Advanced Chiral Resolution Strategies for High-Purity Pharmaceutical Intermediates and Active Ingredients
The landscape of modern pharmaceutical manufacturing is increasingly defined by the demand for enantiomerically pure active ingredients, driven by stringent regulatory requirements and the superior therapeutic profiles of single-isomer drugs. Patent CN1197447A presents a significant technological advancement in the field of chiral separation, detailing a novel process for the resolution of racemates utilizing optical antipodes of phenoxypropionic acid derivatives. This innovation addresses critical bottlenecks in the production of high-value intermediates such as selegiline, ephedrine, and tetrahydropapaverine, offering a pathway to superior optical purity without the complexities associated with traditional resolution agents. By leveraging the unique stereoselective properties of substituted phenoxypropionic acids, manufacturers can achieve robust separation efficiencies that translate directly into enhanced product quality and process reliability.
The core of this invention lies in the specific interaction between the racemic amine substrate and the chiral acid resolver, which forms diastereomeric salts with distinct physical properties, particularly solubility and crystallinity. Unlike previous methods that often struggled with the formation of oils or unstable complexes, this approach ensures the precipitation of well-defined crystalline solids that can be easily isolated through filtration. The structural versatility of the resolving agent, characterized by variable substituents on the phenoxy ring, allows for fine-tuning the resolution process to accommodate a wide range of chemical scaffolds. This adaptability makes the technology highly relevant for diverse applications in the synthesis of complex pharmaceutical intermediates where chirality is a defining feature of biological activity.
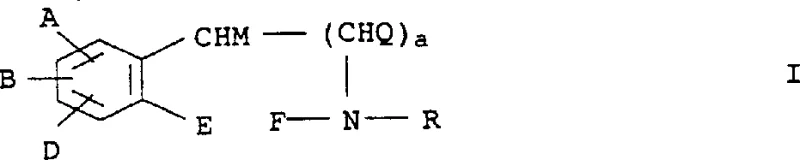
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the fractionation of racemic modifications has been plagued by significant technical hurdles that compromise both yield and operational efficiency. Traditional resolution agents, such as N-Acetylleucine, often exhibit inadequate stability under processing conditions, leading to the formation of diastereomeric salts that are difficult to handle. A major drawback observed in conventional protocols is the tendency for the unwanted enantiomer to precipitate first or for the desired product to remain in the mother liquor as an oil, necessitating complex and costly purification steps. Furthermore, the reliance on naturally occurring optical acids limits the flexibility of the process, as these reagents may not possess the optimal steric or electronic properties required for efficient separation of specific synthetic amines. These inefficiencies result in prolonged production cycles, increased solvent consumption, and ultimately, higher manufacturing costs that erode profit margins in competitive markets.
The Novel Approach
In stark contrast, the methodology disclosed in the patent introduces a streamlined resolution strategy that overcomes these inherent deficiencies through the use of synthetic phenoxypropionic acid derivatives. By employing optical antipodes of compounds like 2-(2,4-dichlorophenoxy)propionic acid, the process achieves high selectivity, ensuring that the desired enantiomer crystallizes preferentially with exceptional purity. The stability of these resolving agents eliminates the risk of degradation during the heating and cooling cycles essential for crystallization, thereby providing a consistent and reproducible outcome. Moreover, the resulting diastereomeric salts possess superior crystalline characteristics, facilitating rapid filtration and washing steps that significantly reduce downstream processing time. This novel approach not only simplifies the operational workflow but also enhances the overall economic viability of producing enantiopure intermediates on a commercial scale.
Mechanistic Insights into Phenoxypropionic Acid-Mediated Chiral Resolution
The efficacy of this resolution technique is rooted in the precise molecular recognition between the chiral acid and the racemic amine, governed by the spatial arrangement of substituents on the phenoxy ring. The presence of electron-withdrawing groups, such as chlorine atoms at the 2 and 4 positions, modulates the acidity and steric bulk of the resolving agent, creating a highly differentiated environment for the formation of diastereomeric ion pairs. This differentiation is critical because it amplifies the solubility differences between the two possible salt forms, driving the equilibrium towards the precipitation of the less soluble diastereomer. The mechanism relies on the thermodynamic stability of the crystal lattice formed by the matched pair, which is energetically favored over the mismatched pair that remains in solution. Understanding these subtle intermolecular interactions allows chemists to predict and optimize resolution outcomes for new substrates by selecting the appropriate substitution pattern on the phenoxypropionic acid scaffold.
Furthermore, the control of impurities is intrinsically linked to the crystallization dynamics facilitated by this specific class of resolving agents. The high selectivity of the phenoxypropionic acid derivatives minimizes the co-crystallization of the undesired enantiomer, effectively acting as a purification step during the salt formation itself. This reduces the burden on subsequent recrystallization stages, which are often required in less selective processes to meet stringent optical purity specifications. The robustness of the salt formation also means that minor variations in reaction conditions, such as temperature fluctuations or solvent ratios, do not drastically impact the enantiomeric excess of the final product. Consequently, this mechanistic advantage translates into a more forgiving and reliable manufacturing process, reducing the risk of batch failures and ensuring consistent quality across large-scale production runs.
How to Synthesize Enantiomerically Pure Tetrahydropapaverine Efficiently
The practical implementation of this resolution technology involves a straightforward sequence of dissolution, crystallization, and liberation steps that are amenable to standard chemical engineering equipment. Operators begin by dissolving the racemic base and the chiral acid in a heated solvent system, typically isopropanol or toluene, to ensure complete homogenization before initiating the crystallization phase. The careful control of cooling rates and the potential use of seeding techniques are crucial for maximizing the yield of the target diastereomeric salt while maintaining high optical purity. Following isolation, the pure enantiomer is liberated from the salt through basic hydrolysis and extracted into an organic phase, ready for final purification or direct use in downstream synthesis. For a detailed breakdown of the specific operational parameters and stoichiometric ratios required for optimal results, please refer to the standardized synthesis guide below.
- Dissolve the racemic base compound (e.g., tetrahydropapaverine) and the optically active resolving agent (e.g., D-2,4-dichlorophenoxypropionic acid) in a suitable solvent like isopropanol or toluene under heating.
- Allow the mixture to cool slowly to induce crystallization of the desired diastereomeric salt, optionally seeding with pure crystals to enhance selectivity.
- Filter the crystalline salt, wash with cold solvent, and liberate the free base by treatment with aqueous alkali followed by extraction with an immiscible organic solvent.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this resolution methodology offers substantial benefits by addressing key pain points related to cost, reliability, and scalability in the supply of chiral intermediates. The use of stable, synthetic resolving agents eliminates the supply chain volatility associated with natural chiral pool materials, ensuring a consistent and predictable source of critical reagents. Additionally, the simplified processing requirements reduce the need for specialized equipment or extended cycle times, allowing manufacturing partners to respond more agilely to fluctuating market demands. This operational efficiency directly contributes to a more resilient supply chain capable of sustaining long-term production commitments without the risk of bottlenecks caused by complex purification challenges.
- Cost Reduction in Manufacturing: The elimination of complex purification steps and the high yield of the crystallization process lead to significant savings in solvent usage and energy consumption. By avoiding the formation of oily residues that require extensive chromatographic separation, manufacturers can drastically reduce waste disposal costs and improve overall material throughput. The stability of the resolving agent also means that reagent loss due to degradation is minimized, further optimizing the cost structure of the production process.
- Enhanced Supply Chain Reliability: The reliance on readily available synthetic chemicals rather than scarce natural products mitigates the risk of raw material shortages that can disrupt production schedules. The robustness of the crystallization process ensures high batch-to-batch consistency, reducing the likelihood of quality deviations that could lead to shipment delays. This reliability is crucial for maintaining just-in-time inventory levels and meeting the strict delivery timelines demanded by global pharmaceutical clients.
- Scalability and Environmental Compliance: The process utilizes common industrial solvents like toluene and isopropanol, which are easily recovered and recycled, aligning with modern environmental sustainability goals. The simplicity of the unit operations involved facilitates seamless scale-up from pilot plant to commercial production volumes without the need for process re-engineering. This scalability ensures that supply can be rapidly expanded to meet growing market needs while maintaining compliance with rigorous environmental and safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the application and optimization of this chiral resolution technology, providing clarity for R&D and technical procurement teams evaluating its potential integration into their manufacturing workflows. These insights are derived directly from the experimental data and theoretical framework presented in the patent documentation, ensuring accuracy and relevance for industrial application. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: What are the primary advantages of using phenoxypropionic acid derivatives for chiral resolution compared to traditional agents?
A: Unlike traditional agents such as N-Acetylleucine which may produce unstable or oily diastereomeric salts that are difficult to separate, phenoxypropionic acid derivatives form highly stable, crystalline salts. This stability ensures high optical purity and simplifies the isolation process through standard filtration techniques.
Q: Which solvents are recommended for the crystallization process in this resolution method?
A: The patent specifies the use of lower alcohols such as isopropanol, as well as aromatic hydrocarbons like toluene. These solvents are chosen for their ability to dissolve the reactants at elevated temperatures while allowing selective crystallization of the target diastereomer upon cooling.
Q: Can this resolution method be applied to compounds other than tetrahydropapaverine?
A: Yes, the method is broadly applicable to various racemic amines including selegiline (deprenyl) and ephedrine. The versatility lies in the specific substitution patterns of the phenoxypropionic acid resolving agent which can be tuned to match the stereochemical requirements of different amine substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Selegiline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-purity chiral intermediates to drive the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated resolution techniques described in patent CN1197447A can be effectively translated into robust manufacturing processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of selegiline, ephedrine, or tetrahydropapaverine meets the exacting standards required by global regulatory bodies. Our commitment to technical excellence ensures that our clients receive materials that are not only chemically pure but also consistently reliable for their critical drug development programs.
We invite you to engage with our technical procurement team to discuss how our advanced capabilities can support your specific project requirements and timeline. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how optimizing your resolution strategy can impact your overall budget and supply security. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique chemical challenges, ensuring a partnership built on transparency, quality, and mutual success.
