Advanced Biocatalytic Synthesis of Chloramphenicol Intermediates for Commercial Scale-Up
The pharmaceutical industry is currently witnessing a paradigm shift towards sustainable manufacturing, particularly in the synthesis of complex chiral intermediates. Patent CN111808893A introduces a groundbreaking biological preparation method for amino alcohol drug intermediates, specifically targeting the key precursors for chloramphenicol. This technology leverages advanced enzyme engineering to replace traditional chemical reduction steps, offering a pathway that is not only chemically superior but also environmentally compliant. For R&D directors and procurement strategists, this patent represents a critical opportunity to optimize supply chains by adopting a route that inherently minimizes waste and maximizes stereochemical purity. The core innovation lies in the use of a specific (R,S)-carbonyl reductase which facilitates a dynamic kinetic resolution, effectively doubling the theoretical yield compared to conventional resolution methods while operating under mild aqueous conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of chloramphenicol intermediates has relied on a multi-step chemical synthesis starting from ethylbenzene. This legacy process involves nitration, oxidation, bromination, and ammonolysis, culminating in a stereoselective reduction using aluminum isopropoxide. The most significant bottleneck in this traditional workflow is the construction of the chiral center, which necessitates a resolution step that theoretically discards 50% of the material as the unwanted enantiomer. Furthermore, the reliance on aluminum-based reagents generates substantial volumes of aluminum-containing wastewater, posing severe environmental disposal challenges and increasing operational costs for waste treatment facilities. The cumulative effect of these inefficiencies results in a longer production cycle, lower overall atom economy, and a higher carbon footprint, making the legacy route increasingly untenable in a regulated global market.
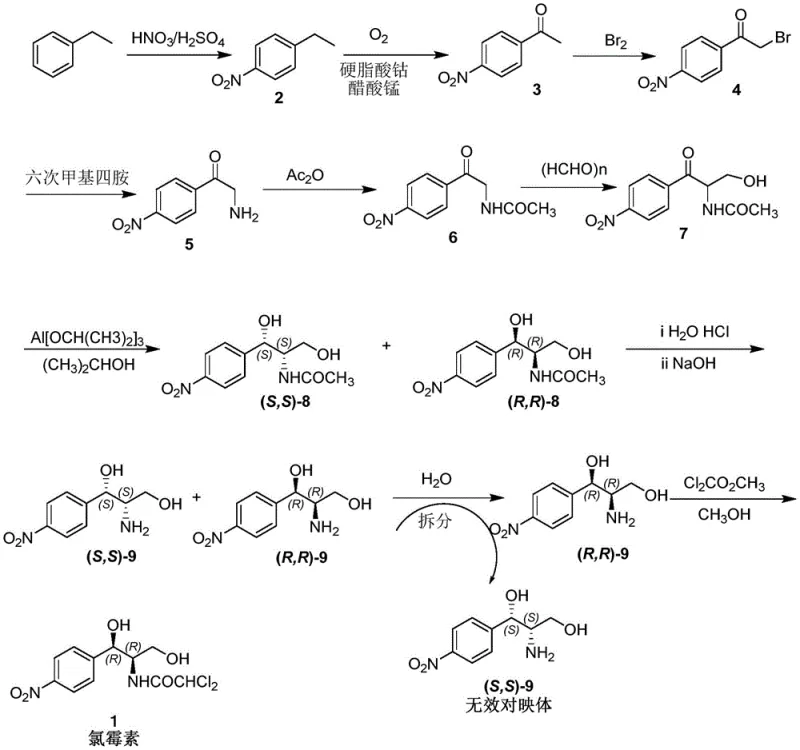
The Novel Approach
In stark contrast, the novel biocatalytic approach described in the patent streamlines the synthesis into a highly efficient enzymatic transformation. By utilizing a recombinant carbonyl reductase, the process achieves the construction of two chiral centers in a single reaction step through dynamic kinetic resolution. This eliminates the need for the wasteful resolution step entirely, allowing for near-quantitative conversion of the substrate into the desired chiral product. The reaction operates in an aqueous system with cofactor regeneration, significantly reducing the need for organic solvents and harsh chemical reagents. This transition from stoichiometric chemical reduction to catalytic biotransformation not only enhances the purity profile of the intermediate but also drastically simplifies the downstream processing requirements, offering a compelling value proposition for cost reduction in pharmaceutical intermediates manufacturing.
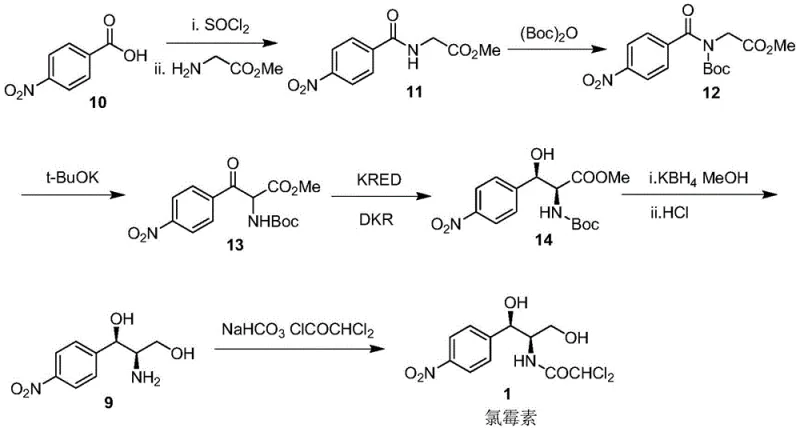
Mechanistic Insights into (R,S)-Carbonyl Reductase Catalyzed Reduction
The scientific foundation of this technology rests on the precise stereoselectivity of the (R,S)-carbonyl reductase enzyme. Unlike chemical catalysts that often require extreme temperatures or pressures to induce chirality, this biocatalyst operates through a sophisticated mechanism of dynamic kinetic resolution. The enzyme selectively reduces the prochiral ketone substrate while simultaneously allowing the non-preferred enantiomer to racemize in situ via enol tautomerism. This continuous interconversion ensures that the entire substrate pool is eventually funneled into the desired product configuration, breaking the 50% yield barrier inherent in static kinetic resolutions. The result is a product with an enantiomeric excess (ee) value exceeding 99% and a diastereomeric excess (de) value greater than 70%, meeting the stringent quality standards required for active pharmaceutical ingredient synthesis.
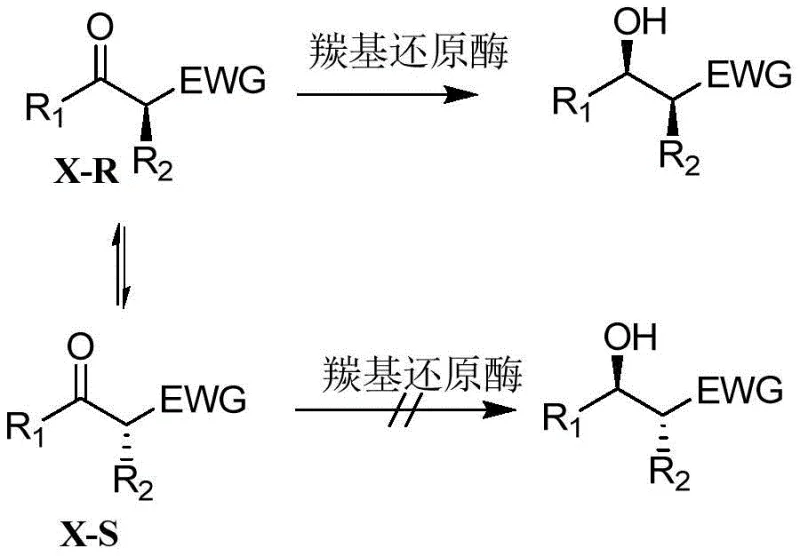
Controlling impurities in this biological system is achieved through the high specificity of the enzyme-substrate interaction. The biocatalyst is engineered to recognize specific structural motifs on the substrate, minimizing side reactions such as over-reduction or non-specific binding that are common in chemical catalysis. The reaction conditions, maintained at a mild pH of 7.0 to 9.0 and temperatures between 20°C to 40°C, further suppress the formation of thermal degradation byproducts. This inherent selectivity reduces the burden on purification teams, as the crude product profile is significantly cleaner than that obtained from chemical routes. For quality control departments, this translates to more consistent batch-to-batch reproducibility and a reduced risk of genotoxic impurities associated with heavy metal catalysts, ensuring a safer final drug product.
How to Synthesize Chloramphenicol Intermediate Efficiently
The implementation of this biocatalytic route requires a systematic approach to enzyme expression and reaction engineering. The process begins with the fermentation of recombinant E. coli strains harboring the carbonyl reductase gene, followed by cell disruption to obtain the crude enzyme or the use of resting cells directly. The substrate is dissolved in a cosolvent system to ensure adequate solubility in the aqueous buffer, and the reaction is initiated by adding the biocatalyst and cofactor regeneration system. Detailed standardized synthesis steps see the guide below.
- Prepare an aqueous reaction system containing compound X substrate, coenzyme NADP+, and glucose dehydrogenase for regeneration.
- Introduce (R,S)-carbonyl reductase enzyme to catalyze the stereoselective reduction under controlled pH and temperature conditions.
- Isolate the resulting compound Y through extraction and concentration, achieving high enantiomeric excess without chemical resolution.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biocatalytic technology offers transformative economic and logistical benefits. The elimination of aluminum-based reagents removes a significant cost center associated with hazardous waste disposal and regulatory compliance. Furthermore, the drastic improvement in yield efficiency means that less raw material is required to produce the same amount of final product, directly lowering the cost of goods sold. The simplified process flow, with fewer unit operations and milder reaction conditions, also reduces energy consumption and equipment wear, contributing to long-term operational sustainability and reliability in the supply of high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The transition to an enzymatic process fundamentally alters the cost structure by eliminating expensive stoichiometric reagents and the associated waste treatment costs. By avoiding the 50% material loss typical of chemical resolution, the effective cost per kilogram of the active intermediate is significantly reduced. Additionally, the use of renewable biocatalysts instead of precious metal catalysts or harsh chemicals mitigates the risk of price volatility in raw material markets, providing a more stable and predictable cost base for long-term procurement planning and budget management.
- Enhanced Supply Chain Reliability: Biocatalytic processes are inherently more scalable and robust compared to complex chemical syntheses involving hazardous reagents. The ability to produce the enzyme via fermentation ensures a consistent and renewable supply of the catalyst, reducing dependency on external chemical suppliers who may face production disruptions. This internal control over the critical catalytic component enhances supply chain resilience, ensuring continuous production capabilities even during periods of market instability or raw material shortages, thereby securing the continuity of supply for downstream API manufacturers.
- Scalability and Environmental Compliance: The aqueous nature of the reaction system and the absence of heavy metals make this process exceptionally easy to scale from pilot to commercial production without encountering the safety and environmental hurdles of traditional chemistry. Facilities can expand capacity with minimal investment in specialized containment or waste treatment infrastructure. This alignment with green chemistry principles not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the corporate sustainability profile, which is increasingly a key criterion for partnerships with major global pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic platform. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of adopting this technology for your specific production needs. Understanding these details is crucial for making informed decisions about process integration and supplier qualification.
Q: How does the enzymatic route improve yield compared to chemical synthesis?
A: The traditional chemical route relies on resolution which theoretically limits yield to 50%. The patented biocatalytic method utilizes dynamic kinetic resolution to convert nearly all substrate into the desired chiral isomer, significantly boosting overall productivity.
Q: What are the environmental benefits of this biological preparation method?
A: This process eliminates the use of aluminum isopropoxide and aluminum trichloride, thereby removing the generation of hazardous aluminum-containing wastewater and reducing the environmental footprint of the manufacturing facility.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method uses robust recombinant enzymes and standard fermentation techniques, making it highly adaptable for commercial scale-up from kilogram to multi-ton annual production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chloramphenicol Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of integrating advanced biocatalytic routes into the global pharmaceutical supply chain. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial realities. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which guarantee that every batch of chloramphenicol intermediate meets the highest international standards for safety and efficacy, providing you with a secure foundation for your drug development programs.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing us to demonstrate how our biocatalytic expertise can drive efficiency and value in your supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
