Advanced Catalytic Synthesis of 2,3-Quinoxaline Dimethanol-1,4-Dioxide for Commercial Scale-Up
Advanced Catalytic Synthesis of 2,3-Quinoxaline Dimethanol-1,4-Dioxide for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and cost-efficient pathways for synthesizing complex heterocyclic intermediates. A significant breakthrough in this domain is documented in Chinese Patent CN111825612B, which details a novel preparation method for 2,3-quinoxaline dimethanol-1,4-dioxide. This compound serves as a critical building block for a wide array of antibacterial drugs and veterinary agents, characterized by their potent biological activity and favorable safety profiles. The patent outlines a sophisticated four-step synthetic strategy that begins with the condensation of o-phenylenediamine and 2,3-butanedione, followed by sequential oxidation and rearrangement reactions. This approach not only addresses the limitations of prior art but also establishes a new benchmark for purity and yield in the manufacturing of quinoxaline nitrogen oxides. By leveraging advanced Lewis acid catalysis and controlled oxidative conditions, this technology offers a compelling value proposition for global supply chains.
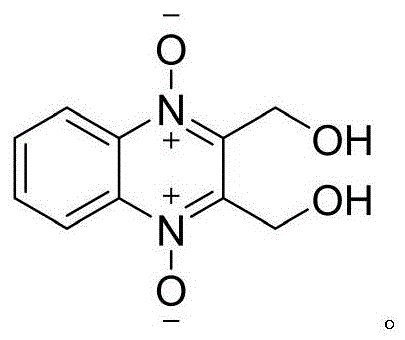
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoxaline-1,4-dioxide derivatives has relied heavily on the Beirut reaction, a pathway that presents substantial economic and operational challenges for large-scale manufacturers. As illustrated in the legacy synthetic route, this conventional method typically involves the reaction of benzofurazan-1-oxide with ketones, followed by cumbersome bromination and substitution steps. The primary drawback of this approach lies in the exorbitant cost and limited availability of the starting material, benzofurazan-1-oxide, which acts as a significant bottleneck for procurement teams aiming to reduce raw material expenses. Furthermore, the involvement of bromination introduces severe safety hazards and generates toxic halogenated waste streams, complicating environmental compliance and increasing the burden on waste treatment facilities. The cumulative yield of such multi-step sequences is often suboptimal, leading to higher production costs per kilogram and reduced overall process efficiency.
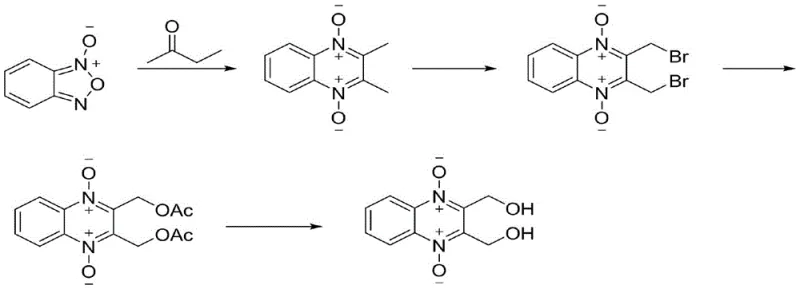
The Novel Approach
In stark contrast to the legacy methods, the innovative process disclosed in the patent utilizes a direct condensation strategy starting from commodity chemicals, fundamentally reshaping the cost structure of production. The new route initiates with the dehydration and cyclization of o-phenylenediamine and 2,3-butanedione, two widely available and inexpensive feedstocks, to form 2,3-dimethylquinoxaline. This is followed by a series of controlled oxidations and a unique rearrangement-hydrolysis sequence that efficiently installs the necessary functional groups without the need for hazardous halogenation. The overall process flow is streamlined into four continuous steps, maximizing atom economy and minimizing the generation of by-products. By eliminating the reliance on expensive precursors and dangerous reagents, this methodology not only enhances the economic viability of the project but also aligns perfectly with modern green chemistry principles, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks seeking sustainable solutions.
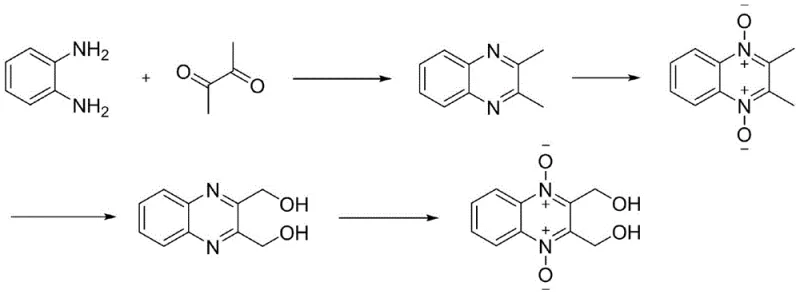
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization and Oxidation
The success of this synthetic pathway hinges on the precise application of Lewis acid catalysis during the initial cyclization step, which dictates the quality of the intermediate and the efficiency of the entire sequence. The patent highlights the efficacy of catalysts such as anhydrous ferric trichloride (FeCl3) or tris(pentafluorophenyl)borane ((C6F5)3B) in facilitating the condensation of o-phenylenediamine with 2,3-butanedione. These catalysts activate the carbonyl groups of the diketone, promoting nucleophilic attack by the amine groups and accelerating the formation of the quinoxaline ring. Crucially, the reaction is conducted under micro-vacuum conditions to continuously remove the water by-product, driving the equilibrium towards completion and ensuring high conversion rates. This mechanistic understanding allows for tight control over impurity profiles, as the choice of catalyst and the rigorous removal of water prevent the formation of oligomeric side products that could complicate downstream purification efforts.
Following the cyclization, the process employs a series of oxidation steps utilizing hydrogen peroxide as a clean and atom-economical oxidant. The transformation of the methyl groups to hydroxymethyl groups involves a complex interplay of oxidation and rearrangement mechanisms, often mediated by catalysts like tungstic acid, phthalic anhydride, or trifluoroacetic acid. In the rearrangement step, the use of acid anhydrides facilitates the migration of the N-oxide oxygen to the adjacent methyl group, a transformation akin to the Boekelheide rearrangement, which is then hydrolyzed to reveal the alcohol functionality. This sequence is meticulously optimized to prevent over-oxidation or degradation of the sensitive quinoxaline N-oxide core. The final oxidation step reinstates the N-oxide functionality on the dimethanol intermediate, completing the synthesis with high regioselectivity. Understanding these mechanistic nuances is vital for R&D directors aiming to replicate this high-purity process in their own facilities.
How to Synthesize 2,3-Quinoxaline Dimethanol-1,4-Dioxide Efficiently
Implementing this synthesis requires strict adherence to the reaction parameters defined in the patent to ensure reproducibility and safety. The process is divided into four distinct stages, each requiring specific attention to temperature control, stoichiometry, and workup procedures. The initial cyclization sets the foundation for the entire synthesis, necessitating the use of dry solvents and effective water removal techniques. Subsequent oxidation steps must be carefully monitored to manage the exothermic nature of hydrogen peroxide reactions, ensuring that the temperature remains within the narrow window of 35-40°C to prevent runaway reactions. The rearrangement step involves handling reactive acid anhydrides at elevated temperatures, requiring robust corrosion-resistant equipment. Finally, the purification strategy relies on recrystallization to achieve the target purity specifications. For a detailed breakdown of the specific operational parameters, reagent quantities, and isolation techniques, please refer to the standardized synthesis guide below.
- Condense o-phenylenediamine with 2,3-butanedione using a Lewis acid catalyst like FeCl3 or (C6F5)3B in toluene under reflux with water removal.
- Oxidize the resulting 2,3-dimethylquinoxaline using hydrogen peroxide with catalysts such as tungstic acid, phthalic anhydride, or TFA in DCM.
- Perform rearrangement and hydrolysis using acid anhydrides (TFAA or Acetic Anhydride) followed by base hydrolysis to form the dimethanol intermediate.
- Conduct a final oxidation of the dimethanol intermediate using hydrogen peroxide and catalysts to yield the target 1,4-dioxide product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers transformative advantages that directly address the pain points of procurement managers and supply chain heads in the fine chemical sector. The shift from expensive, specialized starting materials to bulk commodity chemicals like o-phenylenediamine drastically reduces the raw material cost basis, providing a significant buffer against market volatility. This cost reduction in API manufacturing is further amplified by the elimination of bromination steps, which removes the need for costly bromine handling infrastructure and the associated regulatory compliance costs. The simplified workflow, consisting of standard unit operations like reflux, distillation, and extraction, means that the process can be easily scaled up in existing multipurpose reactors without requiring capital-intensive new installations. This flexibility ensures a more resilient supply chain capable of adapting to fluctuating demand patterns.
- Cost Reduction in Manufacturing: The economic benefits of this route are profound, primarily driven by the substitution of high-cost precursors with low-cost alternatives. By avoiding the Beirut reaction's reliance on benzofurazan-1-oxide, manufacturers can achieve substantial savings on the bill of materials. Additionally, the use of hydrogen peroxide as the primary oxidant is far more economical than traditional metal-based oxidants or halogenating agents, and its by-product is merely water, which simplifies waste disposal. The high total yield reported in the patent, reaching up to 64 percent, means that less raw material is wasted per unit of product, further driving down the effective cost per kilogram and enhancing the overall profitability of the manufacturing campaign.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the scarcity of specialized intermediates, but this new route mitigates that risk by relying on globally available feedstocks. O-phenylenediamine and 2,3-butanedione are produced at massive scales for various industries, ensuring a stable and competitive supply market. This abundance reduces the lead time for high-purity intermediates, as suppliers do not need to wait for the synthesis of niche starting materials. Furthermore, the robustness of the catalytic systems described allows for consistent batch-to-batch quality, reducing the incidence of failed batches that can disrupt delivery schedules. This reliability makes the process highly attractive for long-term supply agreements with major pharmaceutical clients.
- Scalability and Environmental Compliance: The environmental footprint of chemical manufacturing is a critical consideration for modern enterprises, and this process excels in sustainability metrics. By eliminating halogenated reagents and heavy metal catalysts in key steps, the process generates significantly less hazardous waste, simplifying the permitting process for commercial scale-up of complex heterocycles. The use of common solvents like toluene and dichloromethane, which can be efficiently recovered and recycled, aligns with circular economy goals. The operational simplicity of the four-step sequence facilitates rapid scale-up from pilot plant to full commercial production, allowing companies to bring products to market faster while maintaining strict adherence to environmental regulations and safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what potential partners can expect. Understanding these details is crucial for evaluating the feasibility of adopting this route for your specific production needs. The answers cover aspects ranging from raw material sourcing to the technical nuances of the catalytic systems employed.
Q: What are the primary advantages of this new synthesis route over the traditional Beirut reaction?
A: The new route utilizes inexpensive and readily available starting materials like o-phenylenediamine and 2,3-butanedione, avoiding the high-cost benzofurazan-1-oxide required in the Beirut reaction. Furthermore, it eliminates hazardous bromination steps, significantly improving safety and reducing waste treatment costs while achieving a superior total yield of over 60%.
Q: How does the process ensure high purity suitable for pharmaceutical applications?
A: The process incorporates rigorous purification strategies, including recrystallization in the final step and precise control of reaction parameters such as temperature and stoichiometry. The use of specific Lewis acid catalysts minimizes side reactions, ensuring the final product achieves a purity exceeding 99.0%, which is critical for downstream API synthesis.
Q: Is this synthetic method scalable for industrial production?
A: Yes, the methodology relies on standard unit operations common in fine chemical engineering, such as reflux, distillation, and liquid-liquid extraction. The continuous four-step operation is designed for robustness, allowing for seamless transition from laboratory scale to multi-ton commercial production without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Quinoxaline Dimethanol-1,4-Dioxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN111825612B for maintaining a competitive edge in the global market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical intermediates. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including the safe handling of peroxides and acid anhydrides, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards. We are committed to delivering value through technical excellence and operational reliability.
We invite you to collaborate with us to leverage this advanced technology for your drug development programs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable supply of high-quality quinoxaline derivatives for your future projects.
