Scalable Green Synthesis of Arylmethyl Hydrocarbyl Sulfones for Pharmaceutical Intermediates
The landscape of fine chemical synthesis is undergoing a paradigm shift towards greener, more sustainable methodologies, particularly for high-value scaffolds like sulfones. Patent CN110511166B introduces a groundbreaking approach for the synthesis of arylmethyl hydrocarbyl sulfones, utilizing a recyclable ion-exchange resin catalyst, Amberlyst-15, in an aqueous medium. This innovation addresses critical pain points in traditional organic synthesis by replacing toxic organic solvents and expensive transition metal catalysts with a robust, heterogeneous system. The technology enables a direct three-component coupling of readily available aryl or alkyl aldehydes, sodium hydrocarbyl sulfinates, and polymethoxybenzenes. For R&D directors and process chemists, this represents a significant leap forward in atom economy and operational simplicity, delivering high-purity intermediates essential for pharmaceutical and agrochemical applications without the burden of heavy metal residues.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of arylmethyl hydrocarbyl sulfones has relied on multi-step sequences that are both resource-intensive and environmentally taxing. Traditional pathways often involve the initial conversion of aryl aldehydes into reactive intermediates, such as alpha-amidosulfones or sulfonyl hydrazones, before undergoing a subsequent arylation step. These legacy methods frequently necessitate the use of stoichiometric amounts of Lewis acids like iron(III) chloride or precious metal catalysts such as palladium complexes. The reliance on homogeneous metal catalysts introduces severe complications in downstream processing, requiring rigorous purification steps to meet stringent regulatory limits for residual metals in pharmaceutical ingredients. Furthermore, the requirement for anhydrous organic solvents and harsh reaction conditions often leads to lower overall yields and generates substantial hazardous waste, complicating the supply chain and inflating production costs for reliable arylmethyl hydrocarbyl sulfone suppliers.
The Novel Approach
In stark contrast, the methodology disclosed in CN110511166B streamlines the synthesis into a single, efficient pot. By leveraging the strong acidic properties of the macroreticular resin Amberlyst-15, the reaction proceeds smoothly in water at a mild temperature of 60 °C. This one-pot strategy eliminates the need for isolating unstable intermediates, thereby reducing material loss and handling time. The heterogeneous nature of the catalyst allows for simple filtration and reuse, drastically cutting down on catalyst consumption costs. Moreover, the use of water as the primary reaction medium not only enhances safety by removing flammable organic solvents but also simplifies work-up procedures. This novel approach ensures cost reduction in pharmaceutical intermediates manufacturing by minimizing waste treatment expenses and maximizing throughput through a simplified operational workflow.

Mechanistic Insights into Amberlyst-15 Catalyzed Three-Component Coupling
The core of this synthetic breakthrough lies in the dual activation capability of the sulfonic acid groups anchored on the Amberlyst-15 polymer matrix. The mechanism initiates with the protonation of the carbonyl oxygen of the aldehyde, significantly enhancing its electrophilicity. This activated species then undergoes nucleophilic attack by the sulfur atom of the sodium sulfinate, forming a transient hemithioacetal intermediate. Subsequently, the polymethoxybenzene, acting as a highly electron-rich aromatic nucleophile due to the methoxy substituents, attacks the benzylic position. This sequence effectively constructs both the C-S and C-C bonds in a concerted fashion under thermodynamic control. The resin's porous structure facilitates the diffusion of reactants while stabilizing the transition states, ensuring high conversion rates even with sterically hindered substrates. This mechanistic pathway avoids the generation of free radical species often associated with metal-catalyzed processes, resulting in a cleaner reaction profile with fewer side products.
From an impurity control perspective, this metal-free protocol offers distinct advantages for the production of high-purity arylmethyl hydrocarbyl sulfones. The absence of transition metals like palladium or iron eliminates the risk of metal-catalyzed decomposition or unwanted cross-coupling side reactions that can plague complex molecule synthesis. The primary impurities are typically unreacted starting materials or minor regioisomers, which are easily removed via standard silica gel chromatography or crystallization. The patent data demonstrates exceptional substrate tolerance, accommodating electron-withdrawing groups such as nitro and halogen substituents on the aldehyde, as well as electron-donating groups on the sulfinate. For instance, the reaction of 4-chlorobenzaldehyde with sodium benzenesulfinate yielded the corresponding sulfone in 91% yield, showcasing the robustness of the catalytic system against deactivating groups. This consistency is vital for maintaining batch-to-batch reproducibility in commercial scale-up of complex pharmaceutical intermediates.
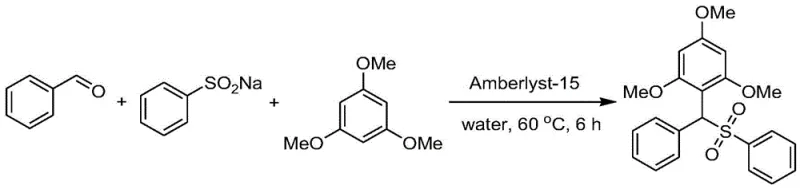
How to Synthesize Arylmethyl Hydrocarbyl Sulfone Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and catalyst longevity. The standard protocol involves charging a reaction vessel with the aldehyde, sodium sulfinate, and polymethoxybenzene in a molar ratio optimized for complete conversion, typically around 1.5:1.0:1.0. Water is added as the solvent, followed by the addition of Amberlyst-15 resin, usually at 50% relative to the mass of the limiting reagent. The mixture is then heated to 60 °C and stirred for approximately 6 hours. Upon completion, the solid catalyst is recovered by simple filtration, washed, and dried for reuse, while the product is extracted from the aqueous filtrate. Detailed standardized synthesis steps see the guide below.
- Combine aryl/alkyl aldehyde, sodium hydrocarbyl sulfinate, and polymethoxybenzene in water with Amberlyst-15 catalyst.
- Heat the reaction mixture to 60 °C and stir for 6 hours to facilitate C-C and C-S bond formation.
- Filter the recyclable resin catalyst, extract the product with ethyl acetate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Amberlyst-15 catalyzed process translates into tangible strategic benefits beyond mere chemical efficiency. The shift from precious metal catalysts to a reusable polymeric resin fundamentally alters the cost structure of the manufacturing process. By eliminating the need for expensive palladium or specialized ligands, the raw material costs are significantly reduced. Furthermore, the ability to recycle the catalyst multiple times without significant loss of activity extends the lifecycle of the catalytic material, driving down the cost per kilogram of the final product. The use of water as a solvent also removes the logistical and safety burdens associated with the storage and disposal of large volumes of volatile organic compounds (VOCs), leading to substantial cost savings in waste management and regulatory compliance.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for costly metal scavenging resins and extensive purification protocols required to meet ppm-level metal specifications. This simplification of the downstream processing train reduces labor hours and consumable usage. Additionally, the one-pot nature of the reaction minimizes solvent swaps and intermediate isolation steps, which are traditionally major drivers of operational expenditure in fine chemical synthesis. The overall process intensity is lowered, allowing for higher throughput in existing reactor infrastructure without the need for specialized high-pressure or cryogenic equipment.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis—simple benzaldehydes, sodium sulfinates, and polymethoxybenzenes—are commodity chemicals with robust global supply chains. Unlike specialized organometallic reagents that may suffer from supply bottlenecks or long lead times, these feedstocks are readily available from multiple vendors. This diversification of supply sources mitigates the risk of production stoppages due to raw material shortages. The mild reaction conditions also reduce the risk of thermal runaways or safety incidents, ensuring consistent delivery schedules and reducing lead time for high-purity arylmethyl hydrocarbyl sulfones.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is straightforward due to the heterogeneous nature of the catalysis and the use of water. Heat transfer and mixing are more manageable in aqueous systems compared to viscous organic slurries. From an environmental standpoint, the process aligns perfectly with green chemistry principles, generating minimal hazardous waste. This facilitates easier permitting for new manufacturing lines and enhances the sustainability profile of the final product, a key metric for modern pharmaceutical buyers who prioritize eco-friendly supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on substrate compatibility and process robustness. Understanding these nuances is critical for process development teams evaluating the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: What are the primary advantages of using Amberlyst-15 over traditional metal catalysts?
A: Amberlyst-15 eliminates heavy metal contamination risks, simplifies purification, and allows for catalyst recycling, significantly reducing environmental impact and downstream processing costs.
Q: Can this synthesis method accommodate diverse substrate scopes?
A: Yes, the protocol demonstrates excellent tolerance for various aryl, alkyl, and heteroaryl aldehydes, as well as different sulfinate salts and polymethoxybenzenes, yielding products with 70% to 93% efficiency.
Q: Is the process suitable for large-scale commercial production?
A: Absolutely. The use of water as a solvent, mild reaction conditions (60 °C), and heterogeneous catalysis make the process inherently safer and easier to scale compared to anhydrous or high-pressure methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arylmethyl Hydrocarbyl Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Amberlyst-15 catalyzed synthesis for producing high-value sulfone intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped to handle heterogeneous catalysis and aqueous processing with the highest standards of safety and efficiency. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of arylmethyl hydrocarbyl sulfone meets the exacting requirements of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this green synthesis technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how we can optimize your supply chain for arylmethyl hydrocarbyl sulfones.
