Advanced Enzymatic Resolution for High-Purity Halogenated Alpha-Amino Acid Manufacturing
The pharmaceutical and fine chemical industries face a persistent challenge in sourcing optically pure halogenated alpha-amino acids, which serve as critical building blocks for synthesizing antibiotics, antivirals, and agrochemicals. Patent CN103074409A introduces a groundbreaking biocatalytic strategy that addresses the limitations of traditional chemical resolution by leveraging the high stereoselectivity of immobilized penicillin acylase. This technology enables the efficient separation of racemic halogenated alpha-amino acids into their respective L and D enantiomers with exceptional optical purity, often exceeding 99% ee. By integrating mild enzymatic hydrolysis with robust chemical recovery steps, this method offers a sustainable pathway for producing high-value intermediates like L-chloroalanine and L-4-bromo-2-aminobutyric acid. For global procurement teams, this represents a shift towards more reliable supply chains characterized by reduced dependency on expensive chiral reagents and improved environmental compliance through minimized solvent usage.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically active halogenated amino acids has relied heavily on chemical resolution or complex asymmetric synthesis, both of which suffer from significant economic and technical drawbacks. Traditional chemical resolution often involves the formation of diastereomeric salts using expensive chiral resolving agents, a process that is inherently limited to a maximum theoretical yield of 50% for the desired enantiomer unless dynamic kinetic resolution is employed. Furthermore, asymmetric synthesis routes frequently require precious metal catalysts or specialized chiral auxiliaries that drive up production costs and introduce heavy metal contamination risks requiring stringent purification. As illustrated in prior art approaches, such as the multi-step chemical conversion of L-Methionine shown below, conventional pathways often involve harsh reagents like phosphorus pentachloride or complex protection-deprotection sequences that generate substantial waste.
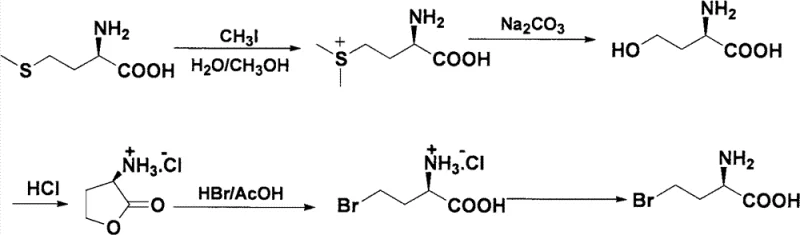
The complexity of these legacy methods is further compounded by difficult separation processes, where removing by-products like dichloro- or trichloro- derivatives requires energy-intensive distillation or chromatography. Additionally, fermentation methods, while green, are often restricted by substrate specificity and low productivity, failing to meet the high-volume demands of modern API manufacturing. These inefficiencies result in prolonged lead times and volatile pricing structures, creating supply chain vulnerabilities for downstream manufacturers who require consistent quality and volume. Consequently, there is an urgent industry need for a resolution technology that combines the selectivity of biology with the scalability of chemical engineering.
The Novel Approach
The patented methodology revolutionizes this landscape by introducing a highly selective enzymatic splitting process centered on immobilized penicillin acylase (PGA). The core innovation lies in the N-acylation of the racemic halo-alpha-amino acid with phenylacetyl chloride, followed by a precise enzymatic hydrolysis step where the enzyme selectively cleaves the amide bond of only the L-enantiomer. This biological discrimination leaves the D-enantiomer intact as an N-phenylacetyl derivative, allowing for a clean physical separation based on solubility differences between the free amino acid and the acylated species. The aqueous phase yields the pure L-amino acid, while the organic phase retains the protected D-enantiomer, which is subsequently recovered via simple acid hydrolysis. This approach effectively bypasses the 50% yield ceiling of traditional resolution by enabling the recycling of the unwanted enantiomer through racemization strategies in future iterations, although the patent focuses on high-yield direct splitting.
Mechanistic Insights into Penicillin Acylase-Catalyzed Resolution
The success of this resolution strategy hinges on the exquisite stereospecificity of penicillin G acylase, an enzyme that has evolved to recognize specific steric configurations within its active site. In the context of N-phenylacetyl-DL-halo-alpha-amino acids, the enzyme's catalytic triad facilitates nucleophilic attack on the carbonyl carbon of the amide bond exclusively when the substrate presents the L-configuration. The bulky halogen substituent on the alpha-carbon of the D-enantiomer creates a steric clash within the enzyme's binding pocket, preventing the proper alignment required for hydrolysis. This kinetic resolution is conducted in a borate buffer system maintained at a pH between 6 and 10, conditions that optimize enzyme stability while ensuring the substrate remains in a soluble ionization state. The reaction temperature is carefully controlled between 20°C and 40°C to balance reaction rate with enzyme longevity, preventing thermal denaturation that could compromise the resolution efficiency.
Impurity control is intrinsically built into this mechanism through the phase separation strategy employed post-reaction. Once the enzymatic hydrolysis is complete, the reaction mixture is acidified to a pH of 0-4, protonating the free L-amino acid and keeping it in the aqueous phase, while the unhydrolyzed N-phenylacetyl-D-amino acid remains neutral and lipophilic. Extraction with hydrophobic solvents like butyl acetate effectively partitions the D-enantiomer into the organic layer, leaving behind inorganic salts and enzyme residues in the water. This physical separation minimizes cross-contamination between enantiomers, a common issue in crystallization-based resolutions. Finally, the chemical hydrolysis of the D-enantiomer using concentrated HCl at elevated temperatures (80-120°C) ensures complete deprotection without racemization, yielding the final D-halo-alpha-amino acid with optical purity levels that consistently surpass 99% ee, meeting the rigorous standards required for chiral drug synthesis.
How to Synthesize Halogenated Alpha-Amino Acid Efficiently
The synthesis protocol outlined in the patent provides a robust framework for manufacturing facilities aiming to transition from chemical to biocatalytic processes. The procedure begins with the N-acylation of the racemic starting material, followed by the critical enzymatic splitting step which dictates the overall optical purity of the product. Operators must maintain strict control over pH and temperature during the biocatalysis phase to ensure maximum enzyme turnover and selectivity. Following the biocatalytic step, the workup involves a straightforward liquid-liquid extraction and acid hydrolysis, eliminating the need for complex chromatographic purifications. For a detailed breakdown of the specific reaction parameters, stoichiometry, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Perform N-acylation of DL-halo-alpha-amino acid with phenylacetyl chloride under basic conditions to form N-phenylacetyl-DL-halo-alpha-amino acid.
- Conduct selective enzymolysis using immobilized penicillin acylase in borate buffer to hydrolyze the L-enantiomer while leaving the D-enantiomer acylated.
- Separate the phases via extraction and chemically hydrolyze the remaining N-phenylacetyl-D-enantiomer using HCl to recover the pure D-amino acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic resolution technology translates into tangible improvements in cost structure and supply reliability. The primary economic driver is the remarkable stability and reusability of the immobilized penicillin acylase, which the patent data indicates can be recycled for over one hundred reaction cycles without significant loss of activity. This drastic reduction in biocatalyst consumption lowers the variable cost per kilogram of product, insulating buyers from the price volatility associated with single-use chiral catalysts or resolving agents. Furthermore, the process operates under mild aqueous conditions, reducing the need for expensive anhydrous solvents and specialized pressure-rated reactors, thereby lowering capital expenditure requirements for scale-up.
- Cost Reduction in Manufacturing: The elimination of expensive chiral resolving agents and precious metal catalysts significantly reduces the raw material bill of materials. By utilizing a reusable immobilized enzyme system, manufacturers avoid the recurring costs of purchasing stoichiometric amounts of chiral auxiliaries, leading to substantial long-term savings. Additionally, the simplified downstream processing, which relies on extraction and crystallization rather than preparative HPLC, reduces utility consumption and solvent recovery costs. These factors combine to create a highly competitive cost profile for high-purity halogenated amino acids, making them more accessible for large-scale API production.
- Enhanced Supply Chain Reliability: The reliance on domestically produced immobilized enzymes, as noted in the patent, mitigates supply chain risks associated with importing specialized reagents from single-source vendors. The robustness of the enzymatic process ensures consistent batch-to-batch quality, reducing the incidence of out-of-specification products that can disrupt production schedules. Moreover, the ability to produce both L and D enantiomers from a single racemic feedstock provides flexibility in meeting varying market demands for different chiral forms. This versatility strengthens the resilience of the supply chain against fluctuations in demand for specific stereoisomers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in multi-liter reactor volumes with straightforward agitation and temperature control requirements. The use of water as the primary reaction medium and the minimal usage of recyclable organic solvents align with green chemistry principles, simplifying wastewater treatment and regulatory compliance. Reduced hazardous waste generation lowers disposal costs and enhances the sustainability profile of the manufacturing site. This environmental advantage is increasingly critical for maintaining operational licenses and meeting the corporate social responsibility goals of multinational pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic resolution technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the process feasibility. Understanding these details is essential for R&D teams assessing the integration of this method into existing production lines and for quality assurance personnel verifying product specifications.
Q: What optical purity can be achieved with this enzymatic resolution method?
A: The patented method consistently achieves optical purity exceeding 99% ee, with specific embodiments reaching up to 99.9% ee for various halogenated substrates like chloroalanine and bromoalanine.
Q: How does the immobilized penicillin acylase contribute to cost efficiency?
A: The immobilized enzyme demonstrates exceptional stability and can be reused for over 100 reaction cycles without significant loss of activity, drastically reducing the cost per kilogram of the final product compared to single-use chiral catalysts.
Q: Which specific halogenated amino acids are suitable for this process?
A: The process is versatile and has been successfully demonstrated for L/D-chloroalanine, L/D-bromoalanine, L/D-fluoroalanine, and L/D-4-halo-2-aminobutyric acids, making it broadly applicable for diverse pharmaceutical intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halogenated Alpha-Amino Acid Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced biocatalytic technologies to deliver superior pharmaceutical intermediates to the global market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of halogenated alpha-amino acid meets the exacting standards required for GMP manufacturing. Our facility is equipped to handle the specific buffering and extraction requirements of this enzymatic process, ensuring consistent quality and supply continuity.
We invite potential partners to engage with our technical procurement team to discuss how this innovative resolution method can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to our enzymatically produced intermediates. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring that your development timelines are met with the highest quality materials available.
