Advanced Four-Step Synthesis of 5-Methoxyindole for Commercial API Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for high-value intermediates like 5-methoxyindole, a critical precursor for melatonin and various tryptamine derivatives. Patent CN100432052C introduces a transformative four-step methodology that utilizes o-nitrotoluene, a ubiquitous and cost-effective bulk chemical, as the foundational starting material. This technical breakthrough addresses long-standing inefficiencies in indole synthesis by replacing hazardous reagents with safer, more scalable alternatives while maintaining high economic added value. By shifting away from complex diazotization processes and toxic methylating agents, this route offers a compelling value proposition for large-scale API manufacturing. The process demonstrates exceptional versatility through two distinct variations, allowing manufacturers to optimize for either maximum purity or streamlined operational simplicity depending on specific production requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-methoxyindole has relied on methodologies such as the Fisher indole synthesis or the Leimgruber-Batcho method, both of which present significant industrial hurdles. Traditional Fisher methods often necessitate the use of aluminum oxide catalysts at extremely high temperatures ranging from 320°C to 330°C, creating severe energy demands and safety risks associated with thermal runaway. Furthermore, alternative routes starting from m-cresol involve nitrosation oxidation steps that utilize carcinogenic sodium nitrite and highly toxic dimethyl sulfate, posing unacceptable environmental and occupational health hazards. These legacy processes frequently suffer from low overall yields, sometimes dropping below 30%, due to side reactions like carbonization during high-temperature decarboxylation steps. The reliance on unstable diazonium intermediates in some classical approaches also introduces explosion risks that complicate regulatory compliance and insurance assessments for modern chemical plants.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages a mild hydroxymethylation followed by a controlled reduction and Bamberger rearrangement to achieve superior outcomes. By initiating the synthesis with o-nitrotoluene, the process bypasses the need for expensive and unstable phenylhydrazone precursors entirely. The reaction conditions are notably温和,operating primarily between 20°C and 100°C across the initial steps, which drastically reduces energy consumption compared to the 300°C+ requirements of older technologies. This methodology effectively eliminates the use of dimethyl sulfate, replacing it with safer methanol-based methoxylation during the rearrangement phase. The result is a streamlined workflow that not only enhances operator safety but also simplifies waste treatment protocols, making it an ideal candidate for green chemistry initiatives in fine chemical manufacturing.
Mechanistic Insights into the Four-Step Cascade Synthesis
The core of this innovation lies in a carefully orchestrated sequence of reactions beginning with the hydroxymethylation of o-nitrotoluene. In this initial phase, a basic catalyst facilitates the condensation of o-nitrotoluene with formaldehyde sources such as paraformaldehyde or aqueous formaldehyde solution. The reaction proceeds efficiently at temperatures between 20°C and 100°C, yielding o-nitrophenylethanol with high selectivity. Following isolation, the nitro group undergoes a selective chemical reduction using zinc powder in the presence of ammonium chloride. This step is critical as it converts the nitro functionality into a hydroxylamine moiety without over-reducing to the amine prematurely, forming 2-(β-hydroxyethyl)-phenylhydroxylamine. The precise control of temperature at 60°C to 65°C during zinc addition ensures minimal formation of azo-byproducts, which are common impurities in nitro reductions.
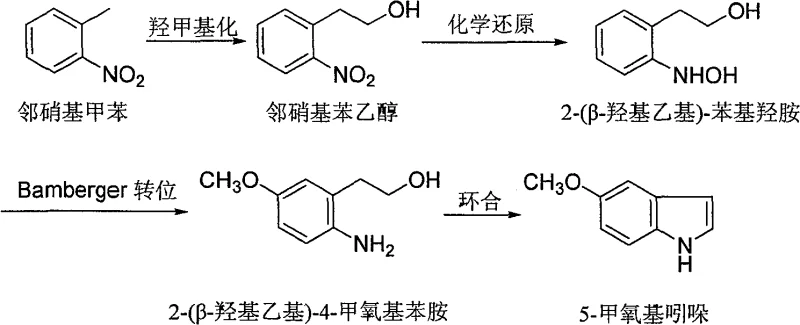
The subsequent Bamberger transposition represents the key structural modification step where the aromatic ring is functionalized. By treating the phenylhydroxylamine intermediate with sulfuric acid in anhydrous methanol under reflux, the molecule undergoes a rearrangement that installs the methoxy group at the para-position relative to the amine, yielding 2-(β-hydroxyethyl)-4-methoxyaniline. This intramolecular migration is highly efficient and avoids the external alkylation steps that typically generate salt waste. Finally, the cyclization to form the indole ring is achieved through a catalytic dehydrogenation process. Depending on the specific embodiment, manufacturers can utilize either a triphenylphosphine ruthenium chloride complex in toluene or a Raney nickel catalyst in liquid paraffin. This flexibility allows for optimization based on catalyst recovery capabilities and desired reaction kinetics, ensuring the final 5-methoxyindole product meets stringent purity specifications required for pharmaceutical applications.
How to Synthesize 5-Methoxyindole Efficiently
Implementing this synthesis route requires strict adherence to the specified stoichiometric ratios and thermal profiles to maximize yield and minimize impurity formation. The process is designed to be scalable, moving seamlessly from laboratory glassware to industrial reactors with minimal modification to the core chemical logic. Operators must pay particular attention to the pH adjustment during the hydroxymethylation workup and the temperature control during the exothermic zinc reduction phase. Detailed standard operating procedures regarding catalyst loading, solvent recovery, and crystallization parameters are essential for consistent batch-to-batch quality. For a comprehensive breakdown of the specific reagent quantities, reaction times, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide provided below.
- Perform hydroxymethylation of o-nitrotoluene with formaldehyde and a basic catalyst at 20-100°C to obtain o-nitrophenylethanol.
- Conduct chemical reduction using zinc powder and ammonium chloride at 60-65°C to synthesize 2-(β-hydroxyethyl)-phenylhydroxylamine.
- Execute Bamberger transposition in methanol with sulfuric acid to form 2-(β-hydroxyethyl)-4-methoxyaniline.
- Complete cyclization using a ruthenium catalyst or Raney nickel at elevated temperatures to finalize 5-methoxyindole production.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial strategic benefits by decoupling production from volatile specialty chemical markets. The primary starting material, o-nitrotoluene, is a commodity chemical produced in massive volumes globally, ensuring a stable and continuous supply chain that is resistant to the shortages often seen with niche intermediates. By eliminating the need for toxic reagents like dimethyl sulfate, facilities can significantly reduce the costs associated with hazardous material handling, storage, and specialized waste disposal. This reduction in regulatory burden translates directly into lower operational overhead and faster permitting times for new production lines. Furthermore, the mild reaction conditions reduce wear and tear on reactor vessels and heating systems, extending equipment lifespan and lowering maintenance expenditures over the long term.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the utilization of low-cost bulk raw materials instead of high-value specialized precursors. By avoiding expensive catalysts and toxic reagents that require complex neutralization and disposal, the overall cost of goods sold is drastically optimized. The high atom economy of the Bamberger rearrangement step ensures that a greater proportion of the input mass is converted into the final valuable product, minimizing raw material waste. Additionally, the ability to recycle solvents like toluene and methanol further contributes to a leaner cost structure, making the final API intermediate more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: Relying on o-nitrotoluene as the feedstock provides a robust foundation for supply continuity, as this chemical is widely available from multiple suppliers worldwide. The simplified four-step sequence reduces the number of intermediate isolation points, thereby decreasing the risk of bottlenecks or yield losses that often plague longer synthetic routes. This streamlined approach allows for faster turnaround times from order to delivery, enabling pharmaceutical partners to maintain tighter inventory controls. The stability of the intermediates also permits safer storage and transport if multi-site manufacturing strategies are employed, adding another layer of resilience to the supply network.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations such as filtration, distillation, and crystallization that are well-understood in chemical engineering. The absence of high-pressure hydrogenation or extreme temperature requirements simplifies the engineering controls needed for large-scale reactors. Environmentally, the elimination of carcinogenic nitrites and toxic sulfates aligns with increasingly strict global environmental regulations, reducing the risk of compliance violations. The aqueous workups and manageable organic waste streams facilitate easier treatment, supporting corporate sustainability goals and reducing the carbon footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its integration into existing production portfolios. The following questions address common concerns regarding safety, scalability, and quality control associated with this specific patent technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to ensure accuracy and relevance. We encourage technical teams to review these points closely when assessing the feasibility of adopting this route for commercial manufacture.
Q: What are the safety advantages of this 5-methoxyindole synthesis route?
A: This method eliminates the use of highly toxic dimethyl sulfate and carcinogenic sodium nitrite required in traditional Leimgruber-Batcho or m-cresol routes, significantly improving operational safety and reducing hazardous waste disposal costs.
Q: Why is o-nitrotoluene preferred as the starting material?
A: o-Nitrotoluene is a basic bulk chemical commodity with stable pricing and high availability, unlike specialized precursors like p-methoxyaniline phenylhydrazones which require complex diazotization steps prone to explosion risks.
Q: How does the Bamberger rearrangement impact yield in this process?
A: The Bamberger transposition step allows for direct methoxylation under mild acidic conditions in methanol, avoiding the harsh decarboxylation temperatures of older methods that often lead to carbonization and low overall yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Methoxyindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for high-value pharmaceutical intermediates like 5-methoxyindole. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify every batch against pharmacopeial standards. Our commitment to technical excellence means we can adapt the patented synthesis route to fit your specific needs, whether you require custom particle sizes or specific impurity profiles tailored to your downstream API synthesis.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can benefit your project timelines and budget. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this o-nitrotoluene-based route. We are ready to provide specific COA data and route feasibility assessments to support your vendor qualification process. Contact us today to secure a sustainable and cost-effective supply of 5-methoxyindole for your next generation of therapeutic products.
