Advanced Purification Technology for Benzcycloquine Ammonium Bromide: Scaling High-Purity Pharmaceutical Intermediates
The pharmaceutical industry continuously demands higher purity standards for active pharmaceutical ingredients and their precursors, particularly for complex quaternary ammonium compounds used in respiratory and cardiovascular therapies. Patent CN111377920B introduces a groundbreaking refinement in the purification of benzcycloquine ammonium bromide, a critical M-receptor blocker. This technology addresses the longstanding challenge of removing inorganic salts and organic byproducts that persist through conventional acetone-based recrystallization methods. By shifting to a mixed solvent system of water and organic alcohols, specifically isopropanol or ethanol, manufacturers can achieve purity levels exceeding 99.80% in a single processing step. This innovation is not merely a laboratory curiosity but a robust industrial solution that aligns with the rigorous quality expectations of a reliable pharmaceutical intermediates supplier. The ability to consistently deliver high-purity materials while minimizing solvent waste represents a significant leap forward in process chemistry, offering tangible benefits for both regulatory compliance and production efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of quinine compounds containing quaternary ammonium groups has relied heavily on acetone as the primary recrystallization solvent. While acetone is a common organic solvent, it exhibits significant thermodynamic limitations when applied to these specific ionic structures. The solubility profile of acetone often fails to differentiate effectively between the target molecule and inorganic salt impurities, leading to co-crystallization or entrapment of unwanted byproducts. Consequently, manufacturers are forced to perform multiple recrystallization cycles to approach acceptable purity standards, which drastically reduces the overall yield and increases operational costs. Furthermore, the extensive use of acetone necessitates complex solvent recovery systems to meet environmental regulations, adding another layer of financial and logistical burden to the production line. These inefficiencies create bottlenecks in the commercial scale-up of complex pharmaceutical intermediates, making the supply chain vulnerable to delays and cost volatility.
The Novel Approach
In stark contrast, the method disclosed in CN111377920B utilizes a mixed solution of water and an organic solvent, preferably isopropanol, to redefine the purification landscape. This binary solvent system creates a unique solubility environment where the target quaternary ammonium compound dissolves readily at elevated temperatures but crystallizes with high selectivity upon cooling. The presence of water plays a crucial role in solvating and retaining inorganic salts in the mother liquor, preventing them from contaminating the final crystal lattice. This approach allows for the effective removal of reaction byproducts and unreacted raw materials in a single pass, eliminating the need for repetitive processing. The result is a streamlined workflow that significantly simplifies the work-up of the solvent and enhances the overall economic viability of the manufacturing process. For procurement teams, this translates to cost reduction in API manufacturing without compromising on the stringent quality specifications required for medicinal raw materials.
Mechanistic Insights into Aqueous Isopropanol Recrystallization
The core of this technological advancement lies in the precise manipulation of solubility parameters through the use of a water-isopropanol mixture. When the crude compound is introduced to this mixed solvent and heated to 3-7°C above the boiling point, the system achieves a state of complete dissolution that is thermodynamically favorable for the target molecule but not for the impurities. The specific volume ratio of organic solvent to water, optimally between 95:5 and 80:20, is critical; it ensures that the dielectric constant of the medium is tuned to keep ionic impurities solvated while allowing the organic salt to precipitate out as the temperature drops. This selective crystallization mechanism is far superior to single-solvent systems, as it leverages the differential solubility of the components to drive purification. By filtering the solution while hot, any insoluble particulate matter is removed before crystallization begins, ensuring that the resulting solid is composed almost exclusively of the desired benzcycloquine ammonium bromide.
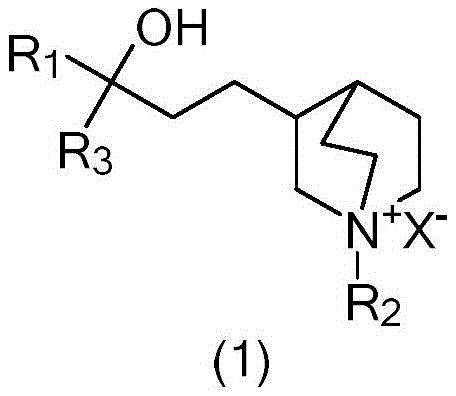
Furthermore, the structural integrity of the quaternary ammonium group is preserved throughout this mild thermal process, avoiding the degradation often seen in harsher purification regimes. The general formula (1) represents a class of compounds where R1, R2, and R3 can vary, yet the refining method remains robust across these variations. Specifically, when R1 is cyclopentyl and R2 is methyl, forming benzcycloquine ammonium bromide, the method demonstrates exceptional efficacy. The crystal lattice formed during the cooling phase is highly ordered, which inherently excludes impurities that do not fit the geometric or electronic constraints of the lattice. This phenomenon is crucial for R&D directors focused on the impurity profile, as it ensures that the final product meets the strict limits for residue on ignition, often as low as 0.010%. The ability to control the polymorphic form and crystal size distribution through cooling rates further enhances the filterability and drying characteristics of the product, facilitating smoother downstream processing.
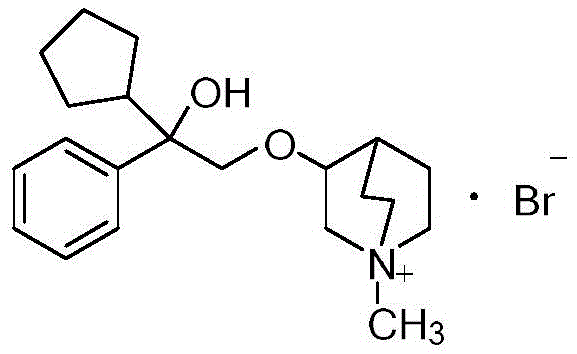
How to Synthesize Benzcycloquine Ammonium Bromide Efficiently
Implementing this refining protocol requires careful attention to solvent ratios and thermal controls to maximize yield and purity. The process begins with the preparation of the crude benzcycloquine ammonium bromide, which typically enters this stage with a purity between 60% and 80%. The key to success lies in the preparation of the 95% aqueous isopropanol solution, which serves as the crystallization medium. Operators must ensure that the heating phase reaches the specified temperature margin above the boiling point to guarantee a clear, homogeneous solution before hot filtration. This step is non-negotiable for achieving the reported yields of up to 98% and purity levels greater than 99.80%. The following guide outlines the standardized operational parameters derived from the patent examples, providing a clear roadmap for technical teams to replicate these results in a pilot or production setting. Detailed standardized synthesis steps are provided in the guide below.
- Prepare a mixed solvent system comprising isopropanol and water in a volume ratio of 95: 5 to 80:20, ensuring the solution is ready for dissolution.
- Mix the crude quaternary ammonium compound with the solvent and heat to 3-7°C above the boiling point until the solution becomes completely clear.
- Perform hot filtration to remove insoluble impurities, then cool the filtrate to induce crystallization, followed by suction filtration and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this refining technology offers profound strategic advantages beyond mere technical specifications. The shift from acetone to an isopropanol-water system directly addresses the volatility of solvent costs and the regulatory pressures surrounding volatile organic compound (VOC) emissions. By reducing the number of recrystallization cycles from multiple steps to a single operation, the process drastically cuts down on energy consumption and labor hours. This efficiency gain is critical for reducing lead time for high-purity active ingredients, allowing manufacturers to respond more agilely to market demands. Moreover, the simplified solvent recovery process due to the lower toxicity and easier handling of isopropanol compared to acetone enhances workplace safety and reduces environmental compliance costs. These factors collectively contribute to a more resilient and cost-effective supply chain, ensuring continuity of supply for critical respiratory medications.
- Cost Reduction in Manufacturing: The elimination of multiple recrystallization steps significantly lowers the operational expenditure associated with solvent purchase, heating, and cooling. Since the process achieves high purity in a single pass, the loss of product typically associated with repeated mother liquor discards is minimized, leading to a substantial increase in overall material yield. Additionally, the reduced volume of organic solvent required per kilogram of product lowers the burden on solvent recovery distillation columns, saving energy and maintenance costs. This qualitative improvement in process efficiency translates directly to a more competitive pricing structure for the final API intermediate without sacrificing quality margins.
- Enhanced Supply Chain Reliability: The robustness of the aqueous isopropanol system ensures consistent batch-to-batch quality, which is essential for maintaining long-term supply contracts with major pharmaceutical companies. The use of readily available and stable solvents like isopropanol mitigates the risk of supply disruptions that can occur with more specialized or regulated solvents. Furthermore, the high yield and predictable crystallization behavior reduce the likelihood of batch failures or the need for re-processing, which are common causes of delivery delays. This reliability strengthens the partnership between the chemical manufacturer and the drug developer, fostering trust and long-term collaboration in the competitive pharmaceutical market.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the use of standard unit operations like heating, filtration, and crystallization. The reduction in organic solvent usage aligns with green chemistry principles, significantly lowering the environmental footprint of the manufacturing facility. This compliance with environmental standards is increasingly becoming a prerequisite for vendor qualification by top-tier global pharmaceutical companies. By adopting this eco-friendly refining method, manufacturers can position themselves as preferred partners for sustainable drug production, meeting both corporate social responsibility goals and regulatory requirements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this refining technology. They are derived from the specific data points and comparative examples provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is essential for technical teams evaluating the integration of this process into existing production lines. The answers reflect the proven capabilities of the method to handle crude inputs with varying impurity profiles while delivering consistent high-quality output.
Q: Why is the isopropanol-water system superior to acetone for refining quaternary ammonium salts?
A: Conventional acetone recrystallization often fails to effectively remove inorganic salts and requires multiple cycles. The isopropanol-water mixed solvent system described in patent CN111377920B allows for single-step recrystallization with significantly higher yield (up to 98%) and superior removal of inorganic impurities, achieving purity levels exceeding 99.80%.
Q: What are the critical temperature controls for this purification process?
A: To ensure maximum dissolution of the crude compound and effective separation of impurities, the solution must be heated to the boiling point of the solvent mixture, specifically 3-7°C above the boiling point. This thermal margin ensures a clear solution prior to hot filtration, which is critical for preventing premature crystallization of impurities.
Q: Can this refining method be scaled for commercial API production?
A: Yes, the process is designed for industrial scalability. It reduces the consumption of organic solvents compared to traditional methods and simplifies the work-up procedure by eliminating the need for multiple recrystallization steps. This makes it highly suitable for commercial scale-up of complex pharmaceutical intermediates while maintaining environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzcycloquine Ammonium Bromide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent data to commercial reality requires deep technical expertise and robust infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the CN111377920B refining process are implemented with precision. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the >99.80% purity and low residue on ignition levels demanded by modern pharmacopoeias. We understand the critical nature of quaternary ammonium intermediates in the synthesis of respiratory therapeutics and are committed to delivering materials that meet the highest global standards. Our team of chemists and engineers works collaboratively to optimize every step of the synthesis and purification, guaranteeing supply continuity for our partners.
We invite global pharmaceutical companies to engage with our technical procurement team to discuss how this advanced refining technology can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain insights into how switching to this aqueous isopropanol method can reduce your overall cost of goods sold. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project needs. Our goal is to provide not just a chemical product, but a comprehensive solution that enhances your R&D efficiency and commercial competitiveness. Let us help you secure a stable, high-quality supply of benzcycloquine ammonium bromide for your next clinical or commercial campaign.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
