Optimizing Cefazedone Sodium Production: A Novel Mixed Anhydride Route for Industrial Scale
Introduction to Advanced Cefazedone Sodium Synthesis
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antibiotics, and the preparation of Cefazedone Sodium stands as a prime example of process innovation. Based on the technological breakthroughs detailed in patent CN109651402B, we observe a significant departure from traditional, costly synthesis routes toward a more efficient mixed anhydride methodology. This novel approach addresses long-standing challenges in cephalosporin production, specifically targeting the optimization of the 7-position acylation and the 3-position substitution steps. By leveraging a unique combination of pivaloyl chloride and controlled pH modulation, this process achieves exceptional purity levels exceeding 99% while utilizing readily available raw materials. For global procurement teams and R&D directors, understanding this shift is vital for securing a reliable cefazedone sodium supplier capable of meeting stringent regulatory standards.
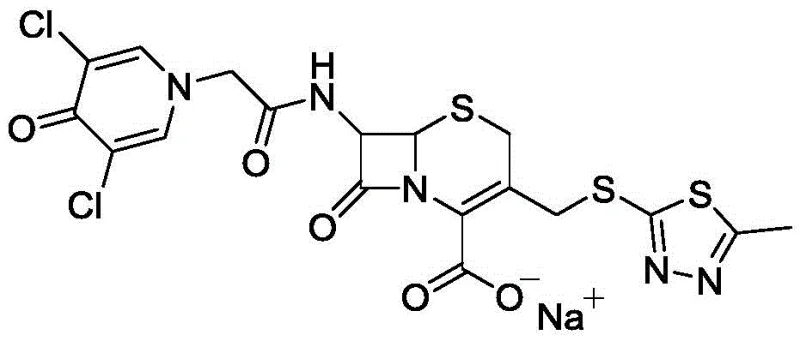
The structural complexity of Cefazedone Sodium requires precise stereochemical control, particularly at the 6R and 7R positions. The patented process ensures that the delicate beta-lactam ring remains intact throughout the synthesis, minimizing ring-opening degradation which is a common failure mode in less optimized routes. This level of control is not merely a technical achievement but a commercial necessity, as it directly correlates to the therapeutic efficacy and safety profile of the final antibiotic product. As we delve deeper into the mechanistic advantages, it becomes clear why this methodology represents the gold standard for modern pharmaceutical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cefazedone Sodium has been plagued by inefficient coupling strategies that hinder industrial scalability. Early methods, such as the direct synthesis using DCC (dicyclohexylcarbodiimide) as a condensing agent, suffered from severe drawbacks including high raw material costs and the necessity for strictly anhydrous reaction conditions. The formation of dicyclohexylurea as a byproduct created significant purification bottlenecks, often requiring complex filtration and washing steps that reduced overall throughput. Furthermore, alternative routes employing trichloroacetyl chloride to form acid chlorides introduced harsh reaction environments that compromised the stability of the cephalosporin nucleus, leading to lower yields and unpredictable impurity profiles. These legacy processes are increasingly untenable in a market demanding both cost efficiency and environmental sustainability.
The Novel Approach
In stark contrast, the novel approach outlined in the patent utilizes a mixed anhydride generated from 3,5-dichloropyridone acetic acid and pivaloyl chloride. This strategy fundamentally alters the reaction kinetics, providing a highly active acylating agent that reacts rapidly and selectively with the 7-amino group of the cephalosporin intermediate. The use of pivaloyl chloride is particularly advantageous due to its steric bulk, which helps prevent unwanted side reactions while maintaining high reactivity. Additionally, the process incorporates a sophisticated three-batch addition protocol for the 7-TDA intermediate, coupled with precise pH adjustments, to maintain optimal reaction conditions throughout the acylation phase. This results in a streamlined workflow that is far more amenable to large-scale production.
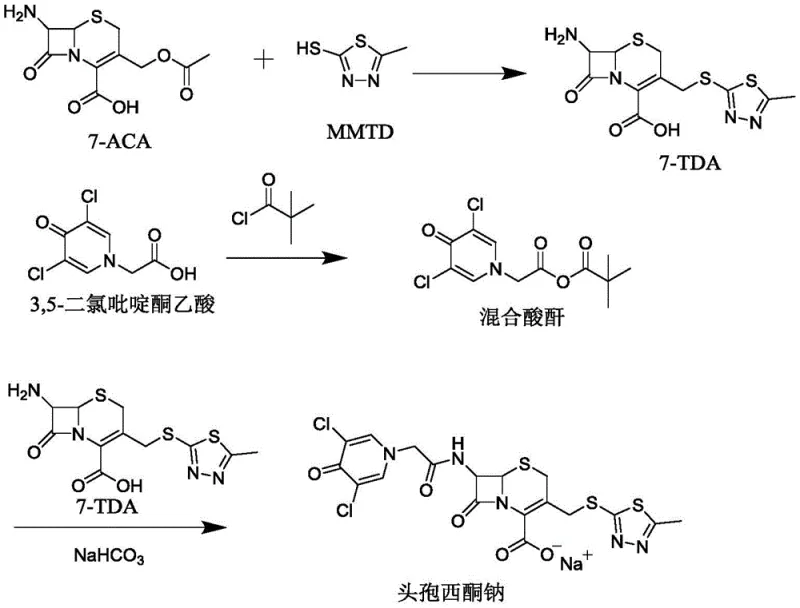
The visual representation of this new pathway highlights the elegance of the design, where the 3-position substitution is decoupled from the 7-position acylation to maximize yield at each stage. By avoiding the instability issues associated with active esters and the toxicity concerns of certain acid chlorides, this method offers a cleaner, safer, and more economically viable route. For supply chain managers, this translates to a more predictable production schedule and reduced risk of batch failures, ensuring a consistent supply of high-quality antibiotic intermediates to the global market.
Mechanistic Insights into Mixed Anhydride Acylation and Substitution
The core of this technological advancement lies in the dual optimization of the 3-position substitution and the 7-position acylation mechanisms. In the first stage, the conversion of 7-ACA to 7-TDA is facilitated by a boron trifluoride-dimethyl carbonate complex catalyst within a mixed solvent system. This specific solvent blend, comprising dimethyl carbonate and organic acids like citric or tartaric acid, plays a critical role in modulating the polarity of the reaction medium. By adjusting the proton concentration and enhancing the solubility of both 7-ACA and MMTD, the system promotes a homogeneous reaction environment that significantly accelerates the substitution rate while suppressing the formation of regio-isomers. This precise control over the reaction microenvironment is essential for achieving the reported purity levels of over 99%.
Following the formation of the 7-TDA intermediate, the acylation step relies on the in situ generation of a mixed anhydride. The mechanism involves the nucleophilic attack of the carboxylate of 3,5-dichloropyridone acetic acid on the carbonyl carbon of pivaloyl chloride. The resulting mixed anhydride possesses a lower activation energy compared to traditional acid chlorides, making it highly susceptible to nucleophilic attack by the amino group of 7-TDA. Crucially, the patent describes a triple-addition protocol where 7-TDA is added in three distinct batches, with pH adjustments made after each addition. This prevents the accumulation of the acid-binding agent and the carboxylic acid byproduct, which would otherwise catalyze the decomposition of the mixed anhydride back to its starting materials. This dynamic control ensures that the concentration of the active acylating species remains high throughout the reaction, driving the equilibrium towards the desired product and minimizing hydrolytic degradation.
How to Synthesize Cefazedone Sodium Efficiently
Implementing this synthesis route requires careful attention to temperature control and stoichiometry, particularly during the exothermic formation of the mixed anhydride. The process begins with the preparation of the 7-TDA intermediate in a dimethyl carbonate solvent, followed by the separate generation of the mixed anhydride at low temperatures ranging from -10°C to 0°C. The convergence of these two streams is managed through a controlled addition strategy that maintains the reaction pH within a narrow window to optimize yield. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below, which outline the precise molar ratios and thermal profiles necessary for successful replication.
- Synthesize 7-TDA by reacting 7-ACA with MMTD in a dimethyl carbonate and organic acid mixed solvent using a boron trifluoride catalyst.
- Prepare the mixed anhydride by reacting 3,5-dichloropyridone acetic acid with pivaloyl chloride under catalytic conditions at low temperature.
- Perform the final acylation by adding 7-TDA in three batches to the mixed anhydride solution with intermittent pH adjustment, followed by salification and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this mixed anhydride methodology offers profound advantages for cost reduction in antibiotic manufacturing. The replacement of expensive condensing agents like DCC with commodity chemicals such as pivaloyl chloride drastically lowers the raw material bill of materials. Furthermore, the elimination of difficult-to-remove byproducts simplifies the downstream purification process, reducing solvent consumption and waste disposal costs. This efficiency gain is compounded by the higher overall yield of the process, which means more product is generated per unit of input, directly improving the margin profile for manufacturers. For procurement managers, this translates into a more competitive pricing structure without compromising on the quality of the final API.
- Cost Reduction in Manufacturing: The substitution of high-cost reagents with economically favorable alternatives like pivaloyl chloride creates a substantial decrease in production expenses. By avoiding the need for specialized anhydrous conditions and expensive catalysts, the process reduces energy consumption and equipment wear. The simplified workup procedure further contributes to cost savings by shortening cycle times and minimizing the labor hours required for purification, making the overall manufacturing process significantly more lean and efficient.
- Enhanced Supply Chain Reliability: The reliance on widely available and stable raw materials ensures a robust supply chain that is less susceptible to market volatility. Unlike specialized active esters that suffer from poor storage stability and short shelf lives, the precursors used in this method are commodity chemicals with established global supply networks. This stability allows for better inventory planning and reduces the risk of production stoppages due to material shortages, thereby enhancing the reliability of delivery for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions, operating primarily between -10°C and 30°C, are easily manageable in standard stainless steel reactors, facilitating seamless commercial scale-up of complex antibiotics. The process generates fewer hazardous byproducts and utilizes solvents that are easier to recover and recycle, aligning with modern green chemistry principles. This environmental compatibility not only reduces regulatory burden but also future-proofs the manufacturing site against tightening environmental regulations, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis pathway. These insights are derived directly from the patent data and practical experience in cephalosporin manufacturing, aiming to clarify the operational benefits and technical nuances of the process. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this technology for their own production lines.
Q: Why is the mixed anhydride method superior to the DCC condensation method for Cefazedone Sodium?
A: The mixed anhydride method utilizes pivaloyl chloride, which is significantly cheaper and more stable than DCC. It avoids the strict anhydrous conditions required by DCC and eliminates the formation of difficult-to-remove urea byproducts, leading to higher purity and easier downstream processing.
Q: How does the three-batch addition of 7-TDA improve the reaction yield?
A: Adding 7-TDA in three batches with intermediate pH adjustments prevents the accumulation of the acid-binding agent and the carboxylic acid byproduct. This strategy maintains the reactivity of the mixed anhydride, prevents its hydrolysis over time, and ensures a more thorough reaction with reduced impurity generation.
Q: What are the critical quality parameters for the 7-TDA intermediate in this process?
A: High purity of the 7-TDA intermediate is crucial, typically exceeding 99% as achieved by the patented mixed solvent system. Impurities in the 3-position substitution step can carry through to the final antibiotic, affecting the overall potency and safety profile of the Cefazedone Sodium API.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefazedone Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis routes requires a partner with deep technical expertise and proven execution capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a practical industrial setting. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Cefazedone Sodium meets the highest international pharmacopoeia standards. Our commitment to quality assurance ensures that your supply chain remains uninterrupted and compliant with global regulatory requirements.
We invite you to collaborate with us to leverage these technological advancements for your product portfolio. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete performance metrics. Let us help you optimize your antibiotic supply chain with a solution that balances cost, quality, and reliability.
