Scaling Ezetimibe Intermediate Production via Advanced Whole-Cell Biocatalysis Technology
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable pathways for producing high-value active pharmaceutical ingredients (APIs) and their critical precursors. A significant technological advancement in this domain is detailed in patent CN112458143B, which discloses a novel method for the whole-cell catalytic synthesis of chiral ezetimibe intermediates. This innovation addresses the longstanding challenges associated with the stereochemical control and environmental impact of traditional synthetic routes. By leveraging the biocatalytic potential of Schizosaccharomyces octosporus ATCC2479, this method achieves exceptional enantiomeric excess and conversion efficiency through a sophisticated staged fermentation process. The strategic implementation of freeze-dried whole cells not only enhances catalytic stability but also streamlines the operational workflow, offering a compelling alternative to legacy chemical methodologies. For global supply chains, this represents a pivotal shift towards greener manufacturing paradigms that do not compromise on yield or purity standards.
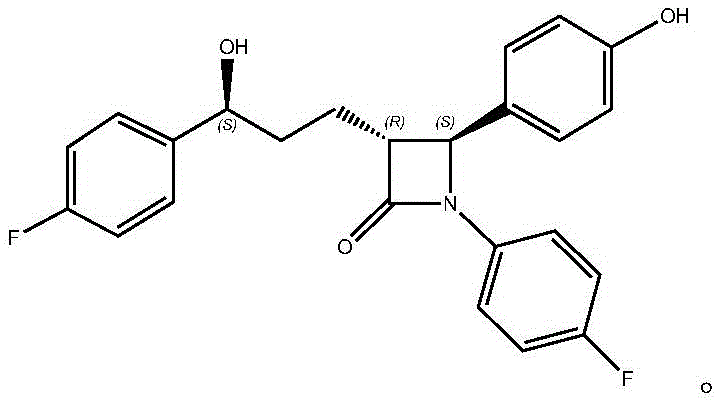
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ezetimibe and its key intermediates, such as the compound known as SCH 58235, has relied heavily on complex multi-step chemical sequences that pose significant operational and environmental hurdles. The conventional route, originally reported by Schering-Plough, involves the condensation of methyl chloroformylbutyrate with specific imines to construct the beta-lactam ring, followed by a Negishi coupling reaction utilizing p-fluorophenylzinc bromide. This organometallic step requires stringent anhydrous conditions and generates substantial amounts of zinc-containing waste, necessitating expensive and energy-intensive purification protocols to meet residual metal specifications. Furthermore, the subsequent reduction of the ketone carbonyl group typically employs borane-dimethyl sulfide complexes, which are hazardous, pyrophoric reagents that demand specialized handling infrastructure and rigorous safety controls. These chemical methods often struggle to maintain high enantiomeric purity without additional resolution steps, leading to lower overall yields and increased production costs due to material loss and waste disposal fees.
The Novel Approach
In stark contrast, the biocatalytic strategy outlined in the patent data introduces a paradigm shift by replacing hazardous chemical reductants with a highly selective enzymatic system housed within whole cells. This approach utilizes Schizosaccharomyces octosporus ATCC2479 as a biocatalyst to perform an asymmetric reduction of the ketone precursor (Compound II) directly to the desired chiral alcohol (Compound I). The use of freeze-dried cells provides a stable, shelf-stable catalyst form that simplifies transportation and dosing compared to liquid fermentation broths. Crucially, the process incorporates a unique staged stirring cultivation technique where acetone, a byproduct or co-solvent component, is actively removed via vacuum distillation midway through the reaction. This dynamic removal drives the reaction equilibrium forward, preventing product inhibition and significantly enhancing the substrate conversion rate. By operating at mild temperatures around 33°C and neutral pH, this method drastically reduces energy consumption and eliminates the generation of toxic heavy metal waste, aligning perfectly with modern green chemistry principles.
Mechanistic Insights into Whole-Cell Asymmetric Reduction
The core of this technological breakthrough lies in the intricate interplay between the whole-cell catalyst and the cofactor regeneration system. The Schizosaccharomyces octosporus cells act as micro-reactors containing the necessary oxidoreductases that facilitate the stereoselective transfer of hydride ions to the prochiral ketone substrate. To sustain this catalytic cycle, the system employs an auxiliary enzyme, isopropanol dehydrogenase, coupled with NADP+ as the electron carrier. Isopropanol serves a dual role in this mechanism: it acts as a co-solvent to improve the solubility of the organic substrate in the aqueous buffer and simultaneously functions as the hydrogen donor for cofactor regeneration. As the primary reductase converts the ketone to the alcohol, NADPH is oxidized to NADP+, which is then immediately reduced back to NADPH by the dehydrogenase using isopropanol, producing acetone as a byproduct. This efficient cofactor cycling ensures that only catalytic amounts of the expensive nicotinamide cofactor are required, making the process economically viable for large-scale applications.
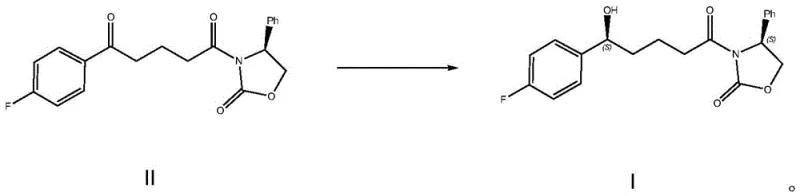
A critical mechanistic feature of this process is the management of reaction byproducts to optimize yield. In standard biocatalytic reductions, the accumulation of acetone can inhibit enzyme activity or shift the thermodynamic equilibrium back towards the starting material. The patent describes a sophisticated solution where the reaction is paused after an initial period, typically around 6 hours, to perform a vacuum distillation that selectively removes the acetone generated during the first phase. Following this removal, the volume is replenished with fresh isopropanol, and the reaction resumes for a second phase. This intervention effectively resets the thermodynamic landscape, allowing the enzymes to drive the conversion to near-completion levels, achieving substrate conversion rates as high as 97% with exceptional enantiomeric excess values exceeding 99%. This level of control over the reaction environment demonstrates a deep understanding of bioprocess engineering, ensuring that the biological catalyst performs at its theoretical maximum efficiency.
How to Synthesize Ezetimibe Chiral Intermediate Efficiently
The synthesis protocol described in the patent offers a reproducible framework for manufacturing high-purity chiral intermediates suitable for downstream pharmaceutical processing. The procedure begins with the preparation of the biocatalyst, where Schizosaccharomyces octosporus ATCC2479 is cultured in a defined YPD medium, harvested via centrifugation, and lyophilized to create a stable powder. This freeze-dried biomass is then suspended in a phosphate buffer system along with the ketone substrate, the cofactor recycling enzymes, and the necessary co-solvents. The reaction is conducted under controlled agitation and temperature to ensure optimal mass transfer and enzyme activity. While the general parameters are well-defined, precise optimization of stirring rates and distillation timing is essential for maximizing throughput in a commercial setting. The detailed standardized synthesis steps for implementing this protocol are provided in the guide below.
- Preparation of freeze-dried Schizosaccharomyces octosporus ATCC2479 cells using YPD liquid medium followed by centrifugation and lyophilization.
- Reaction setup involving Compound II, freeze-dried cells, isopropanol dehydrogenase, NADP+, and phosphate buffer at pH 7.0.
- Execution of a staged stirring culture at 33°C, incorporating a vacuum distillation step to remove acetone before completing the reaction cycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from traditional chemical synthesis to this whole-cell biocatalytic route offers profound strategic advantages that extend beyond simple unit cost calculations. The elimination of hazardous reagents such as organozinc compounds and borane complexes fundamentally alters the risk profile of the manufacturing process, reducing the need for specialized containment facilities and expensive safety monitoring systems. This simplification of the safety infrastructure translates directly into lower capital expenditure and operational overheads. Furthermore, the reliance on renewable biological catalysts and benign solvents like isopropanol positions the supply chain to be more resilient against regulatory tightening regarding environmental emissions and waste disposal. Companies adopting this technology can anticipate smoother regulatory audits and a reduced carbon footprint, which are increasingly critical metrics for maintaining partnerships with top-tier pharmaceutical innovators.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the purification train. Traditional chemical routes require extensive workup procedures to remove heavy metal residues like zinc and boron to parts-per-million levels, often involving multiple extraction, filtration, and chromatography steps that result in significant yield loss. By utilizing a metal-free biocatalytic system, these costly purification stages are rendered unnecessary, leading to substantial savings in solvent usage, filtration media, and labor hours. Additionally, the high stereoselectivity of the enzyme minimizes the formation of unwanted diastereomers, thereby reducing the material loss associated with chiral resolution or recrystallization steps. The ability to use catalytic amounts of cofactors through efficient recycling further lowers the raw material cost per kilogram of product, creating a leaner and more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: Supply chain continuity is significantly bolstered by the use of freeze-dried whole cells, which offer superior stability and shelf-life compared to liquid enzyme preparations or fresh cultures. This stability allows for bulk production and stocking of the biocatalyst, decoupling the catalyst supply from immediate production schedules and mitigating the risk of batch-to-batch variability. The robustness of the Schizosaccharomyces octosporus strain under the specified reaction conditions ensures consistent performance even when scaling up to larger reactor volumes. Moreover, the raw materials required for this process, such as isopropanol and phosphate buffers, are commodity chemicals with stable global availability, reducing the vulnerability of the supply chain to geopolitical disruptions or shortages of specialized reagents that often plague complex organic syntheses.
- Scalability and Environmental Compliance: From an environmental compliance perspective, this biocatalytic route represents a best-in-class solution for sustainable manufacturing. The aqueous nature of the reaction medium and the absence of chlorinated solvents or heavy metals simplify wastewater treatment processes, allowing for easier compliance with increasingly stringent environmental regulations. The staged distillation step, while adding a unit operation, effectively manages volatile organic compound (VOC) emissions by capturing acetone for potential reuse or safe disposal. The process is inherently scalable, as the principles of mass transfer and enzyme kinetics remain consistent from laboratory flasks to industrial fermenters, facilitating a seamless technology transfer. This scalability ensures that manufacturers can rapidly respond to market demand surges without the need for extensive process re-engineering or the construction of new specialized hazardous handling facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is essential for R&D teams planning pilot studies and procurement officers assessing vendor capabilities.
Q: What are the advantages of using whole-cell catalysis over traditional chemical synthesis for Ezetimibe intermediates?
A: Whole-cell catalysis offers superior stereospecificity with ee values exceeding 99%, eliminates the need for hazardous heavy metal reagents like zinc and borane used in traditional Negishi reactions, and operates under mild, environmentally friendly conditions.
Q: How does the staged stirring culture method improve product purity?
A: The patented process utilizes a specific two-stage cultivation where acetone is removed via vacuum distillation after the initial phase. This removal prevents side reactions and equilibrium limitations, significantly boosting substrate conversion rates from roughly 78% to over 97%.
Q: Is this biocatalytic process scalable for commercial manufacturing?
A: Yes, the use of freeze-dried whole cells simplifies logistics and storage compared to fresh cultures, and the reaction utilizes standard industrial equipment for stirring and distillation, making it highly amenable to scale-up from pilot to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ezetimibe Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biocatalysis in the production of complex pharmaceutical intermediates like those required for Ezetimibe. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial realities. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which are equipped to analyze chiral purity and trace impurities with the highest degree of accuracy. Our facility is designed to handle both traditional chemical synthesis and advanced biocatalytic processes, offering our clients the flexibility to choose the most efficient route for their specific project requirements.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your product quality while optimizing your overall production costs.
