Advanced Naphthalene-Based D-π-A Fluorescent Materials for High-Performance OLED Manufacturing
The rapid evolution of the organic optoelectronics industry demands materials that balance high performance with manufacturability, a challenge addressed comprehensively in patent CN108148055B. This intellectual property introduces a novel class of naphthalene-based D-π-A type organic fluorescent materials designed specifically for next-generation organic light-emitting diodes (OLEDs). By utilizing a naphthalene ring as a rigid π-conjugated platform and strategically introducing electron donor (D) and electron acceptor (A) units at the 1 and 8 positions, the invention achieves a unique molecular architecture. This specific structural arrangement fosters large steric hindrance and a twisted conformation, which are critical for minimizing intermolecular aggregation and maximizing luminescence efficiency. The technical breakthrough lies not only in the photophysical properties but also in the synthetic accessibility, offering a viable pathway for reliable OLED material supplier partnerships seeking to diversify their emitter portfolios beyond traditional phosphorescent complexes.
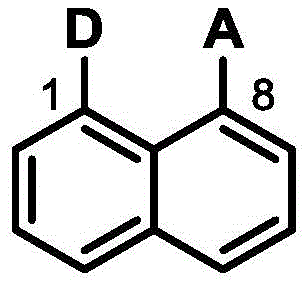
The development of high-efficiency emitting layers has historically been bottlenecked by the reliance on expensive heavy metal complexes and complex synthesis routes that suffer from low yields and difficult purification. Conventional methods often struggle with thermal instability during the vacuum deposition process, leading to device degradation and shortened operational lifetimes. Furthermore, the planar structures of many traditional fluorophores promote strong π-π stacking interactions, resulting in aggregation-caused quenching (ACQ) which drastically reduces quantum efficiency in solid-state films. These limitations necessitate a shift towards materials that inherently possess twisted geometries to suppress non-radiative decay pathways. The novel approach detailed in the patent overcomes these hurdles by leveraging the inherent rigidity of the naphthalene core combined with bulky donor and acceptor substituents, ensuring that the material maintains its integrity and emissive properties under the rigorous conditions of device fabrication and operation.
Mechanistic Insights into Suzuki Coupling and Molecular Engineering
The core synthetic strategy relies on palladium-catalyzed Suzuki cross-coupling reactions, a robust method for constructing carbon-carbon bonds between aryl halides and organoboron compounds. In this specific application, the mechanism involves the oxidative addition of the palladium catalyst to the carbon-bromine bond of the 1,8-dibromonaphthalene core, followed by transmetallation with the boronate ester-functionalized donor unit. The presence of a base, typically potassium carbonate, is crucial as it activates the boron species to form a reactive tetravalent borate intermediate, facilitating the transfer of the organic group to the palladium center. This mechanistic pathway allows for the precise installation of diverse electron-donating groups such as phenothiazine, carbazole, or acridine derivatives without compromising the sensitive functional groups present on the aromatic rings. The subsequent reductive elimination step regenerates the active catalyst and releases the coupled product, establishing the foundational D-π framework necessary for charge transport.
Beyond the coupling mechanics, the molecular engineering focuses on the spatial separation of frontier molecular orbitals to optimize the energy gap. The 1,8-substitution pattern on the naphthalene ring forces the donor and acceptor moieties into a non-planar, three-dimensional arrangement. This twisted intramolecular charge transfer (TICT) state is instrumental in reducing the overlap between the Highest Occupied Molecular Orbital (HOMO) and the Lowest Unoccupied Molecular Orbital (LUMO). By minimizing this overlap, the material can achieve a smaller singlet-triplet energy gap (ΔEST), which is beneficial for harvesting triplet excitons through thermally activated delayed fluorescence (TADF) mechanisms, although the primary focus here is on high-efficiency fluorescence. Additionally, the bulky nature of substituents like thianthrene dioxide acts as a physical barrier, preventing the close packing of molecules that typically leads to self-quenching, thereby preserving high photoluminescence quantum yields even in concentrated solid films.
How to Synthesize Naphthalene-Based D-π-A Fluorescent Materials Efficiently
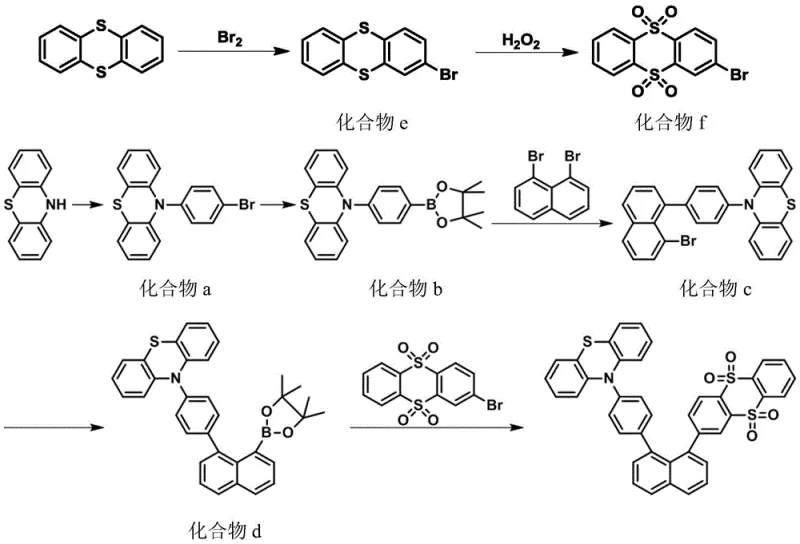
The synthesis protocol outlined in the patent provides a scalable and reproducible method for producing these advanced emitters, starting from commercially available precursors like 1,8-dibromonaphthalene and various brominated donor units. The process is divided into distinct stages involving boronic esterification, initial coupling to form the donor-naphthalene intermediate, and final coupling with the acceptor unit. Reaction conditions are carefully optimized, typically employing inert gas atmospheres to prevent catalyst oxidation and utilizing specific solvent systems like tetrahydrofuran or toluene mixtures to ensure solubility of the increasingly large conjugated intermediates. Temperature control is vital, with coupling reactions generally proceeding at reflux temperatures between 75°C and 90°C to drive the reaction to completion while minimizing side reactions. The detailed procedural steps below illustrate the standardized workflow required to achieve the high purity levels necessary for electronic grade applications.
- Preparation of Electron Donor Precursors: Perform Ullmann reaction or direct substitution to attach donor units like phenothiazine or carbazole to bromophenyl groups, followed by boronic esterification.
- Suzuki Coupling with Naphthalene Core: React the boronated donor intermediate with 1,8-dibromonaphthalene using a palladium catalyst and potassium carbonate base at 75-90°C.
- Final Assembly with Acceptor Unit: Couple the naphthalene-donor intermediate with electron acceptor units such as thianthrene dioxide derivatives to complete the D-π-A structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the transition to this naphthalene-based technology offers significant strategic advantages regarding cost structure and supply chain resilience. Traditional phosphorescent OLED materials often rely on scarce and price-volatile precious metals like iridium and platinum, which introduce substantial financial risk and supply bottlenecks. In contrast, the materials described in this patent are purely organic, eliminating the dependency on these critical raw materials and thereby enabling cost reduction in electronic chemical manufacturing. The synthesis utilizes widely available commodity chemicals such as naphthalene derivatives and common heterocycles, which are produced at scale by the global petrochemical industry. This abundance ensures a stable supply base that is less susceptible to geopolitical disruptions or mining constraints, providing procurement managers with greater predictability in long-term planning and budgeting for material costs.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts in the final product structure directly lowers the Bill of Materials (BOM) cost, while the use of standard Suzuki coupling chemistry leverages existing infrastructure in fine chemical plants. The synthetic route avoids exotic reagents or extreme conditions, allowing for production in standard glass-lined or stainless steel reactors without the need for specialized high-pressure equipment. Furthermore, the high thermal stability of the products simplifies the purification process, as sublimation can be performed efficiently without thermal degradation, reducing yield losses associated with purification steps. These factors collectively contribute to a more economical production model that can be passed down as value to downstream display manufacturers.
- Enhanced Supply Chain Reliability: The reliance on robust, well-understood chemical transformations like Ullmann and Suzuki couplings means that the technology can be easily transferred between different contract development and manufacturing organizations (CDMOs). This flexibility prevents single-source dependency, a critical factor for reducing lead time for high-purity organic fluorescent materials. The starting materials, such as 1,8-dibromonaphthalene and various bromo-heterocycles, are commodity items with multiple global suppliers, ensuring that raw material shortages are unlikely to halt production. This diversified supply base enhances the overall security of supply for OLED panel makers who require consistent, uninterrupted delivery of emitting materials to meet aggressive production schedules.
- Scalability and Environmental Compliance: The synthetic processes described operate at moderate temperatures and pressures, facilitating straightforward commercial scale-up of complex organic intermediates from gram-scale laboratory synthesis to multi-ton annual production. The use of common organic solvents like toluene and THF allows for established recovery and recycling protocols, minimizing waste generation and aligning with increasingly stringent environmental regulations. Additionally, the metal-free nature of the final product simplifies waste stream management, as there is no need for complex heavy metal removal or disposal procedures required for phosphorescent emitters. This environmental compatibility reduces the regulatory burden on manufacturing sites and supports sustainability goals within the electronics supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these naphthalene-based fluorescent materials in OLED device architectures. Understanding these details is essential for R&D teams evaluating the compatibility of these emitters with existing host materials and device stacks. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the thermal stability characteristics of these naphthalene-based materials?
A: According to patent CN108148055B, the synthesized compounds exhibit excellent thermal stability with decomposition temperatures (Td) reaching up to 399°C at 5% weight loss, making them highly suitable for vacuum deposition processes in OLED fabrication.
Q: How does the 1,8-substitution pattern on naphthalene improve performance?
A: The 1,8-position substitution creates significant steric hindrance, resulting in a twisted molecular structure. This geometry effectively separates the HOMO and LUMO energy levels, reduces concentration quenching, and enhances exciton utilization for higher quantum efficiency.
Q: Are precious metals required in the final fluorescent material structure?
A: No, the final organic fluorescent material structure is metal-free. While palladium catalysts are utilized during the Suzuki coupling synthesis steps, the resulting D-π-A molecules consist entirely of organic elements, eliminating the cost and supply chain risks associated with iridium or platinum-based phosphorescent emitters.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthalene-Based Fluorescent Material Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercially viable electronic chemicals, leveraging deep expertise in organic synthesis to deliver high-performance OLED materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to mass manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the exacting standards required for display applications. Our commitment to quality assurance ensures that the naphthalene-based D-π-A materials we supply deliver consistent color coordinates, efficiency, and lifetime performance in final devices.
We invite industry partners to collaborate with us to explore the full potential of these metal-free fluorescent emitters in your next-generation display projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals. Let us help you accelerate your product development cycle with reliable, high-quality materials that drive innovation in the organic optoelectronics sector.
