Scaling Bio-Based Monomers: Advanced Visible Light Carboxylation for 2,5-Furandicarboxylic Acid Production
Scaling Bio-Based Monomers: Advanced Visible Light Carboxylation for 2,5-Furandicarboxylic Acid Production
The global shift towards sustainable polymer feedstocks has placed 2,5-furandicarboxylic acid (FDCA) at the forefront of green chemistry innovation, serving as a critical bio-based alternative to terephthalic acid in the production of polyethylene furanoate (PEF). A groundbreaking development in this sector is detailed in patent CN113801081A, which discloses a novel preparation method utilizing visible light photocatalysis and carbon dioxide fixation. This technology represents a paradigm shift from traditional thermal oxidation processes, offering a pathway to high-purity intermediates without the reliance on scarce noble metals. For R&D directors and procurement strategists, understanding the nuances of this metal-free protocol is essential for evaluating future supply chain resilience and cost structures in the fine chemical and polymer sectors.
The core innovation lies in the utilization of tetraamine ethylene super electron donors as photocatalysts, which drive the carboxylation of non-edible furfural derivatives under anaerobic conditions. By leveraging the reducing power of these organic super-electron donors, the process activates stable C-H or C-X bonds on the furan ring, allowing for direct insertion of CO2. This approach not only valorizes a greenhouse gas as a C1 building block but also operates under remarkably mild conditions, typically between 20°C and 80°C. The implications for industrial manufacturing are profound, suggesting a route that is both energetically efficient and environmentally benign, aligning perfectly with modern ESG mandates for chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,5-furandicarboxylic acid has been dominated by the oxidation of 5-Hydroxymethylfurfural (HMF), a pathway fraught with significant technical and economic challenges. The oxidation process often suffers from poor selectivity, generating a complex spectrum of by-products such as formic acid and levulinic acid, which complicates downstream purification and drastically reduces overall yield. Furthermore, alternative carboxylation strategies reported in prior art frequently depend on expensive noble metal catalysts, such as ruthenium or iridium complexes, which introduce substantial raw material costs and pose risks of heavy metal contamination in the final API or polymer grade material. These traditional methods often require harsh reaction conditions, including high temperatures and pressures, leading to elevated energy consumption and increased safety hazards in large-scale reactors.
The Novel Approach
In stark contrast, the methodology described in patent CN113801081A circumvents these bottlenecks by employing a visible-light-driven organocatalytic system. The process utilizes readily available furfural derivatives as substrates, which react with CO2 in the presence of a tetraamine ethylene super electron donor. This eliminates the need for transition metals entirely, thereby removing the cost burden associated with precious metal procurement and the regulatory burden of residual metal testing. The reaction proceeds smoothly in aprotic solvents like dimethylsulfoxide or N,N-dimethylformamide, utilizing simple inorganic bases as auxiliaries. This transition to a metal-free, photochemical platform not only simplifies the impurity profile but also democratizes access to FDCA synthesis for facilities lacking specialized high-pressure hydrogenation or oxidation infrastructure.
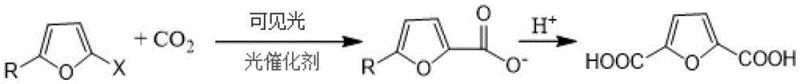
The visual representation of this transformation highlights the elegance of the direct carboxylation strategy. As depicted in the reaction scheme, the substrate, characterized by a furan ring with variable substituents (R and X), undergoes a clean conversion to the dicarboxylic acid structure. The versatility of the method is evident in its tolerance for various halogen substituents (F, Cl, Br, I) and ester groups, allowing for a broad scope of feedstock utilization. This flexibility is crucial for supply chain managers who may need to pivot between different biomass-derived starting materials based on regional availability and price fluctuations, ensuring continuous production capability without extensive process re-validation.
Mechanistic Insights into Tetraamine Ethylene Photocatalysis
From a mechanistic perspective, the efficacy of this process relies on the unique electronic properties of the tetraamine ethylene super electron donor. Upon irradiation with visible light, these organic molecules enter an excited state with a sufficiently low reduction potential to transfer a single electron to the furan substrate. This single electron transfer (SET) generates a radical anion intermediate on the furan ring, which is highly nucleophilic and capable of attacking the electrophilic carbon of the CO2 molecule. This radical-polar crossover mechanism is distinct from traditional two-electron oxidative pathways and allows for the functionalization of positions on the heterocycle that are typically inert under standard thermal conditions. The regeneration of the catalyst and the subsequent protonation steps are facilitated by the auxiliary base and the solvent environment, closing the catalytic cycle efficiently.
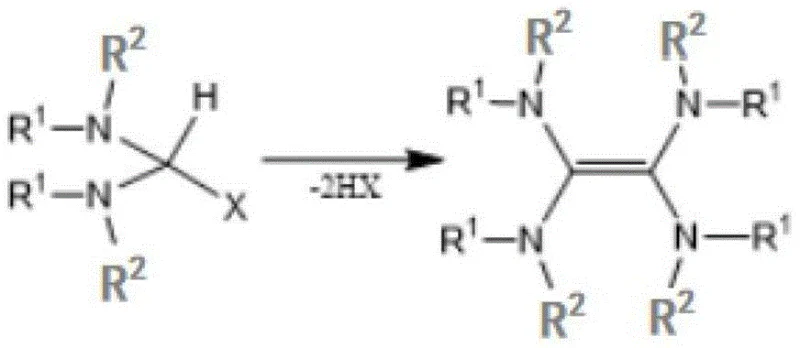
The preparation of the photocatalyst itself is a testament to the process's economic viability, as shown in the catalyst synthesis diagram. The tetraamine ethylene derivative is generated through the elimination of HX from readily accessible amine precursors or via condensation with orthoformates. This synthetic accessibility ensures that the catalyst can be produced in-house or sourced at a fraction of the cost of commercial photoredox complexes. For quality control teams, the organic nature of the catalyst means that any residual catalyst in the final product is easier to manage and remove compared to heavy metals, which often require specialized scavenging resins or recrystallization steps. The robustness of this catalytic system underpins the high yields observed in the patent examples, ranging from 65% to 95%, demonstrating excellent reproducibility across different substrate variations.
How to Synthesize 2,5-Furandicarboxylic Acid Efficiently
Implementing this synthesis route requires careful attention to the exclusion of oxygen, as the radical intermediates generated during the photocatalytic cycle are sensitive to aerobic quenching. The standard operating procedure involves dissolving the furfural derivative, the super electron donor, and an inorganic base in a dry, deoxygenated aprotic solvent under a nitrogen or inert gas blanket. Once the homogeneous mixture is established, the system is exposed to visible light sources, such as blue LEDs, while CO2 is introduced, either via balloon or continuous flow. Detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined in the technical guide below to ensure consistent replication of the high-purity results reported in the patent literature.
- Prepare the reaction mixture by dissolving the furfural derivative substrate, tetraamine ethylene super electron donor, and inorganic base auxiliary in an aprotic solvent under an oxygen-free nitrogen or inert gas environment.
- Expose the mixture to visible light irradiation while introducing CO2 gas, maintaining a temperature between 20°C and 80°C to facilitate the carboxylation reaction.
- Upon completion of the reaction, perform an acidification step to precipitate and isolate the final 2,5-furandicarboxylic acid product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible-light carboxylation technology offers a compelling value proposition centered on cost stability and operational simplicity. By decoupling production from the volatile markets of precious metals like ruthenium and iridium, manufacturers can achieve significant cost reduction in polymer additive manufacturing. The reliance on commodity chemicals such as CO2 and basic amines for catalyst generation creates a more predictable cost structure, shielding the supply chain from geopolitical disruptions that often affect the availability of specialized transition metal catalysts. Furthermore, the mild reaction temperatures reduce the energy load on production facilities, contributing to lower utility costs and a smaller carbon footprint per kilogram of product produced.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts serves as a primary driver for lowering the Bill of Materials (BOM). Traditional photoredox methods often require catalyst loadings that, while low, represent a high unit cost due to the intrinsic value of the metals. By switching to an organic super electron donor system, the direct material costs are drastically simplified. Additionally, the avoidance of heavy metals removes the need for costly downstream purification steps, such as metal scavenging columns or extensive recrystallization sequences required to meet strict ppm limits for pharmaceutical or food-contact applications. This streamlined workflow translates directly into improved gross margins and a more competitive pricing structure for the final 2,5-furandicarboxylic acid.
- Enhanced Supply Chain Reliability: The feedstock profile for this process enhances supply security by utilizing non-edible furfural derivatives, which are abundant biomass processing by-products. Unlike food-competing feedstocks, these materials offer a stable supply baseline that is less susceptible to agricultural yield fluctuations. Moreover, the use of CO2 as a reagent leverages an essentially infinite and low-cost carbon source, further insulating the production process from raw material scarcity. The simplicity of the reagents—common inorganic bases and standard aprotic solvents—means that sourcing can be localized in most major chemical manufacturing hubs, reducing lead times for high-purity intermediates and minimizing logistics risks associated with importing specialized reagents.
- Scalability and Environmental Compliance: The operational parameters of this method are inherently scalable, operating at near-ambient temperatures and pressures that do not require exotic high-pressure reactor vessels. This facilitates easier technology transfer from laboratory to pilot and commercial scales, reducing the capital expenditure (CAPEX) required for plant retrofitting. From an environmental compliance standpoint, the process is greener by design; it fixes CO2, avoids toxic heavy metals, and utilizes visible light, a renewable energy input. This alignment with green chemistry principles simplifies regulatory filings and environmental impact assessments, accelerating time-to-market for new bio-based polymer applications and ensuring long-term viability in increasingly regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic technology. These insights are derived directly from the experimental data and claims within patent CN113801081A, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the primary advantages of using tetraamine ethylene super electron donors over traditional metal catalysts?
A: The use of tetraamine ethylene super electron donors eliminates the need for expensive noble metal catalysts like ruthenium or iridium complexes. This organic photocatalyst offers a lower excited state potential and stronger reducibility, enabling milder reaction conditions and significantly reducing raw material costs while avoiding heavy metal contamination in the final product.
Q: Can this photocatalytic method be scaled for industrial production of polymer monomers?
A: Yes, the process is highly conducive to industrial scale-up. It operates at mild temperatures (20-80°C), utilizes readily available non-edible furfural derivatives and CO2, and avoids complex high-pressure or high-temperature conditions typical of traditional oxidation methods, thereby simplifying reactor design and safety protocols.
Q: How does this method address impurity profiles compared to HMF oxidation?
A: Unlike the oxidation of 5-Hydroxymethylfurfural (HMF), which often yields complex mixtures of oxidation products that are difficult to separate, this carboxylation approach targets specific C-H activation sites. The mild visible light conditions minimize thermal degradation and side reactions, resulting in a cleaner crude reaction mixture and simplified downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Furandicarboxylic Acid Supplier
As the demand for bio-based polymers accelerates, the ability to produce high-quality 2,5-furandicarboxylic acid efficiently becomes a critical competitive advantage. NINGBO INNO PHARMCHEM stands ready to support your transition to this advanced manufacturing paradigm. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, our engineering team is equipped to optimize this visible-light protocol for your specific capacity requirements. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch meets the exacting standards required for polymerization and pharmaceutical applications, guaranteeing consistency and performance in your final products.
We invite you to engage with our technical procurement team to explore how this metal-free synthesis can enhance your supply chain resilience. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching from traditional oxidation or metal-catalyzed routes to this sustainable photocatalytic method. Contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, and let us partner with you to drive the next generation of green chemical innovation.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
