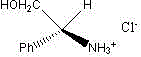
Advanced Synthesis of Chiral (S)-Phenylglycinol Hydrochloride for Commercial Scale-up
Advanced Synthesis of Chiral (S)-Phenylglycinol Hydrochloride for Commercial Scale-up
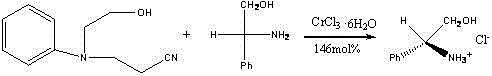
The landscape of chiral intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable synthetic routes. A pivotal development in this sector is documented in patent CN102952026B, which discloses a novel preparation method for chirality (S)-benzene ammonia alcohol hydrochloride, commonly known as (S)-phenylglycinol hydrochloride. This technology represents a significant departure from traditional multi-step syntheses, offering a streamlined one-pot approach that leverages the unique properties of transition metal salts. The patent details a specific reaction system where N-(2-cyanoethyl)-N-(2-hydroxyethyl)aniline reacts with (S)-phenylglycinol in the presence of chromium chloride hexahydrate. This unexpected yet highly effective pathway not only simplifies the production of this critical chiral building block but also highlights the compound's dual utility as a potent organocatalyst for downstream transformations such as Henry reactions. For global procurement and R&D teams, understanding the nuances of this patented methodology is essential for securing a reliable supply chain of high-purity chiral intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral amino alcohols and their corresponding hydrochloride salts has been fraught with challenges that impact both cost and throughput. Conventional methods often rely on the resolution of racemic mixtures using expensive chiral resolving agents, a process that inherently limits the maximum theoretical yield to 50% unless dynamic kinetic resolution is employed. Furthermore, traditional synthetic routes frequently necessitate the use of hazardous reagents, stringent low-temperature conditions, and multiple purification steps involving column chromatography or repeated recrystallizations. These factors contribute to a bloated cost structure and extended lead times, creating bottlenecks for pharmaceutical manufacturers who require large quantities of intermediates for API production. The reliance on stoichiometric amounts of chiral auxiliaries rather than catalytic systems further exacerbates waste generation, posing significant environmental compliance hurdles for modern chemical facilities aiming to reduce their carbon footprint.
The Novel Approach
In stark contrast, the methodology outlined in CN102952026B introduces a paradigm shift by utilizing a Lewis acid-mediated transformation that proceeds in a single operational step. By employing chromium chloride hexahydrate (CrCl3·6H2O) as a catalyst, the process facilitates the in situ generation of hydrogen chloride, which subsequently reacts with excess (S)-phenylglycinol to form the target hydrochloride salt. This approach eliminates the need for external gaseous HCl sources, which are difficult to handle and pose safety risks on a large scale. The reaction operates under reflux conditions in chlorobenzene, a solvent choice that allows for high-temperature operation conducive to driving the equilibrium towards product formation. The simplicity of the workup, which involves merely standing the reaction solution for three days to induce crystallization, drastically reduces labor costs and solvent waste, positioning this technology as a superior alternative for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into CrCl3-Catalyzed Salt Formation
The core innovation of this patent lies in the mechanistic role of chromium chloride hexahydrate acting as a robust Lewis acid within the reaction matrix. Under the thermal energy provided by refluxing chlorobenzene, the hydrated chromium salt interacts with the hydroxyl groups present in the reactants, specifically N-(2-cyanoethyl)-N-(2-hydroxyethyl)aniline. This interaction weakens the O-H bonds and facilitates the release of protons, effectively generating hydrogen chloride within the closed system. This in situ generated acid then protonates the amino group of the (S)-phenylglycinol, leading to the precipitation of the stable ammonium chloride salt. The use of 146 mol% of the chromium catalyst ensures that the acidic environment is maintained throughout the 48-hour reaction period, driving the conversion to completion despite the reversible nature of salt formation. This mechanism bypasses the need for harsh mineral acids, thereby preserving the integrity of the chiral center and preventing racemization, a critical quality attribute for any chiral pharmaceutical ingredient.
Furthermore, the impurity profile of the resulting product is exceptionally clean due to the specificity of the Lewis acid interaction. The reaction conditions are mild enough to avoid degradation of the sensitive cyano and hydroxyl functionalities present in the starting aniline derivative, yet vigorous enough to ensure complete salification of the amine. The crystallization process, driven by the slow standing of the reaction mixture over three days, acts as a self-purification step. As the chiral clear crystals of (S)-phenylglycinol hydrochloride nucleate and grow, they exclude non-chiral impurities and unreacted starting materials from the crystal lattice. This phenomenon results in a product with high optical purity, as evidenced by the specific rotation values reported in the patent data. For R&D directors, this implies that downstream processing can be minimized, potentially allowing the crude crystalline product to be used directly in subsequent catalytic applications without extensive recrystallization.
How to Synthesize (S)-Phenylglycinol Hydrochloride Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and reaction conditions to maximize yield and purity. The patent specifies a precise molar ratio of 1:4.2:1.5 for the aniline derivative, (S)-phenylglycinol, and chromium chloride hexahydrate, respectively. Deviating from these ratios could result in incomplete conversion or difficulties in product isolation. The process begins with the preparation of an anhydrous and oxygen-free environment, typically achieved by purging the reactor with inert gas, to prevent oxidation of the chromium species or hydrolysis of the nitrile group. Following the addition of chlorobenzene as the solvent, the mixture is subjected to reflux for 48 hours. The detailed standardized synthesis steps, including specific temperature controls and workup procedures, are outlined below to ensure reproducibility at scale.
- Mix N-(2-cyanoethyl)-N-(2-hydroxyethyl)aniline, (S)-phenylglycinol, and chromium chloride hexahydrate in a molar ratio of 1: 4.2:1.5 in chlorobenzene solvent.
- Reflux the mixture under anhydrous and oxygen-free conditions for approximately 48 hours to allow the Lewis acid catalysis to proceed.
- Allow the reaction solution to stand undisturbed for 3 days to facilitate the crystallization of the target chiral clear crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility. The shift towards a one-pot catalytic process fundamentally alters the cost structure of producing this chiral intermediate. By eliminating the need for multiple reaction vessels, intermediate isolations, and complex purification trains, the overall manufacturing cycle time is significantly compressed. This efficiency translates directly into lower operational expenditures and a reduced working capital requirement for inventory holding. Moreover, the use of chromium chloride hexahydrate, a commodity chemical, replaces more expensive or hazardous acid sources, contributing to substantial cost savings in raw material procurement. The robustness of the process also enhances supply chain reliability, as it is less susceptible to variations in raw material quality compared to sensitive enzymatic or biocatalytic routes.
- Cost Reduction in Manufacturing: The elimination of stoichiometric resolving agents and the reduction of solvent usage through a simplified workup procedure lead to a drastic decrease in production costs. The ability to generate the necessary acidic conditions in situ removes the capital expenditure associated with specialized corrosion-resistant equipment required for handling gaseous HCl, further optimizing the CAPEX profile of the manufacturing facility.
- Enhanced Supply Chain Reliability: The starting materials, including (S)-phenylglycinol and the aniline derivative, are commercially available from multiple global suppliers, mitigating the risk of single-source dependency. The stability of the reagents allows for long-term storage without significant degradation, enabling manufacturers to maintain strategic stockpiles and buffer against market volatility or logistics disruptions.
- Scalability and Environmental Compliance: The process operates in chlorobenzene, a solvent with well-established recovery and recycling protocols in the fine chemical industry, facilitating easy scale-up from pilot plants to multi-ton production. Additionally, the solid-state crystallization minimizes liquid waste generation, aligning with increasingly stringent environmental regulations and supporting corporate sustainability goals without compromising output volume.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of (S)-phenylglycinol hydrochloride via this novel method. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these aspects is crucial for evaluating the fit of this intermediate within your specific synthetic workflows and supply chain strategies.
Q: What is the primary advantage of the CrCl3-catalyzed method for (S)-phenylglycinol hydrochloride?
A: The primary advantage is the one-step synthesis capability which eliminates complex multi-step protection and deprotection sequences, significantly simplifying the operational workflow and reducing solvent consumption.
Q: Can this chiral salt be used as a catalyst for other reactions?
A: Yes, the synthesized compound demonstrates excellent catalytic performance in nitrile silylation reactions (up to 77.5% yield) and Henry reactions (up to 99.2% conversion), adding significant value beyond its role as an intermediate.
Q: Is the process scalable for industrial production?
A: The process utilizes common solvents like chlorobenzene and stable reagents like chromium chloride hexahydrate, making it highly amenable to scale-up from laboratory to commercial tonnage without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-Phenylglycinol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our technical team has thoroughly analyzed the potential of the CrCl3-catalyzed route described in CN102952026B and is prepared to leverage this knowledge for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market launch is seamless. Our state-of-the-art rigorous QC labs and adherence to stringent purity specifications guarantee that every batch of (S)-phenylglycinol hydrochloride meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to explore how this advanced synthesis method can optimize your manufacturing costs and timelines. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate our commitment to being your trusted partner in fine chemical innovation.
