturation. Deviating from this range can lead to a drop in the E-value, compromising the optical purity of the product. Similarly, the molar ratio of the substrates—racemic ibuprofen to alcohol—is a critical parameter that influences the equilibrium of the esterification reaction. Maintaining a balanced ratio, often favoring a slight excess of alcohol, helps drive the reaction forward without inhibiting the enzyme or promoting non-selective background reactions. These mechanistic nuances underscore the importance of precise process engineering to fully exploit the catalytic potential of the Yarrowia lipolytica system.
How to Synthesize S-Ibuprofen Efficiently
To implement this high-efficiency synthesis route, manufacturers must adhere to a standardized protocol that maximizes the yield of the S-enantiomer while minimizing impurities. The process begins with the preparation of a reaction mixture containing racemic ibuprofen and a selected alcohol, such as ethanol or n-heptanol, dissolved in a hydrophobic organic solvent like n-hexane or isooctane. The choice of solvent is critical, as solvents with a Log P value greater than 3 are preferred to preserve enzyme activity and prevent the stripping of essential water molecules from the enzyme's surface. Once the substrate solution is prepared, the immobilized or free extracellular lipase from Yarrowia lipolytica is introduced, optionally alongside phase-transfer catalysts or crown ethers to boost selectivity. The detailed standardized synthesis steps see the guide below.
- Prepare the reaction system by mixing racemic ibuprofen and alcohol (e.g., ethanol or n-heptanol) in an organic solvent with Log P ≥ 3, such as n-hexane or isooctane.
- Add extracellular lipase from Yarrowia lipolytica (either as crude powder or immobilized form) and optional additives like 18-crown-6 to enhance stereoselectivity.
- Incubate the mixture at 25-40°C for 24-48 hours, then separate the S-ibuprofen ester using alkaline extraction and hydrolyze it to obtain high-purity S-ibuprofen.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Yarrowia lipolytica-based biocatalytic process offers transformative benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the production workflow, which eliminates the need for hazardous chemical resolving agents and the complex waste treatment associated with them. This streamlining translates directly into operational efficiencies, as fewer unit operations are required to achieve the same level of purity compared to traditional methods. Furthermore, the robustness of the yeast-derived enzyme ensures consistent batch-to-batch quality, reducing the risk of production delays caused by catalyst variability. By securing a reliable S-ibuprofen supplier who utilizes this advanced technology, companies can mitigate supply chain risks associated with volatile raw material markets and stringent environmental regulations, ensuring a steady flow of high-quality intermediates for downstream drug formulation.
- Cost Reduction in Manufacturing: The implementation of this enzymatic route leads to substantial cost savings by removing the dependency on expensive chiral chemical auxiliaries and precious metal catalysts. Since the lipase can be produced via fermentation using cost-effective substrates and potentially reused through immobilization, the overall cost of goods sold (COGS) for the API intermediate is significantly lowered. Additionally, the high stereoselectivity reduces the burden on downstream purification processes like chromatography or recrystallization, which are often the most energy and solvent-intensive steps in pharmaceutical manufacturing. This holistic reduction in material and energy consumption creates a leaner, more profitable production model.
- Enhanced Supply Chain Reliability: Utilizing a GRAS (Generally Recognized As Safe) organism like Yarrowia lipolytica ensures that the biocatalyst source is stable, scalable, and free from regulatory hurdles associated with pathogenic strains. This biological stability guarantees a continuous supply of the enzyme, preventing bottlenecks that often occur when relying on specialized chemical reagents with long lead times. Moreover, the process operates under mild conditions (ambient pressure and moderate temperatures), reducing the reliance on specialized high-pressure equipment and lowering the barrier for contract manufacturing organizations (CMOs) to adopt the technology. This accessibility broadens the potential supplier base, enhancing the resilience of the global supply network for NSAID intermediates.
- Scalability and Environmental Compliance: The transition to this biocatalytic method aligns perfectly with global sustainability goals by minimizing the generation of toxic waste and reducing the carbon footprint of the manufacturing process. The use of recyclable organic solvents and the absence of heavy metals simplify wastewater treatment and discharge compliance, avoiding costly fines and remediation efforts. From a scalability perspective, the fermentation-based production of the enzyme and the straightforward reaction setup allow for seamless scale-up from pilot batches to multi-ton commercial production without losing efficiency. This scalability ensures that the technology can meet the growing global demand for S-ibuprofen without compromising on environmental standards or product quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented biocatalytic technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on reaction optimization and product specifications. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: Why is Yarrowia lipolytica lipase superior to other enzymes for ibuprofen resolution?
A: According to patent CN101104861B, lipase from Yarrowia lipolytica demonstrates significantly higher stereoselectivity (E values up to 94.87 with additives) compared to conventional enzymes like Rhizomucor miehei or Candida rugosa, which often show poor selectivity (E < 5).
Q: What are the optimal reaction conditions for maximizing S-ibuprofen yield?
A: The patent indicates that using organic solvents with Log P ≥ 3 (like isooctane), maintaining temperatures around 40°C, and utilizing a substrate molar ratio of 1:2 (acid:alcohol) provides the best balance of conversion and enantioselectivity.
Q: Is this biocatalytic process suitable for large-scale industrial production?
A: Yes, the process utilizes GRAS (Generally Recognized As Safe) yeast strains and robust immobilization techniques, allowing for enzyme reuse and simplified downstream processing, which are critical factors for scalable commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-Ibuprofen Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthesis technologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has extensively studied the Yarrowia lipolytica lipase pathway and possesses the technical expertise to scale this complex biocatalytic route from laboratory benchtop to full commercial production. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-purity intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to verify stringent purity specifications, guaranteeing that every batch of S-ibuprofen meets the highest international pharmacopoeia standards.
We invite potential partners to engage with our technical procurement team to discuss how this innovative biocatalytic process can be tailored to your specific production needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this enzymatic resolution method. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will optimize your supply chain and enhance your product portfolio's profitability.
