Advanced Two-Step Synthesis of Single Nitrogen Heterocyclic Compounds for Commercial Scale-Up
Advanced Two-Step Synthesis of Single Nitrogen Heterocyclic Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient, environmentally benign pathways to access critical nitrogen-containing scaffolds. Patent CN105753768B introduces a transformative production method for single nitrogen heterocyclic compounds, leveraging cheap and readily available lactones and fatty amines as primary feedstocks. This technology represents a significant departure from traditional synthesis routes, offering a streamlined two-step process that combines ammoniation displacement with subsequent hydrogenation and deoxygenation. By utilizing a totally enclosed process flow, the method ensures continuous industrial production capabilities while minimizing the generation of high-concentration salt wastes, addressing key pain points in modern chemical manufacturing regarding safety and environmental protection. The innovation lies not only in the feedstock selection but also in the discovery of key control parameters that optimize energy consumption and maximize yield across a diverse range of heterocyclic structures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of single nitrogen heterocyclic compounds such as tetrahydropyrrole and tetrahydroquinoline has relied on methods that present substantial operational and environmental challenges. Common prior art techniques often involve the direct condensation of tetrahydrofuran with liquid ammonia, which requires severe reaction conditions and frequently suffers from moderate selectivity issues. Alternative routes utilizing dihalogenated hydrocarbons and amines generate stoichiometric amounts of inorganic salts, creating a massive burden on wastewater treatment facilities and increasing the overall cost of goods sold. Furthermore, existing hydrodeoxygenation processes frequently employ copper-chromium catalysts; while effective, the presence of chromium introduces severe toxicity concerns, necessitating expensive removal steps and complex disposal protocols to meet stringent global environmental regulations. These legacy methods often struggle to balance high purity requirements with economic feasibility, particularly when scaling to multi-ton quantities where waste management becomes a dominant cost factor.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes lactones as the foundational building blocks, reacting them with primary aliphatic amines or ammonia to first synthesize an intermediate amide, referred to as the Type I compound. This initial step is followed by a highly selective hydrogenation and deoxygenation reaction to yield the final nitrogen heterocycle, effectively removing the oxygen atom from the lactone ring structure. 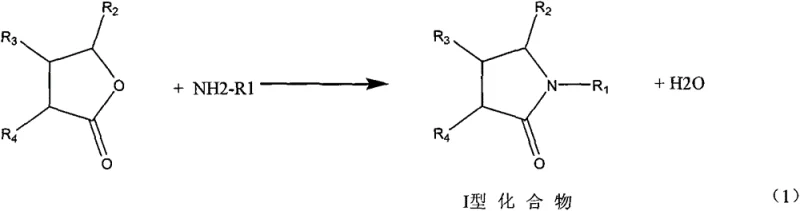 This two-step sequence is characterized by its simplicity and high atom economy, avoiding the generation of difficult-to-treat by-products. The process is designed for continuity, utilizing tubular reactors that enhance heat and mass transfer efficiency compared to traditional batch kettle systems. By eliminating the need for toxic chromium catalysts and reducing the formation of inorganic salts, this method offers a greener alternative that aligns with modern sustainability goals while maintaining high throughput and product quality standards essential for reliable pharmaceutical intermediate supplier operations.
This two-step sequence is characterized by its simplicity and high atom economy, avoiding the generation of difficult-to-treat by-products. The process is designed for continuity, utilizing tubular reactors that enhance heat and mass transfer efficiency compared to traditional batch kettle systems. By eliminating the need for toxic chromium catalysts and reducing the formation of inorganic salts, this method offers a greener alternative that aligns with modern sustainability goals while maintaining high throughput and product quality standards essential for reliable pharmaceutical intermediate supplier operations.
Mechanistic Insights into Lactone Ammoniation and Hydrodeoxygenation
The core of this technology rests on a sophisticated understanding of heterogeneous catalysis applied to ring-opening and ring-closing mechanisms. In the first stage, the lactone undergoes an ammoniation displacement reaction where the cyclic ester bond is cleaved and reformed into a cyclic amide (lactam) structure under elevated temperatures ranging from 150 to 270°C. The use of specific heterogeneous catalysts, such as super acids or basic ion exchange resins, allows this transformation to proceed under milder pressures compared to uncatalyzed thermal reactions, reducing the energy intensity of the process. The second stage involves the hydrodeoxygenation of the Type I compound, where a specialized copper-based catalyst facilitates the removal of the carbonyl oxygen. 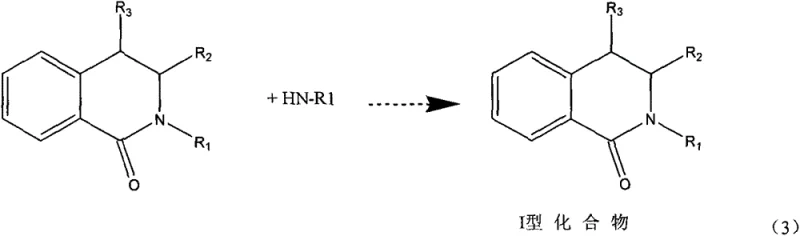 This step is critical, as it converts the stable amide bond into the desired amine functionality without breaking the carbon-nitrogen ring structure, a common side reaction in less selective systems. The catalyst formulation, comprising copper oxide promoted by zinc, tin, and alkaline earth metal oxides, is engineered to maximize activity while suppressing ring-opening degradation, ensuring that the heterocyclic integrity is preserved throughout the reduction.
This step is critical, as it converts the stable amide bond into the desired amine functionality without breaking the carbon-nitrogen ring structure, a common side reaction in less selective systems. The catalyst formulation, comprising copper oxide promoted by zinc, tin, and alkaline earth metal oxides, is engineered to maximize activity while suppressing ring-opening degradation, ensuring that the heterocyclic integrity is preserved throughout the reduction.
Impurity control is another pillar of this mechanistic design, specifically regarding the management of water and free amine content in the intermediate stream. The inventors discovered that maintaining a specific residual amount of free amine and water in the crude Type I compound prior to hydrogenation surprisingly improves the selectivity of the subsequent reduction step. Too little water can deactivate the hydrogenation catalyst, while too much can lead to darker product color and reduced activity; thus, the process employs a precise flash separation technique to tune these parameters within an optimal window.  Furthermore, the final purification utilizes vacuum rectification combined with molecular sieve drying or membrane separation, which effectively breaks azeotropes and removes trace moisture without the need for energy-intensive extractive distillation solvents. This rigorous control over the impurity profile ensures that the final high-purity nitrogen heterocycles meet the stringent specifications required for downstream API synthesis, minimizing the risk of catalyst poisoning in subsequent customer processes.
Furthermore, the final purification utilizes vacuum rectification combined with molecular sieve drying or membrane separation, which effectively breaks azeotropes and removes trace moisture without the need for energy-intensive extractive distillation solvents. This rigorous control over the impurity profile ensures that the final high-purity nitrogen heterocycles meet the stringent specifications required for downstream API synthesis, minimizing the risk of catalyst poisoning in subsequent customer processes.
How to Synthesize Single Nitrogen Heterocyclic Compounds Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing various heterocycles, from pyrrolidines to tetrahydroisoquinolines, with consistent quality. The process begins with the continuous feeding of lactone and amine reactants into a heated tubular reactor packed with a heterogeneous catalyst, where residence time and temperature are tightly controlled to ensure complete conversion to the Type I intermediate. Following the initial reaction, the stream undergoes flash separation to remove excess light components, preparing the crude intermediate for the critical hydrogenation stage in a second tubular reactor. For a comprehensive breakdown of the specific operating parameters, catalyst loading densities, and separation sequences required to replicate this high-yield process, please refer to the standardized technical guide below.
- React lactone with aliphatic amine or ammonia in a tubular reactor using a heterogeneous catalyst at 150-270°C to form Type I amide intermediates.
- Separate light components via flash tank to obtain crude Type I compound with controlled water and free amine content.
- Hydrogenate the Type I compound over a chromium-free copper-based catalyst at 200-280°C to yield the final nitrogen heterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this lactone-based synthesis route offers compelling strategic advantages that extend beyond simple unit cost metrics. The shift away from chromium-based catalysis fundamentally alters the waste management profile of the facility, eliminating the need for costly hazardous waste disposal services associated with heavy metal contamination. This transition not only reduces regulatory compliance risks but also simplifies the supply chain for waste treatment vendors, ensuring uninterrupted production schedules even under tightening environmental audits. Additionally, the use of continuous tubular reactors enhances equipment utilization rates, allowing for a smaller physical footprint to achieve the same output volume as larger batch facilities, which translates to lower capital expenditure and overhead costs per kilogram of product manufactured.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts containing chromium significantly lowers the raw material cost base while simultaneously removing the downstream expenses related to metal scavenging and purification. By utilizing cheap and widely available lactones and fatty amines as starting materials, the process insulates the supply chain from volatility in specialty reagent markets, ensuring stable pricing structures for long-term contracts. The high selectivity of the reaction minimizes the formation of by-products, which reduces the load on separation units and lowers utility consumption for distillation and solvent recovery. Consequently, the overall cost of goods sold is drastically optimized, providing a competitive edge in price-sensitive markets without compromising on the quality of the fine chemical intermediates supplied.
- Enhanced Supply Chain Reliability: The reliance on commodity-grade feedstocks such as lactones and ammonia ensures a robust and diversified supply base, reducing the risk of shortages that often plague manufacturers dependent on niche synthetic precursors. The continuous nature of the process allows for flexible production scheduling, enabling rapid response to fluctuating market demands and shortening the lead time for high-purity pharmaceutical intermediates. Furthermore, the stability of the heterogeneous catalysts extends their operational lifespan, reducing the frequency of reactor shutdowns for catalyst change-outs and maintenance. This operational continuity guarantees a steady flow of materials to downstream customers, reinforcing the reliability of the supplier partnership and supporting just-in-time inventory strategies.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction parameters that translate linearly from pilot plants to multi-ton commercial reactors, mitigating the technical risks usually associated with technology transfer. The absence of high-concentration salt waste and toxic heavy metals simplifies the environmental permitting process, facilitating faster expansion into new geographic regions with strict ecological regulations. Energy efficiency is further enhanced through heat integration between the exothermic hydrogenation step and the endothermic vaporization requirements, lowering the carbon footprint of the manufacturing site. This alignment with green chemistry principles not only future-proofs the production asset against evolving regulatory landscapes but also appeals to end-users seeking sustainable sourcing options for their own ESG reporting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented production method. These insights are derived directly from the experimental data and process descriptions found in the patent documentation, providing clarity on how this technology compares to incumbent methods. Understanding these details is crucial for technical teams evaluating the feasibility of integrating these intermediates into their own synthesis pipelines or for procurement specialists assessing the long-term viability of the supply source.
Q: What are the primary advantages of the lactone-based route over traditional tetrahydrofuran amination?
A: The lactone-based route described in CN105753768B avoids the harsh conditions and lower selectivity often associated with direct THF amination, utilizing milder heterogeneous catalysis and achieving yields over 95% without toxic chromium byproducts.
Q: How does the new copper-based catalyst improve environmental compliance?
A: By replacing traditional copper-chromium catalysts with a specialized copper-zinc-oxide formulation, the process eliminates hexavalent chromium waste, significantly reducing hazardous waste treatment costs and environmental impact.
Q: Is this process suitable for continuous industrial production?
A: Yes, the patent explicitly designs the method for continuous tubular reactors, allowing for steady-state operation, consistent product quality, and easier scale-up from pilot to commercial tonnage compared to batch processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Single Nitrogen Heterocyclic Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to maintain competitiveness in the global fine chemicals market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN105753768B are fully realized in practical, large-scale operations. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of single nitrogen heterocyclic compound meets the exacting standards required for pharmaceutical and agrochemical applications. Our commitment to process excellence means we can deliver these complex intermediates with the consistency and reliability that global supply chains demand.
We invite you to engage with our technical procurement team to discuss how this innovative lactone-based route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this greener, more efficient manufacturing method for your specific application. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your upstream supply chain.
