Advanced Synthesis of L-Penicillamine Hydrochloride for Commercial Scale-Up
Introduction to the Breakthrough in L-Penicillamine Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for critical chiral amino acids, and the recent disclosure in patent CN113307754A represents a significant leap forward in the production of L-penicillamine hydrochloride. This optically active compound serves as a pivotal building block for a diverse array of therapeutic agents, including cardiovascular medications like SD3321 and potent anticancer candidates such as AG 3340, alongside its established utility in HIV-1 protease inhibitors. Historically, the synthesis of this high-value intermediate has been plagued by technical bottlenecks, specifically regarding reaction severity and scalability limitations that hinder consistent commercial supply. The novel methodology outlined in this intellectual property addresses these challenges head-on by introducing a streamlined five-step sequence that prioritizes operational simplicity and high throughput. By shifting away from cumbersome traditional protocols, this approach offers a viable solution for manufacturers aiming to secure a reliable pharma intermediate supplier capable of meeting stringent global demand without compromising on quality or delivery timelines.
At the core of this innovation is a strategic reimagining of the synthetic timeline, where the initial steps combine racemization and functional group protection into a singular, efficient operation. This consolidation not only reduces the total number of unit operations required but also minimizes the exposure of sensitive intermediates to potentially degrading conditions. For R&D directors and process chemists, the implication is a more predictable reaction profile with fewer opportunities for impurity generation, directly translating to higher purity profiles in the final active pharmaceutical ingredient. Furthermore, the use of common industrial solvents and reagents throughout the sequence ensures that the transition from laboratory bench to pilot plant and eventually to full commercial scale is seamless. This patent effectively bridges the gap between academic feasibility and industrial reality, positioning L-penicillamine hydrochloride as a more accessible commodity for downstream drug formulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art technologies, such as those described in U.S. Pat. No. 3,398,066, have long relied on starting materials like DL-penicillamine hydrochloride which necessitate complex resolution strategies involving multiple solvent systems and specific salt formations. These legacy processes often suffer from harsh reaction conditions that require precise temperature control and extended reaction times, creating significant energy burdens and safety risks in a manufacturing environment. Additionally, the reliance on specialized resolving agents or multi-step protection-deprotection sequences introduces cumulative yield losses that can drastically impact the overall economics of the synthesis. The accumulation of byproducts and the difficulty in purifying intermediates at each stage further complicate the workflow, leading to increased waste generation and higher disposal costs. For supply chain managers, these inefficiencies manifest as longer lead times and greater vulnerability to raw material shortages, making it difficult to guarantee consistent availability for critical drug production lines.
The Novel Approach
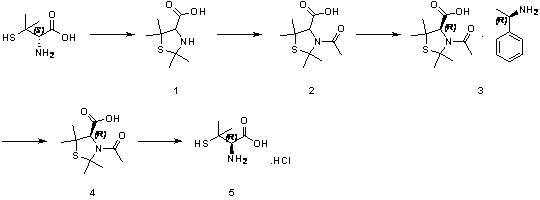
In stark contrast, the methodology presented in CN113307754A introduces a paradigm shift by utilizing D-penicillamine as a starting point and subjecting it to a reflux reaction in an acetic acid and acetone system. This single step ingeniously accomplishes both the racemization of the starting material and the simultaneous protection of the sulfhydryl and amino groups, forming a stable thiazolidine derivative known as Compound 1. This consolidation eliminates the need for separate protection steps, thereby reducing solvent consumption and processing time significantly. Subsequent acetylation and resolution steps are optimized to use readily available reagents like acetyl chloride and (D)-(+)-1-phenylethylamine, which are not only cost-effective but also easy to source in bulk quantities. The final deprotection is achieved through a straightforward hydrolysis in hydrochloric acid, avoiding the need for exotic catalysts or extreme conditions. This streamlined architecture ensures that cost reduction in pharmaceutical intermediates manufacturing is achieved through process intensification rather than mere raw material substitution.
Mechanistic Insights into Thiazolidine Formation and Chiral Resolution
The chemical elegance of this synthesis lies in the formation of the thiazolidine ring during the initial reflux stage, which serves as a robust protecting group for the highly reactive thiol moiety of penicillamine. Under the acidic conditions provided by the acetic acid and acetone mixture, the amino and sulfhydryl groups condense with the ketone to form a five-membered heterocyclic structure that effectively masks the nucleophilicity of the sulfur atom. This protection is crucial for preventing unwanted oxidation or side reactions during the subsequent acetylation phase, where acetyl chloride is introduced in the presence of triethylamine. The mechanistic pathway ensures that the stereochemical integrity is managed carefully, allowing for the eventual isolation of the desired L-enantiomer through diastereomeric salt formation. The stability of this intermediate allows for rigorous purification steps, such as filtration and washing, which are essential for removing trace impurities before the critical resolution step.
Following acetylation, the introduction of (D)-(+)-1-phenylethylamine triggers a classical resolution process based on the differential solubility of diastereomeric salts in acetone. The interaction between the chiral amine and the acetylated penicillamine derivative creates distinct crystalline forms, allowing the desired isomer to precipitate out of the solution while the unwanted enantiomer remains in the mother liquor. This physical separation is far more scalable than chromatographic methods and is highly favored in industrial settings for its simplicity and reproducibility. Once the salt is isolated, treatment with dilute hydrochloric acid dissociates the complex, releasing the protected amino acid which is then subjected to final hydrolysis. The hydrolytic cleavage of the thiazolidine ring and the acetyl group under reflux in concentrated hydrochloric acid regenerates the free amino and thiol groups, yielding the target L-penicillamine hydrochloride with high optical purity. This mechanistic understanding underscores the reliability of the process for producing high-purity L-penicillamine hydrochloride suitable for sensitive pharmaceutical applications.
How to Synthesize L-Penicillamine Hydrochloride Efficiently
Executing this synthesis requires strict adherence to the molar ratios and solvent volumes specified in the patent to maximize yield and purity at every stage. The process begins with the reflux of D-penicillamine in a specific volume ratio of acetone to acetic acid, followed by careful temperature management during the acetylation to prevent exothermic runaway. The resolution step is particularly sensitive to the stoichiometry of the resolving agent, where a molar ratio of roughly 1:0.45 is maintained to optimize the precipitation of the target diastereomer. Detailed standard operating procedures for each unit operation, including filtration, washing, and drying parameters, are critical for maintaining batch-to-batch consistency. For a comprehensive breakdown of the exact experimental conditions and workup procedures, please refer to the standardized synthesis guide below.
- Reflux D-penicillamine in an acetic acid and acetone system to achieve simultaneous racemization and thiazolidine ring protection, yielding Compound 1.
- React Compound 1 with acetyl chloride in tetrahydrofuran using triethylamine as a base to form the N-acetylated intermediate, Compound 2.
- Perform chiral resolution by salifying Compound 2 with (D)-(+)-1-phenylethylamine in acetone to isolate the diastereomeric salt, Compound 3.
- Dissociate Compound 3 using dilute hydrochloric acid to release the free acid form, Compound 4, removing the resolving agent.
- Hydrolyze Compound 4 in concentrated aqueous hydrochloric acid under reflux to remove protecting groups and obtain the final L-penicillamine hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial economic benefits primarily driven by the utilization of commodity-grade chemicals that are abundantly available in the global market. The elimination of expensive chiral catalysts or rare earth metals from the process flow means that the bill of materials is significantly optimized, leading to a more competitive pricing structure for the final intermediate. Furthermore, the high yields reported in the patent, particularly the near-quantitative conversion in the final hydrolysis step, ensure that raw material utilization is maximized, reducing the effective cost per kilogram of the product. This efficiency translates directly into margin improvements for downstream API manufacturers who rely on consistent and affordable supply chains for their own production schedules. The robustness of the chemistry also implies fewer batch failures, which is a critical factor in maintaining inventory levels and avoiding costly production stoppages.
- Cost Reduction in Manufacturing: The process achieves cost optimization by integrating the racemization and protection steps, which removes the need for separate reactor charges and reduces overall energy consumption. By avoiding the use of precious metal catalysts or complex enzymatic systems, the capital expenditure required for equipment lining and waste treatment is drastically simplified. The use of standard solvents like acetone and tetrahydrofuran allows for efficient recovery and recycling systems to be implemented, further driving down the variable costs associated with solvent procurement and disposal. Consequently, the overall manufacturing footprint is leaner, enabling producers to offer more aggressive pricing without sacrificing quality standards.
- Enhanced Supply Chain Reliability: The reliance on (D)-(+)-1-phenylethylamine as a resolving agent is a strategic advantage, as this chemical is produced on a massive scale for various industrial applications, ensuring a stable and uninterrupted supply. Unlike proprietary resolving agents that might be sourced from a single vendor, this common reagent can be procured from multiple qualified suppliers, mitigating the risk of supply chain disruptions. The simplicity of the reaction conditions, which do not require cryogenic temperatures or high-pressure vessels, also means that the process can be manufactured in a wider range of facilities, increasing the overall capacity available to the market. This flexibility is essential for reducing lead time for high-purity pharmaceutical intermediates during periods of peak demand.
- Scalability and Environmental Compliance: The synthetic pathway is inherently designed for commercial scale-up of complex amino acids, utilizing unit operations such as reflux, filtration, and crystallization that are easily transferred from pilot to production scale. The waste stream is primarily composed of aqueous acid and organic solvents which can be treated using standard effluent treatment protocols, ensuring compliance with increasingly stringent environmental regulations. The high atom economy of the reaction sequence minimizes the generation of hazardous byproducts, aligning with green chemistry principles and reducing the environmental liability for the manufacturer. This sustainability profile is increasingly important for multinational corporations seeking to partner with suppliers who demonstrate a commitment to responsible manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the patent specifications and beneficial effects. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific production needs. The answers provided reflect the documented performance metrics and operational parameters established in the intellectual property, offering a clear picture of what can be expected during technology transfer.
Q: How does this new synthesis method improve upon traditional L-penicillamine production?
A: Traditional methods often involve harsh reaction conditions and complex multi-step resolutions that are difficult to scale. This patented process integrates racemization and protection into a single reflux step and utilizes a cost-effective resolving agent, significantly simplifying the workflow and enhancing suitability for large-scale industrial manufacturing.
Q: What are the key yield advantages of this specific synthetic route?
A: The process demonstrates exceptional efficiency, with individual step yields exceeding 90% for most stages, including the initial protection and final hydrolysis steps. Even the critical chiral resolution step maintains a robust yield of approximately 73%, contributing to a high overall process mass intensity that minimizes waste and maximizes output.
Q: Is the resolving agent used in this process commercially viable for bulk production?
A: Yes, the method employs (D)-(+)-1-phenylethylamine, which is an industrially produced, inexpensive, and readily available chiral amine. This contrasts with older methods that may require exotic or costly chiral auxiliaries, thereby ensuring a stable supply chain and reduced raw material costs for long-term production campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Penicillamine Hydrochloride Supplier
As the global demand for antiviral and cardiovascular therapeutics continues to rise, securing a dependable source of high-quality intermediates is paramount for pharmaceutical success. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent results. Our facility is equipped with state-of-the-art reactors and rigorous QC labs that ensure every batch meets stringent purity specifications, adhering to the highest international standards for pharmaceutical ingredients. We understand the critical nature of supply continuity and have optimized our logistics and inventory management to support just-in-time delivery models for our global partners.
We invite you to collaborate with us to explore how this advanced synthetic route can be integrated into your supply chain to drive efficiency and value. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and commercial reliability.
