Advanced Synthesis of 10-Deacetylate-9(R)-Hydrogenation-1-Deoxypaclitaxel Analogues for Commercial Scale
The pharmaceutical industry continuously seeks robust alternatives to natural taxanes to overcome supply constraints and resistance issues, and patent CN101353333B presents a significant breakthrough in this domain. This specific intellectual property details the preparation of a 10-deacetylate-9(R)-hydrogenation-1-deoxypaclitaxel analogue, a compound engineered to retain the potent antitumor activity of natural paclitaxel while mitigating its notorious multidrug resistance and severe side effects. The innovation lies in a semi-synthetic approach that utilizes 1-deoxy baccatin VI as a foundational precursor, a strategic choice that leverages more abundant natural resources compared to the extraction of paclitaxel itself. By modifying the taxane skeleton at the C9 and C10 positions, this technology offers a viable pathway for producing high-purity pharmaceutical intermediates that are critical for next-generation oncology treatments. For R&D directors and procurement specialists, understanding this patent is essential as it represents a shift towards more sustainable and chemically efficient manufacturing of complex antitumor agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for producing paclitaxel and its derivatives often rely heavily on the direct extraction from Taxus plants, a process plagued by extremely low natural content and significant ecological impact. Furthermore, conventional semi-synthetic routes frequently encounter challenges related to the scarcity of key starting materials like baccatin III, leading to supply chain bottlenecks and volatile pricing structures. The chemical complexity of the taxane core also means that traditional synthesis often involves harsh conditions that can compromise yield and generate difficult-to-remove impurities, thereby increasing the cost of goods sold. Additionally, the inherent multidrug resistance associated with natural paclitaxel limits its clinical efficacy, necessitating the development of analogues that can bypass P-glycoprotein mediated efflux. These cumulative factors create a precarious environment for supply chain heads who require consistent, high-volume access to reliable pharmaceutical intermediate supplies without the risk of raw material shortages.
The Novel Approach
In contrast, the method disclosed in CN101353333B introduces a streamlined synthetic strategy that begins with 1-deoxy baccatin VI, a precursor available in higher concentrations from specific Taxus species like Taxus cuspidata. This novel approach maintains the essential ring skeleton and functional groups required for biological activity while introducing specific modifications at the C9 and C10 positions to enhance therapeutic profiles. The process is characterized by simple and convenient operations, utilizing standard reagents such as hydrazine hydrate and dimethylamino pyridine under controlled conditions that ensure good selectivity. By avoiding the need for rare natural extracts and focusing on a modular semi-synthetic pathway, this method drastically simplifies the manufacturing landscape. Consequently, this represents a substantial opportunity for cost reduction in API manufacturing, as the improved yields and accessible raw materials translate directly into more stable and economical production cycles for commercial scale-up of complex taxanes.
Mechanistic Insights into Semi-Synthetic Taxane Modification
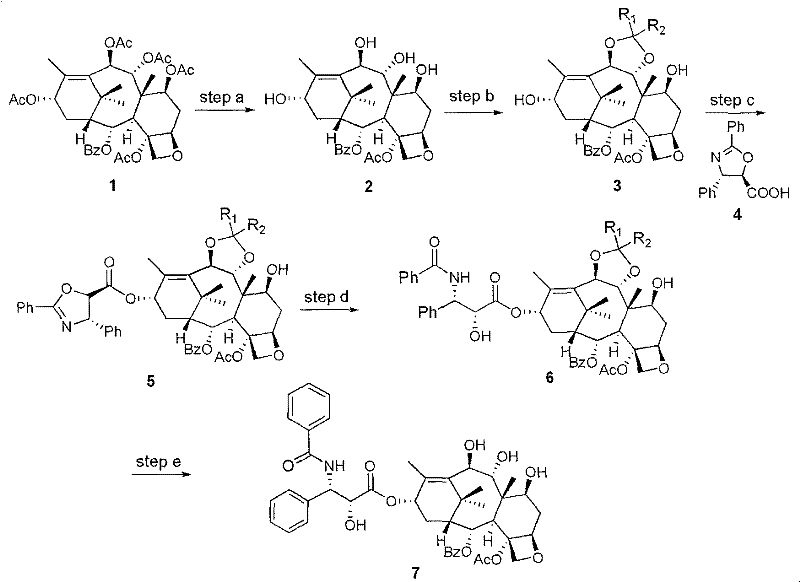
The core of this technology involves a precise sequence of chemical transformations that modify the taxane core without disrupting its bioactive conformation. The process initiates with the deacetylation of 1-deoxy baccatin VI using hydrazine hydrate in ethanol, a reaction that selectively removes acetyl groups to expose hydroxyl functionalities necessary for subsequent modifications. This is followed by a protection step where the 9,10-diol system is converted into an acetonide using 2,2-dimethoxypropane and an acid catalyst like camphorsulfonic acid or Montmorillonite K10. This protection is crucial as it directs the subsequent esterification reaction specifically to the C13 hydroxyl group, ensuring the correct attachment of the paclitaxel side chain. The stereochemistry at the C9 position is carefully managed to achieve the 9(R)-hydrogenation configuration, which is vital for the analogue's interaction with tubulin and its ability to overcome resistance mechanisms. Such mechanistic precision ensures that the final product meets the stringent purity specifications required for clinical applications.
Impurity control is inherently built into this synthetic design through the use of crystallization and chromatography steps that leverage the distinct physical properties of the intermediates. For instance, the intermediate compounds are often isolated as white solids through recrystallization from solvents like ethyl acetate and hexane, which effectively removes non-polar byproducts and unreacted starting materials. The final hydrolysis steps are monitored via TLC to ensure complete deprotection without degrading the sensitive oxetane ring or the ester linkages. By maintaining a pH range of 3 to 4 during hydrolysis, the process minimizes the formation of degradation products that could complicate downstream purification. This rigorous control over reaction conditions and workup procedures results in a high-purity paclitaxel analogue that is suitable for further formulation, addressing the critical concerns of R&D directors regarding impurity profiles and batch-to-batch consistency.
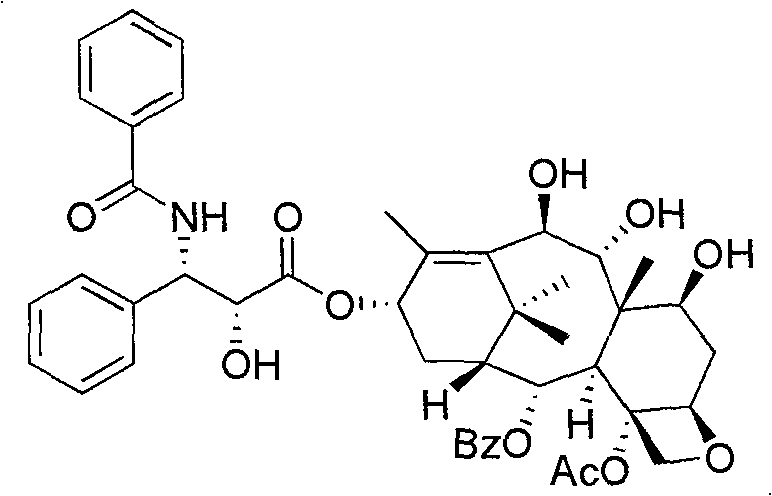
How to Synthesize 10-Deacetylate-9(R)-Hydrogenation-1-Deoxypaclitaxel Efficiently
The synthesis of this valuable analogue follows a logical progression of protection, coupling, and deprotection steps that are amenable to industrial scaling. The process begins with the preparation of the core nucleus from 1-deoxy baccatin VI, followed by the attachment of the N-benzoyl-3-phenylisoserine side chain which is essential for antitumor activity. Each step is optimized for yield and purity, utilizing common organic solvents and reagents that are readily available in a standard chemical manufacturing facility. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios, temperatures, and workup procedures required to achieve the reported high yields. This structured approach allows for reproducible results, making it an ideal candidate for technology transfer and commercial production.
- Deacetylation of 1-deoxy baccatin VI using hydrazine hydrate in ethanol to yield 7,9,10,13-tetra-deacetyl-1-deoxy baccatin VI.
- Protection of the 9,10-diol system using 2,2-dimethoxypropane and an acid catalyst to form the acetonide intermediate.
- Esterification with the paclitaxel side chain followed by acidic hydrolysis to remove protecting groups and yield the final analogue.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling advantages that extend beyond mere chemical elegance. The primary benefit lies in the utilization of 1-deoxy baccatin VI, a starting material that is more abundant and easier to source than the precursors required for natural paclitaxel production. This shift in raw material strategy significantly mitigates the risk of supply disruptions caused by seasonal variations or ecological restrictions on Taxus harvesting. Furthermore, the simplified operational steps reduce the overall processing time and resource consumption, leading to substantial cost savings in the long run. By integrating this technology, organizations can secure a more reliable pharmaceutical intermediate supplier relationship that is resilient to market fluctuations and capable of meeting growing demand for oncology therapeutics.
- Cost Reduction in Manufacturing: The elimination of complex extraction processes and the use of high-yield semi-synthetic steps directly contribute to a lower cost base for production. By avoiding the need for expensive transition metal catalysts or specialized enzymatic processes, the method relies on cost-effective chemical reagents that are easily sourced globally. The high selectivity of the reactions minimizes waste generation and reduces the burden on purification systems, which are often the most expensive part of pharmaceutical manufacturing. Consequently, this leads to a more economical production model that allows for competitive pricing without compromising on the quality of the final active ingredient.
- Enhanced Supply Chain Reliability: Sourcing 1-deoxy baccatin VI from established plantations ensures a consistent flow of raw materials, thereby reducing lead time for high-purity pharmaceutical intermediates. The robustness of the synthetic route means that production can be scaled up rapidly in response to market demand without the long lead times associated with biological extraction. This reliability is crucial for maintaining continuous manufacturing schedules and ensuring that downstream drug formulation processes are not interrupted. Supply chain heads can therefore plan with greater confidence, knowing that the precursor availability is decoupled from the volatile dynamics of natural product harvesting.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reaction vessels and workup techniques that are easily transferred from pilot plant to commercial scale. The use of common organic solvents allows for efficient recovery and recycling, aligning with modern environmental compliance standards and reducing the overall environmental footprint. Additionally, the high atom economy of the key coupling steps ensures that raw materials are utilized efficiently, minimizing the generation of hazardous waste. This makes the technology not only commercially viable but also sustainable, meeting the increasing regulatory and corporate social responsibility requirements of the global pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patent technology in a production environment. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of the method. Understanding these details is crucial for stakeholders evaluating the potential integration of this analogue into their product pipelines. The information below serves as a preliminary guide for technical discussions and feasibility assessments.
Q: What is the primary advantage of this paclitaxel analogue over natural paclitaxel?
A: The analogue is designed to reduce multidrug resistance (MDR) and toxic side effects while maintaining antitumor bioactivity, addressing key limitations of natural taxol.
Q: Is the starting material for this synthesis readily available?
A: Yes, the process utilizes 1-deoxy baccatin VI, which is found in higher content in Taxus cuspidata resources, ensuring a more sustainable and accessible supply chain compared to extracting natural paclitaxel.
Q: How does this method improve manufacturing efficiency?
A: The method features simple operation conditions, good selectivity, and high yields across multiple steps, significantly simplifying the purification process and reducing overall production complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 10-Deacetylate-9(R)-Hydrogenation-1-Deoxypaclitaxel Analogue Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this synthetic route for the global oncology market and are prepared to support its commercialization. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to market is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of this complex taxane analogue meets the highest international standards. We understand the critical nature of antitumor intermediates and are committed to delivering consistent quality that supports your clinical and commercial goals.
We invite you to collaborate with us to optimize your supply chain and leverage the benefits of this advanced technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and production timelines. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can enhance your manufacturing efficiency. Together, we can accelerate the availability of this promising analogue to patients worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
