Advanced Semi-Synthesis of C4-Modified 9(R)-Hydrogenated-1-Deoxypaclitaxel Derivatives for Commercial Scale
Advanced Semi-Synthesis of C4-Modified 9(R)-Hydrogenated-1-Deoxypaclitaxel Derivatives for Commercial Scale
The pharmaceutical industry continuously seeks next-generation antineoplastic agents that overcome the limitations of first-generation chemotherapies, particularly regarding solubility and drug resistance. Patent CN102558105A introduces a groundbreaking methodology for the preparation of C4-modified 9(R)-hydrogenated-1-deoxypaclitaxel derivatives, representing a significant evolution in taxane chemistry. This technology leverages the abundant natural precursor 1-dehydroxybaccatin VI, extracted efficiently from Taxus chinensis, to construct complex taxane skeletons with enhanced biological profiles. Unlike traditional extraction methods that rely on scarce resources, this semi-synthetic approach offers a sustainable pathway to produce high-purity intermediates. The resulting compounds exhibit potent antitumor activity comparable to natural paclitaxel but with improved physicochemical properties, addressing critical pain points in oncology drug development such as poor aqueous solubility and the emergence of multidrug-resistant tumor phenotypes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the supply chain for paclitaxel and its analogs has been constrained by the reliance on natural extraction from the bark of Pacific yew trees or the needles of European yew, processes that are ecologically damaging and yield extremely low quantities of the active pharmaceutical ingredient. Furthermore, total synthesis of taxanes is notoriously difficult due to the molecule's intricate stereochemistry and the presence of multiple chiral centers, leading to prohibitively high costs and low overall yields that are unsuitable for commercial manufacturing. Conventional semi-synthetic routes often utilize 10-deacetylbaccatin III (10-DAB) or baccatin III, which, while more available than paclitaxel itself, still face supply bottlenecks and require harsh reaction conditions that can compromise the integrity of the sensitive oxetane ring. These legacy methods frequently struggle with regioselectivity during functionalization, resulting in complex impurity profiles that necessitate expensive and wasteful purification steps, ultimately inflating the cost of goods sold for the final API.
The Novel Approach
The innovative strategy detailed in the patent circumvents these bottlenecks by utilizing 1-dehydroxybaccatin VI as the foundational building block, a precursor that is present in much higher concentrations in renewable plant sources like Taxus chinensis. This route employs a sophisticated sequence of selective protection and deprotection strategies, specifically targeting the C4, C7, C9, C10, and C13 positions to achieve precise structural modifications without affecting the core taxane skeleton. By introducing specific acyl groups at the C4 position and modifying the C9 carbonyl to a hydroxyl group (9(R)-hydrogenation), the process generates derivatives with distinct pharmacological advantages. 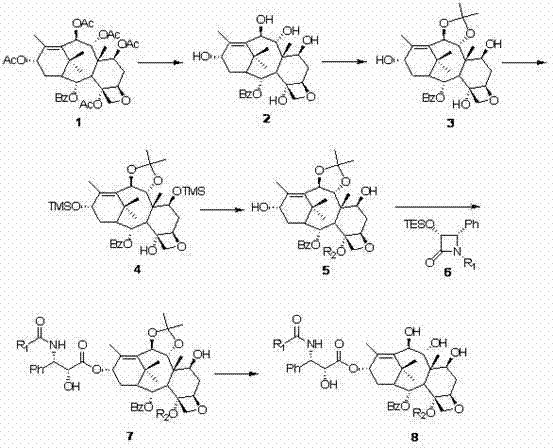
This method not only streamlines the synthesis but also enhances the water solubility of the final product, a critical factor for formulation development. The ability to couple various side chains, such as N-caproyl or N-benzoyl beta-lactams, allows for the rapid generation of a library of analogs to optimize efficacy against resistant cancer cell lines, providing a versatile platform for drug discovery and development.
Mechanistic Insights into Selective Protection and Beta-Lactam Coupling
The core of this synthetic success lies in the meticulous control of regioselectivity during the functionalization of the polyol taxane core. The process initiates with the global deacetylation of 1-dehydroxybaccatin VI using hydrazine hydrate, followed by the selective formation of a 9,10-O-isopropylidene ketal using 2,2-dimethoxypropane and Montmorillonite K10 clay. This step is crucial as it masks the C9 and C10 hydroxyls, preventing unwanted side reactions during subsequent acylation steps. Following this, the C7 and C13 hydroxyl groups are protected as trimethylsilyl (TMS) ethers using TMSCl and imidazole, leaving the C4 hydroxyl group uniquely exposed for selective acylation. This orthogonal protection strategy ensures that the introduction of cyclopropanecarbonyl or acetoacetyl groups occurs exclusively at the C4 position, eliminating the formation of regioisomeric impurities that are common in less controlled syntheses.
Following the C4 modification and removal of the silyl protecting groups using tetrabutylammonium fluoride (TBAF), the pivotal C13 side-chain installation is performed via a beta-lactam coupling reaction. Utilizing strong bases like sodium hexamethyldisilazide (NaHMDS) at low temperatures (-30°C), the C13 hydroxyl attacks the beta-lactam ring, opening it to form the essential ester linkage found in all bioactive taxanes. This mechanism preserves the stereochemistry at the C13 position, which is vital for binding to the microtubule protein. The final deprotection of the isopropylidene group under mild acidic conditions reveals the 9,10-diol system, completing the transformation into the target 9(R)-hydrogenated-1-deoxypaclitaxel derivative. This mechanistic precision ensures high purity and consistent batch-to-batch reproducibility, which are non-negotiable requirements for regulatory approval.
How to Synthesize C4-Modified 9(R)-Hydrogenated-1-Deoxypaclitaxel Efficiently
The synthesis of these advanced taxane derivatives requires a disciplined approach to reaction conditions and stoichiometry to maximize yield and minimize waste. The process is designed to be robust, utilizing commercially available reagents and standard laboratory equipment, making it highly adaptable for pilot and commercial scale operations. Operators must pay close attention to temperature control during the beta-lactam coupling phase and the careful monitoring of pH during the final deprotection steps to prevent degradation of the sensitive oxetane ring. The following guide outlines the standardized operational procedure derived from the patent examples, ensuring that technical teams can replicate the high selectivity and yield reported in the intellectual property.
- Deacetylation of 1-dehydroxybaccatin VI using hydrazine hydrate in ethanol to obtain the penta-deacetylated intermediate.
- Selective protection of the 9,10-diol system using 2,2-dimethoxypropane and Mont K10 catalyst to form the isopropylidene derivative.
- Silylation of the 7,13-hydroxyl groups using TMSCl and imidazole, followed by selective C4-acylation with cyclopropanecarbonyl chloride or diketene.
- Deprotection of silyl groups using TBAF to reveal the C4-acylated baccatin core ready for side-chain attachment.
- Coupling with protected beta-lactam side chains (e.g., N-caproyl or N-benzoyl) using NaHMDS base, followed by global deprotection to yield the final taxane derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift towards this specific semi-synthetic route offers tangible strategic benefits that extend beyond simple chemical novelty. By anchoring the supply chain on 1-dehydroxybaccatin VI, a precursor with superior natural abundance compared to traditional baccatins, manufacturers can significantly mitigate the risks associated with raw material scarcity and price volatility. This abundance translates directly into a more stable supply continuity, reducing the likelihood of production stoppages due to precursor shortages. Furthermore, the streamlined nature of the synthesis, which avoids exotic catalysts or extreme conditions, simplifies the manufacturing process, thereby lowering the barrier to entry for contract manufacturing organizations (CMOs) and enhancing overall supply chain resilience.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive and scarce starting materials in favor of the readily available 1-dehydroxybaccatin VI. The high selectivity of the protection-deprotection sequence minimizes the formation of difficult-to-separate impurities, which drastically reduces the consumption of chromatography media and solvents during purification. Additionally, the use of heterogeneous catalysts like Mont K10 allows for easier recovery and reuse, further driving down the operational expenditure per kilogram of produced intermediate. These efficiencies compound to offer a substantially lower cost of goods sold compared to legacy taxane synthesis routes.
- Enhanced Supply Chain Reliability: Sourcing 1-dehydroxybaccatin VI from cultivated Taxus chinensis provides a renewable and scalable feedstock that is less susceptible to the geopolitical and environmental fluctuations that affect wild-harvested yew species. The synthetic route itself relies on commodity chemicals such as hydrazine hydrate, TMSCl, and standard beta-lactams, ensuring that the supply chain is not dependent on single-source specialty reagents. This diversification of input materials strengthens the supply chain against disruptions, ensuring reliable delivery schedules for downstream API manufacturers and pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing reaction conditions that are easily managed in large-scale reactors, such as ambient temperature acylations and controlled low-temperature couplings. The avoidance of heavy metal catalysts and the use of recyclable solid supports align with modern green chemistry principles, simplifying waste treatment and reducing the environmental footprint of the manufacturing facility. This compliance with stringent environmental regulations facilitates faster regulatory approvals and reduces the long-term liability associated with hazardous waste disposal, making it an attractive option for sustainable pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is critical for stakeholders evaluating its potential for integration into their existing portfolios. The following questions address common inquiries regarding the biological efficacy, raw material sourcing, and process robustness of the C4-modified derivatives. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering a transparent view of the technology's capabilities and limitations for informed decision-making.
Q: What is the primary advantage of using 1-dehydroxybaccatin VI over traditional taxol precursors?
A: 1-Dehydroxybaccatin VI is found in significantly higher concentrations in Taxus chinensis compared to baccatin III or 10-DAB, making it a more abundant and cost-effective starting material for large-scale semi-synthesis while maintaining the essential taxane skeleton.
Q: How does the C4 modification impact the biological profile of the derivative?
A: Modifying the C4 position, combined with 9(R)-hydrogenation and 1-deoxygenation, has been shown to improve water solubility and potentially reduce multidrug resistance (MDR) compared to natural paclitaxel, offering a better therapeutic index for resistant cancer lines.
Q: Is this synthetic route scalable for industrial production?
A: Yes, the process utilizes standard organic transformations such as acylation, silylation, and beta-lactam coupling which are well-established in GMP environments. The use of robust reagents like Mont K10 and straightforward purification steps supports commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9(R)-Hydrogenated-1-Deoxypaclitaxel Derivative Supplier
As the demand for next-generation oncology therapeutics grows, the ability to reliably produce complex taxane intermediates at scale becomes a decisive competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this capability, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this patented technology to life. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of C4-modified 9(R)-hydrogenated-1-deoxypaclitaxel derivative meets the highest standards required for clinical and commercial applications. We understand the critical nature of timeline and quality in the pharmaceutical sector and are committed to delivering consistent, high-performance intermediates.
We invite global pharmaceutical partners to collaborate with us to explore the full potential of this innovative synthetic route. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and project timelines. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, allowing you to make data-driven decisions that optimize your supply chain and accelerate your drug development programs.
