Advanced Biocatalytic Production of High-Purity Chiral 1,2-Diols for Pharmaceutical Intermediates
Advanced Biocatalytic Production of High-Purity Chiral 1,2-Diols for Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient routes to access chiral building blocks, particularly 1,2-diol derivatives which serve as critical scaffolds for complex natural products and active pharmaceutical ingredients. A significant technological breakthrough in this domain is documented in Chinese Patent CN108753851B, which discloses a highly selective biocatalytic method for preparing chiral monohydroxyl-derivatized 1,2-alcohol compounds. This patent introduces the use of a specific carbonyl reductase, designated as ChKRED12, to catalyze the asymmetric reduction of alpha-alkoxy-beta-ketoesters. Unlike traditional chemical synthesis which often relies on hazardous reagents or precious metals, this biological approach operates under mild aqueous conditions, delivering products with exceptional optical purity. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a paradigm shift towards greener, more cost-effective manufacturing processes that align with modern regulatory and environmental standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral 1,2-diols has been plagued by significant technical and economic hurdles that limit their widespread application in commercial scale-up of complex polymer additives or drug candidates. Traditional chemical routes, such as glycolic acid aldol condensation, require the use of stoichiometric amounts of silicon tetrachloride and complex diphosphoramide catalysts to generate active enol silyl ethers, resulting in繁琐 reaction sequences and difficult waste disposal issues. Alternative methods involving epoxidation-reduction sequences often demand harsh reaction conditions and excessive catalyst loading, sometimes up to 30 mol%, which drastically inflates production costs and complicates downstream purification. Furthermore, while transition metal-catalyzed asymmetric reduction offers good stereoselectivity, the presence of heavy metal residues poses severe toxicity risks for pharmaceutical applications, necessitating expensive removal steps. Even biological approaches using wild-type yeast cells have historically suffered from low catalytic efficiency, with substrate concentrations limited to merely 2-4 g/L and reaction times extending over several days, rendering them unsuitable for high-throughput industrial manufacturing.
The Novel Approach
In stark contrast to these legacy technologies, the novel biocatalytic route utilizing ChKRED12 offers a streamlined and robust solution for cost reduction in chiral diol manufacturing. This method employs a genetically engineered carbonyl reductase that exhibits superior activity and stability compared to crude yeast extracts. The process eliminates the need for toxic heavy metals and harsh chemical reagents, operating instead in a simple phosphate buffer system at neutral pH and moderate temperatures ranging from 20 to 40°C. By coupling the reduction with a glucose dehydrogenase (GDH) cofactor regeneration system, the method ensures a continuous supply of the necessary reducing equivalents without the prohibitive cost of adding stoichiometric NADPH. This innovation not only simplifies the reaction workflow but also significantly enhances the overall atom economy, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates where purity and environmental compliance are paramount concerns for supply chain heads.
Mechanistic Insights into ChKRED12-Catalyzed Asymmetric Reduction
The core of this technological advancement lies in the exquisite stereoselectivity of the ChKRED12 enzyme, which belongs to the carbonyl reductase family. Mechanistically, the enzyme facilitates the hydride transfer from the reduced cofactor NADPH to the prochiral ketone carbonyl of the alpha-alkoxy-beta-ketoester substrate. The active site of ChKRED12 is structurally tuned to accommodate the substrate in a specific orientation that favors the formation of the (2S,3S)-configured anti-1,2-diol product. This high degree of stereocontrol is evidenced by the patent data, which reports enantiomeric excess (ee) values consistently greater than 99% and diastereomeric ratios (dr) predominantly exceeding 95/5 across a broad range of substrates. Such precision minimizes the formation of unwanted stereoisomers, thereby reducing the burden on downstream chromatographic separation and ensuring a cleaner impurity profile for the final API intermediate.
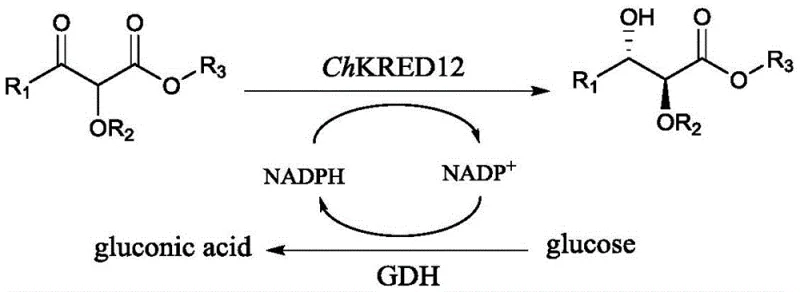
A critical component of this mechanism is the efficient cofactor regeneration loop depicted in the reaction scheme. Since NADPH is an expensive reagent, its stoichiometric use would be economically unfeasible for large-scale production. The patented process solves this by incorporating glucose dehydrogenase (GDH) and glucose into the reaction mixture. As ChKRED12 oxidizes NADPH to NADP+ during the reduction of the ketone, GDH simultaneously reduces NADP+ back to NADPH using glucose as the sacrificial electron donor, producing gluconic acid as a benign byproduct. This cyclic regeneration allows for the use of catalytic amounts of the cofactor, dramatically lowering the raw material costs. Furthermore, the aqueous nature of the reaction medium facilitates heat dissipation and mass transfer, contributing to the overall robustness and scalability of the process for producing high-purity OLED material precursors or pharmaceutical building blocks.
How to Synthesize Chiral 1,2-Diols Efficiently
The implementation of this biocatalytic process is straightforward and designed for ease of adoption in standard fermentation or chemical processing facilities. The general protocol involves suspending the recombinant E. coli cells expressing ChKRED12 in a phosphate buffer, followed by cell disruption to release the crude enzyme. The substrate, typically an alpha-alkoxy-beta-ketoester, is then introduced along with the cofactor system components. Detailed operational parameters regarding substrate loading, enzyme concentration, and reaction kinetics are critical for optimizing yield and throughput. For R&D teams looking to replicate or adapt this chemistry, the following standardized synthesis steps outline the fundamental procedure derived from the patent examples, ensuring reproducible results and high-quality output suitable for further synthetic elaboration.
- Prepare the biocatalytic system by mixing phosphate buffer (pH 7-8), substrate (alpha-alkoxy-beta-ketoester), carbonyl reductase ChKRED12, and cofactor NADP+.
- Add glucose dehydrogenase (GDH) and glucose to the reaction mixture to enable continuous regeneration of the reduced cofactor NADPH.
- Maintain the reaction at 20-40°C with stirring for 1-24 hours until conversion is complete, then extract the product using methyl tert-butyl ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the ChKRED12 biocatalytic platform offers tangible strategic benefits beyond mere technical performance. The shift from chemical catalysis to enzymatic synthesis fundamentally alters the cost structure and risk profile of the supply chain. By eliminating the reliance on precious transition metals like ruthenium or rhodium, manufacturers can avoid the volatility associated with commodity metal prices and the stringent regulatory requirements for heavy metal clearance in final drug substances. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to a lower carbon footprint and aligning with corporate sustainability goals. The simplicity of the work-up procedure, often requiring only extraction and crystallization, further accelerates the production cycle, enabling faster response times to market demands.
- Cost Reduction in Manufacturing: The economic viability of this process is driven primarily by the elimination of expensive chiral ligands and stoichiometric reducing agents. In traditional chemical asymmetric reduction, the cost of the chiral catalyst and the need for rigorous purification to remove metal traces can account for a substantial portion of the total manufacturing cost. The ChKRED12 system utilizes a biocatalyst that can be produced via fermentation at a fraction of the cost of synthetic catalysts. Moreover, the in-situ cofactor regeneration system means that the expensive NADPH is used catalytically rather than stoichiometrically, leading to substantial cost savings in raw materials. The high stereoselectivity also implies that less material is wasted on incorrect isomers, improving the overall yield and reducing the cost per kilogram of the final high-purity pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of multi-step chemical syntheses that rely on specialized reagents with long lead times. The biocatalytic route simplifies the supply chain by consolidating multiple transformation steps into a single enzymatic reaction. The starting materials, such as glucose and phosphate buffers, are commodity chemicals with stable and abundant global supplies, reducing the risk of shortages. Furthermore, the robustness of the ChKRED12 enzyme allows for flexible manufacturing schedules, as the reaction can be paused or adjusted more easily than sensitive chemical reactions. This flexibility ensures reducing lead time for high-purity pharmaceutical intermediates, allowing suppliers to meet tight delivery windows and maintain consistent inventory levels for their clients.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is generally more straightforward than scaling complex organometallic reactions, which often face heat transfer and mixing limitations at large volumes. The aqueous nature of the ChKRED12 reaction facilitates efficient heat management, allowing for safe scale-up from laboratory benchtop to multi-ton production scales. From an environmental perspective, this method generates significantly less hazardous waste compared to chemical alternatives that produce heavy metal sludge or require chlorinated solvents. The primary byproduct, gluconic acid, is non-toxic and biodegradable, simplifying wastewater treatment and ensuring compliance with increasingly strict environmental regulations. This green profile enhances the brand value of the final product and mitigates regulatory risks for the supply chain head.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the stereochemical outcomes of the ChKRED12 catalyzed reduction?
A: The ChKRED12 enzyme specifically produces the (2S,3S)-configured anti-1,2-diol derivatives with exceptional stereoselectivity, typically achieving ee values greater than 99% and dr values exceeding 95/5.
Q: How does this biocatalytic method compare to traditional yeast reduction?
A: Unlike traditional yeast methods which suffer from low substrate loading (2-4 g/L) and long reaction times, the ChKRED12 system allows for higher substrate concentrations (up to 20 g/L) and faster conversion rates under mild conditions.
Q: Is the cofactor system cost-effective for large scale production?
A: Yes, the process utilizes an efficient cofactor regeneration system involving glucose dehydrogenase and glucose, eliminating the need for stoichiometric amounts of expensive NADPH and making the process economically viable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 1,2-Diols Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biocatalysis in modern pharmaceutical synthesis and are committed to delivering these advanced solutions to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of chiral 1,2-diols meets the highest quality standards required for drug development. Our commitment to excellence extends beyond mere production; we act as a strategic partner, helping you navigate the complexities of supply chain management and regulatory compliance.
We invite you to explore how our ChKRED12-based manufacturing platform can optimize your project economics and accelerate your time to market. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our innovative biocatalytic solutions can drive value for your organization.
