Advanced Separation of Cephalomannine from Taxol: A Commercial Scale-Up Perspective
Introduction to Advanced Separation Technologies
The isolation of high-purity pharmaceutical intermediates from complex natural extracts remains one of the most critical challenges in modern drug manufacturing. Patent CN102020619B introduces a groundbreaking methodology for the separation of Cephalomannine (also known as Taxol B) from Taxol, addressing the long-standing inefficiencies associated with traditional purification techniques. This technology leverages a selective 1,4-addition reaction to chemically differentiate these structurally analogous compounds, enabling their efficient separation under mild conditions. For R&D directors and procurement specialists, this represents a significant shift away from hazardous heavy metal catalysis toward a more sustainable and economically viable process. The ability to achieve purity levels exceeding 98% with yields greater than 92% positions this method as a superior alternative for the commercial production of taxane intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the separation of Cephalomannine from Taxol has been plagued by severe technical and safety drawbacks that hinder scalable manufacturing. Prior art methods frequently relied on the use of osmium tetroxide (OsO4) to catalyze the oxidation of side-chain double bonds, a process fraught with extreme toxicity and environmental hazards unsuitable for large-scale pharmaceutical operations. Alternative approaches utilizing ozonolysis required expensive, specialized equipment and suffered from poor selectivity, often leading to undesirable oxidation of other functional groups within the sensitive taxane core. Furthermore, halogen addition methods necessitated cryogenic conditions around 0-5°C and the handling of volatile, toxic bromine, creating substantial operational risks and energy costs. These conventional pathways not only compromised worker safety but also resulted in lower overall yields and complex waste treatment requirements, driving up the total cost of ownership for manufacturers.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a mild, room-temperature 1,4-addition reaction that fundamentally alters the polarity of Cephalomannine without affecting Taxol. By dissolving the crude mixture in inexpensive short-chain fatty alcohols such as methanol, ethanol, or isopropanol, and introducing a catalytic amount of an organic base like DBU or N-methylmorpholine, the process selectively derivatizes the Cephalomannine side chain. This chemical modification creates a distinct difference in chromatographic behavior, allowing for the straightforward isolation of the Cephalomannine derivative and the recovery of unreacted Taxol via standard column chromatography. The simplicity of using ambient temperature and common solvents drastically reduces energy consumption and eliminates the need for specialized cryogenic or high-pressure infrastructure.
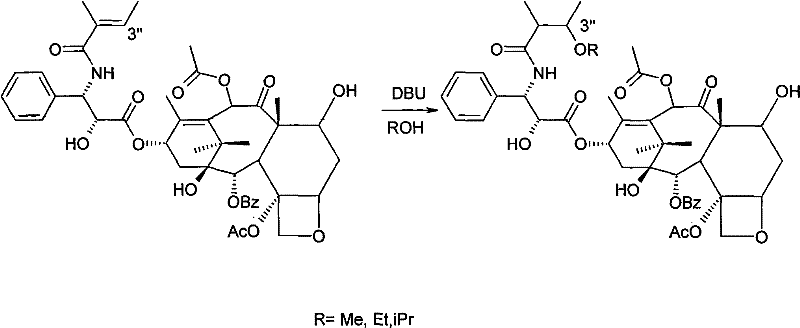
Mechanistic Insights into Organic Base-Catalyzed Derivatization
The core of this technological breakthrough lies in the selective nucleophilic attack on the alpha,beta-unsaturated carbonyl system present in the C-13 side chain of Cephalomannine. Under the influence of an organic alkali catalyst, the short-chain alcohol acts as a nucleophile, undergoing a conjugate addition to the double bond of the cinnamoyl group specific to Cephalomannine. This reaction converts the lipophilic olefin into a more polar ether derivative, specifically a 3'-alkoxy Taxol B derivative, while leaving the saturated or structurally distinct Taxol molecule largely unaffected. This differential reactivity is the key to the high selectivity observed, as it exploits the subtle electronic differences between the two molecules to create a separable physical property gap. The use of mild organic bases ensures that the sensitive oxetane ring and ester linkages of the taxane core remain intact, preserving the integrity of the valuable starting materials.
Following the initial separation, the process employs a clever reversal mechanism to regenerate the native Cephalomannine structure with exceptional fidelity. The isolated 3'-alkoxy derivative is dissolved in toluene and subjected to reflux in the presence of acidified activated carbon, which serves a dual purpose as both an acid catalyst source and a decolorizing agent. This acidic environment facilitates the elimination of the alcohol moiety, effectively reversing the 1,4-addition and restoring the original conjugated double bond system of Cephalomannine. The subsequent recrystallization from an ethanol-water mixture yields the final product with purity levels exceeding 98%, demonstrating the robustness of this reversible derivatization strategy for impurity control.
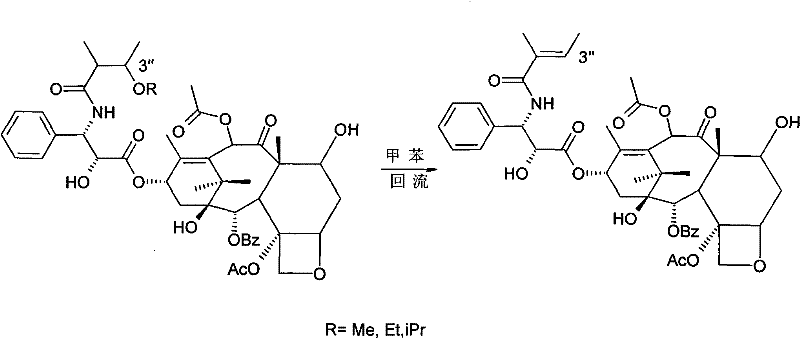
How to Synthesize Cephalomannine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory discovery to pilot-scale production. The procedure begins with the dissolution of the Taxol/Cephalomannine mixture in a selected short-chain alcohol, followed by the controlled addition of the organic base catalyst to initiate the derivatization. After a brief stirring period at room temperature, the reaction mixture is concentrated and subjected to silica gel column chromatography using a gradient of ethyl acetate and petroleum ether to separate the components. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and purification parameters, are provided in the guide below to ensure reproducibility and quality control.
- Dissolve the Taxol and Cephalomannine mixture in a short-chain fatty alcohol and add an organic alkali catalyst to initiate a 1,4-addition reaction at room temperature.
- Concentrate the reaction solution and perform column chromatography separation to isolate the Cephalomannine derivative and recover pure Taxol.
- Dissolve the Cephalomannine derivative in toluene, reflux with acidified activated carbon, and recrystallize from ethanol/water to obtain pure Cephalomannine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this separation technology offers profound strategic advantages regarding cost structure and operational reliability. By eliminating the dependency on scarce and regulated heavy metal catalysts like osmium, manufacturers can significantly reduce raw material costs and avoid the complex regulatory compliance issues associated with heavy metal residue limits in pharmaceutical products. The substitution of expensive ozonizers and cryogenic cooling systems with standard reactor vessels and ambient temperature processing leads to a drastic simplification of the capital equipment requirements. This reduction in process complexity directly translates to lower maintenance overheads and reduced downtime, ensuring a more consistent and reliable supply of high-purity intermediates for downstream synthesis.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of high-cost reagents with commodity chemicals. Short-chain alcohols and organic bases are inexpensive and globally available, contrasting sharply with the volatility and cost of osmium salts or bromine. Furthermore, the mild reaction conditions minimize energy consumption by removing the need for continuous refrigeration or high-pressure gas generation. The high recovery yield of both Taxol and Cephalomannine ensures that valuable starting materials are not lost to degradation, maximizing the return on investment for every kilogram of raw extract processed.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally accessible reagents that are not subject to the same geopolitical or logistical constraints as specialized catalytic metals. The robustness of the reaction against minor variations in temperature or mixing rates reduces the risk of batch failures, thereby stabilizing production schedules. Additionally, the simplified workflow reduces the number of unit operations required, shortening the overall cycle time from raw material intake to finished intermediate. This efficiency allows manufacturers to respond more agilely to market demand fluctuations without compromising on product quality or delivery timelines.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this method aligns perfectly with green chemistry principles by reducing toxic waste generation. The absence of heavy metals simplifies wastewater treatment protocols and lowers the burden on effluent processing facilities, facilitating easier permitting for expansion. The process is inherently scalable because it relies on standard liquid-phase reactions that translate linearly from liter-scale to cubic-meter-scale reactors. This scalability ensures that production capacity can be increased to meet commercial demands of 100 kgs to 100 MT annually without requiring fundamental changes to the chemical pathway or safety infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this separation technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the method's suitability for your specific manufacturing needs. Understanding these nuances is essential for R&D teams planning to integrate this pathway into their existing production lines.
Q: Why is separating Cephalomannine from Taxol traditionally difficult?
A: Traditional separation is challenging because Cephalomannine and Taxol possess extremely similar chemical structures, differing only by the carboxamido-group side chain. Conventional methods often rely on toxic heavy metals like osmium tetroxide or non-selective ozonolysis, which pose significant safety hazards and fail to handle unpurified mixtures effectively.
Q: What are the advantages of the 1,4-addition separation method?
A: This novel method utilizes mild reaction conditions at room temperature using inexpensive short-chain alcohols and organic bases. It avoids toxic reagents and expensive equipment, achieving high yields (>95%) and high purity (>98%) while significantly simplifying the downstream purification process.
Q: Is this process suitable for industrial scale-up?
A: Yes, the process is highly amenable to industrialization. It uses cheap, readily available raw materials and avoids harsh conditions like cryogenic temperatures or high-pressure ozone generation, making it safer and more cost-effective for large-scale commercial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cephalomannine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-quality taxane intermediates for the global pharmaceutical market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Cephalomannine or Taxol B delivered adheres to the highest international standards. We are committed to leveraging advanced separation technologies like the one described in CN102020619B to optimize our manufacturing processes and deliver superior value to our partners.
We invite you to collaborate with us to explore how these innovative separation strategies can enhance your supply chain efficiency and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in complex natural product isolation can support your long-term business goals.
