Advanced Biocatalytic Production of Crizotinib Intermediates for Global Pharmaceutical Supply Chains
Advanced Biocatalytic Production of Crizotinib Intermediates for Global Pharmaceutical Supply Chains
The pharmaceutical industry is currently witnessing a paradigm shift towards greener, more sustainable manufacturing processes, particularly for high-value oncology drugs like Crizotinib. A pivotal development in this sector is documented in patent CN108285908B, which details a novel method for synthesizing (S)-1-(2,6-dichloro-3-fluoro-phenyl) ethanol, a critical chiral intermediate. This patent introduces a sophisticated immobilized dual-enzyme catalytic system that overcomes the significant limitations of traditional chemical synthesis. By leveraging calcium phosphate-enzyme crystals in synergy with coenzyme NADP+, this technology enables the production of optically pure intermediates under mild conditions. For R&D directors and procurement specialists, this represents a transformative opportunity to enhance supply chain resilience while adhering to stringent environmental regulations. The ability to achieve yields of 95% with 100% enantiomeric excess (ee) positions this biocatalytic route as a superior alternative for commercial-scale API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral alcohol intermediates for kinase inhibitors has relied heavily on transition metal-catalyzed asymmetric hydrogenation. As illustrated in prior art such as Chinese patent CN103319311A, these conventional routes typically employ expensive Iridium-based chiral catalysts and require harsh reaction conditions. The process often necessitates high-pressure hydrogen atmospheres, frequently around 10 atm, which imposes severe safety constraints and requires specialized, costly reactor infrastructure. Furthermore, the use of strong alkalis like potassium tert-butoxide creates significant challenges in waste treatment and post-reaction processing. The reliance on precious metals not only inflates the raw material costs but also introduces the risk of heavy metal contamination in the final Active Pharmaceutical Ingredient (API), necessitating additional purification steps to meet regulatory limits. These factors collectively result in a complex, high-cost manufacturing workflow that is difficult to scale efficiently.
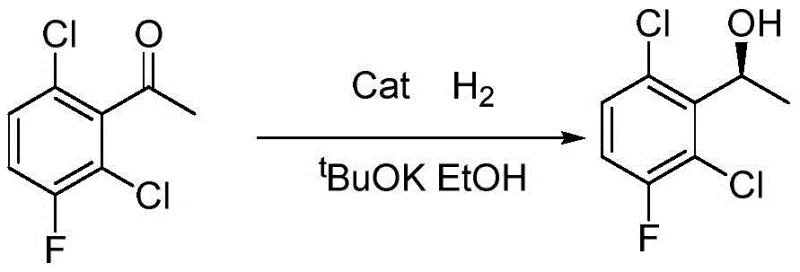
The Novel Approach
In stark contrast, the methodology disclosed in CN108285908B utilizes a bio-engineered approach that eliminates the need for high-pressure equipment and toxic heavy metals. By employing a dual-enzyme system comprising aldehyde ketone reductase and alcohol dehydrogenase, immobilized within a robust calcium phosphate matrix, the reaction proceeds smoothly at atmospheric pressure and moderate temperatures ranging from 30 to 60°C. This shift from chemocatalysis to biocatalysis fundamentally alters the economic and safety profile of the synthesis. The immobilization technique ensures that the enzymes retain high activity while gaining the mechanical stability required for industrial handling. This allows for a simplified downstream processing workflow where the catalyst can be easily separated via filtration, washed, and potentially recycled. The result is a streamlined process that significantly reduces the environmental footprint and operational complexity associated with producing this key pharmaceutical building block.
Mechanistic Insights into Immobilized Dual-Enzyme Catalysis
The core innovation of this technology lies in the synergistic action of two distinct enzymes working in tandem to drive the reduction and cofactor regeneration cycles. The primary catalyst, aldehyde ketone reductase (AKR), is responsible for the stereoselective reduction of the ketone substrate, 2,6-dichloro-3-fluoroacetophenone, into the desired (S)-alcohol. This step consumes the reduced cofactor NADPH, oxidizing it to NADP+. To make the process economically viable, the expensive cofactor must be regenerated in situ. This is achieved by the second enzyme, alcohol dehydrogenase (ADH), which oxidizes a sacrificial alcohol donor, typically isopropanol, back into acetone, simultaneously reducing NADP+ back to NADPH. This closed-loop cofactor recycling ensures that only catalytic amounts of NADP+ are required, drastically lowering material costs. The calcium phosphate crystal matrix serves as a protective scaffold, preventing enzyme denaturation and facilitating mass transfer.
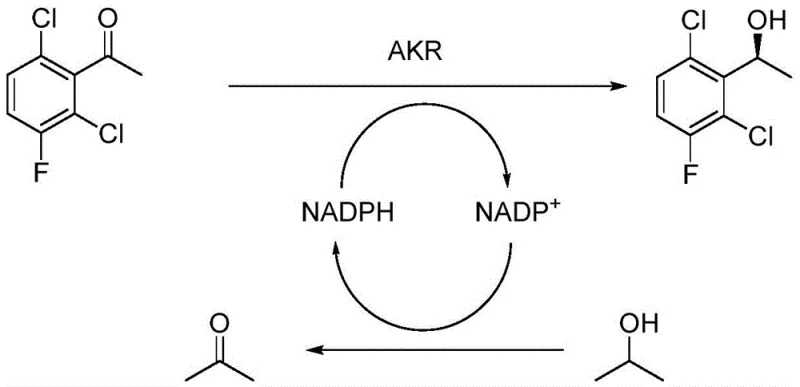
From an impurity control perspective, the high specificity of the enzymatic active sites offers unparalleled selectivity compared to small-molecule catalysts. The enzyme's chiral pocket strictly accommodates the substrate in a specific orientation, ensuring that only the (S)-enantiomer is produced, thereby achieving 100% ee without the formation of the unwanted (R)-isomer. This intrinsic selectivity minimizes the formation of diastereomeric impurities that are common in chemical hydrogenation. Furthermore, the mild aqueous-organic solvent system used in this process prevents side reactions such as dehalogenation or elimination, which can occur under the strongly basic conditions of traditional synthesis. The stability of the calcium phosphate-enzyme crystal also means that the catalytic performance remains consistent over extended reaction times, leading to reproducible batch quality and simplified quality control protocols for the final intermediate.
How to Synthesize (S)-1-(2,6-dichloro-3-fluoro-phenyl) ethanol Efficiently
Implementing this biocatalytic route requires precise control over reaction parameters to maximize yield and optical purity. The process begins with the preparation of the immobilized catalyst, where calcium chloride and phosphate buffer are mixed with the specific enzymes to form white precipitate crystals. These crystals are then introduced into a reaction vessel containing the ketone substrate and the cofactor system. The reaction is typically conducted in a biphasic or mixed solvent system involving potassium phosphate buffer and isopropanol, which serves both as a cosolvent for the organic substrate and the hydrogen donor for cofactor regeneration. Maintaining the temperature between 30°C and 60°C is critical; lower temperatures may slow the reaction kinetics, while higher temperatures could risk enzyme denaturation despite the immobilization. Following the reaction period of 6 to 24 hours, the solid catalyst is removed by filtration, and the product is extracted into an organic phase, dried, and concentrated to yield the high-purity alcohol.
- Prepare the reaction system by dissolving 2,6-dichloro-3-fluoroacetophenone and coenzyme NADP+ in a solvent mixture of potassium phosphate buffer and isopropanol.
- Add the immobilized calcium phosphate-enzyme crystals containing aldehyde ketone reductase and alcohol dehydrogenase to the mixture.
- Maintain the reaction at 30-60°C with shaking for 6-24 hours, then filter, extract with n-heptane, and dry to isolate the optically pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this immobilized enzymatic process offers substantial strategic benefits beyond mere technical feasibility. The most immediate impact is seen in the drastic simplification of the supply chain for raw materials. By removing the dependency on scarce and volatile precious metals like Iridium, manufacturers can insulate themselves from geopolitical supply shocks and price fluctuations in the commodities market. Additionally, the elimination of high-pressure hydrogenation steps removes the need for specialized autoclaves and rigorous safety certifications, allowing production to be scaled in standard stainless steel reactors. This flexibility significantly reduces capital expenditure (CAPEX) for new production lines and lowers the barrier to entry for contract manufacturing organizations (CMOs) looking to offer this intermediate. The overall process intensity is reduced, leading to a leaner, more responsive manufacturing operation.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling, primarily driven by the elimination of expensive chiral ligands and transition metals. In traditional chemical synthesis, the cost of the Iridium catalyst and its associated chiral phosphine ligands constitutes a significant portion of the bill of materials. By replacing this with immobilized enzymes, which can be produced via fermentation and reused multiple times due to their stability, the variable cost per kilogram of the product is significantly lowered. Furthermore, the mild reaction conditions reduce energy consumption for heating and cooling, and the simplified workup procedure reduces solvent usage and waste disposal costs. These cumulative efficiencies translate into a more competitive pricing structure for the final API, providing a clear margin advantage for downstream drug manufacturers.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for pharmaceutical sourcing, and this biocatalytic route enhances reliability through catalyst stability. Free enzymes are often sensitive to temperature and shear forces, making them difficult to transport and store. However, the calcium phosphate immobilization technique converts the biocatalyst into a robust solid powder that can be shipped and stored with standard logistics, similar to traditional chemical reagents. This stability ensures that the catalyst supply is not a bottleneck in the production schedule. Moreover, the high conversion rates and 100% optical purity reduce the risk of batch failures or the need for reprocessing, ensuring a steady flow of qualified material to the next stage of synthesis. This predictability allows supply chain planners to optimize inventory levels and reduce safety stock requirements.
- Scalability and Environmental Compliance: As regulatory bodies worldwide tighten restrictions on pharmaceutical waste and solvent emissions, the green credentials of this process become a major asset. The aqueous-based nature of the enzymatic reaction minimizes the volume of hazardous organic solvents required compared to traditional organic synthesis. The absence of heavy metal residues simplifies the wastewater treatment process and ensures compliance with strict limits on elemental impurities (ICH Q3D). From a scalability standpoint, the exothermic nature of enzymatic reactions is generally easier to manage than high-pressure hydrogenation, reducing the risk of thermal runaways during scale-up from pilot to commercial tons. This makes the technology highly suitable for large-scale production facilities aiming to meet global demand for Crizotinib while maintaining a sustainable corporate image.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this immobilized dual-enzyme technology. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear understanding of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific manufacturing needs. The answers highlight the robustness of the catalyst system and the high quality of the resulting intermediate.
Q: How does the immobilized enzyme method compare to traditional chemical hydrogenation in terms of safety?
A: Unlike traditional methods requiring high-pressure hydrogen (10 atm) and strong bases like potassium tert-butoxide, this enzymatic process operates at atmospheric pressure and mild temperatures (30-60°C), significantly reducing operational hazards and equipment costs.
Q: What is the optical purity achievable with this dual-enzyme system?
A: The patented method utilizing calcium phosphate-enzyme crystals achieves exceptional stereoselectivity, consistently delivering products with 100% ee (enantiomeric excess) and conversion rates up to 95%, eliminating the need for complex chiral resolution steps.
Q: Can the biocatalyst be reused for multiple batches?
A: Yes, the immobilization of enzymes within calcium phosphate crystals enhances their stability and facilitates easy separation via filtration, allowing the catalyst to be recovered and reused, which drastically lowers the cost per kilogram of the final intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-1-(2,6-dichloro-3-fluoro-phenyl) ethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced, sustainable technologies to meet the evolving demands of the global pharmaceutical market. Our team of expert process chemists has extensively evaluated the immobilized dual-enzyme pathway described in CN108285908B and possesses the technical expertise to implement this route effectively. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee 100% ee for every batch of (S)-1-(2,6-dichloro-3-fluoro-phenyl) ethanol we produce. We are committed to delivering high-quality intermediates that support the uninterrupted manufacture of life-saving oncology therapies.
We invite potential partners to engage with our technical procurement team to discuss how this innovative biocatalytic process can be integrated into your supply chain. By collaborating with us, you can leverage our manufacturing capabilities to achieve significant operational efficiencies. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized production of this key Crizotinib intermediate can drive value for your organization.
