Advanced Chemical Synthesis of Levo-5-Hydroxytryptophan for Scalable Pharmaceutical Manufacturing
Advanced Chemical Synthesis of Levo-5-Hydroxytryptophan for Scalable Pharmaceutical Manufacturing
The pharmaceutical and nutraceutical industries have long sought a robust, scalable, and cost-effective method for producing Levo-5-Hydroxytryptophan (5-HTP), a critical precursor for serotonin synthesis. Traditional reliance on plant extraction has created significant bottlenecks in the global supply chain, prompting a shift towards advanced chemical synthesis. Patent CN102351775A introduces a groundbreaking preparation method that utilizes L-tryptophan as a starting material, bypassing the limitations of agricultural dependency. This innovative route employs a sophisticated sequence of esterification, acetylation, and a unique indole ring reduction-oxidation-rearrangement strategy. By leveraging this technology, manufacturers can achieve product purity levels reaching 99.2% with a total yield exceeding 45%, establishing a new benchmark for quality in the production of neurological drug intermediates.
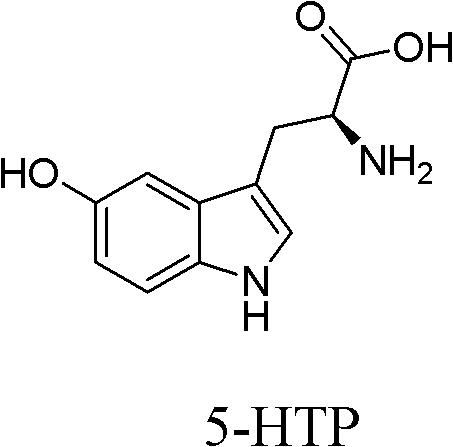
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 5-HTP has been dominated by extraction from the seeds of Griffonia Simplicifolia, a leguminous plant native to West Africa. While this method yields high-purity products, it is inherently flawed due to its complete dependence on agricultural output. The availability of raw materials fluctuates wildly with seasonal changes, climate conditions, and geopolitical factors in the region of origin, leading to unstable pricing and unpredictable delivery schedules. Furthermore, alternative biological methods, such as fermentation using engineered strains like BL21-DE3, suffer from low expression levels and complex metabolic byproducts. These biological impurities necessitate rigorous and costly downstream purification processes, often resulting in significant waste of the precious tryptophan substrate and failing to meet the stringent demands of large-scale industrial manufacturing.
The Novel Approach
In stark contrast, the novel chemical synthesis pathway described in the patent data offers a deterministic and controllable alternative that decouples production from biological variables. By starting with L-tryptophan, a widely available and cost-effective bulk chemical, the process ensures a stable and continuous supply of feedstock. The synthetic route is designed with operational simplicity in mind, utilizing standard organic transformations that are easily adaptable to reactor systems of varying sizes. This approach not only mitigates the risks associated with raw material scarcity but also significantly reduces environmental pollution by avoiding the massive solvent volumes typically required for plant extraction. The result is a streamlined manufacturing process that delivers consistent quality and quantity, making it an ideal solution for reliable pharmaceutical intermediate supplier networks seeking to optimize their procurement strategies.
Mechanistic Insights into Indole Ring Reduction and Rearrangement
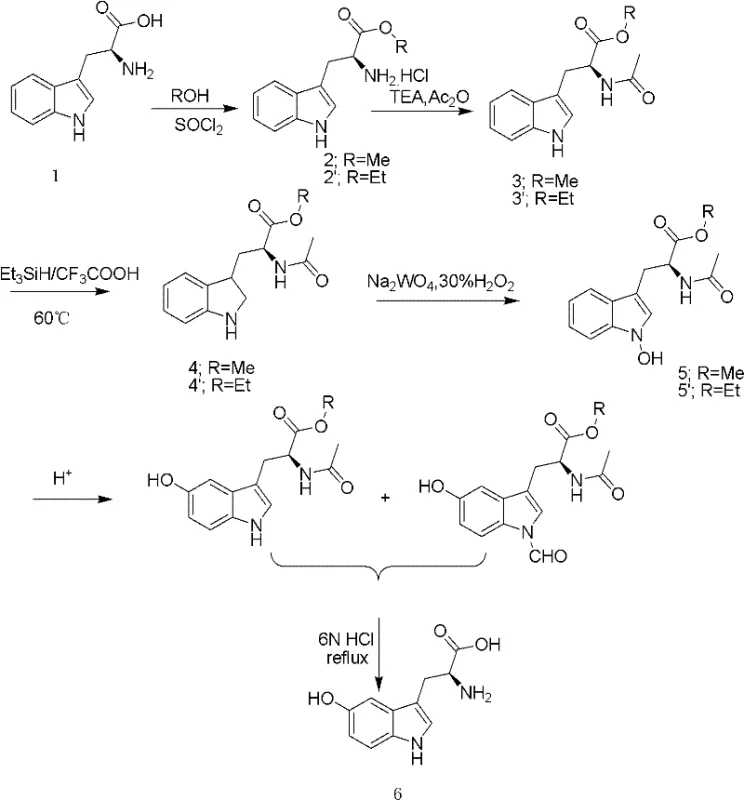
The core innovation of this synthesis lies in the precise manipulation of the indole nucleus to introduce the hydroxyl group at the 5-position. The process begins with the protection of the amino and carboxyl groups of L-tryptophan through esterification and acetylation, creating a stable N-acetyl-L-tryptophan ester intermediate. This protection is crucial as it prevents unwanted side reactions during the subsequent harsh reduction conditions. The indole ring is then selectively reduced using a triethylsilane-trifluoroacetic acid system. This specific reagent combination is highly effective at saturating the double bond of the pyrrole ring without affecting the aromatic benzene ring, generating a dihydroindole (indoline) derivative. This step sets the stage for the critical oxidation event, demonstrating a high level of chemoselectivity that is essential for maintaining the stereochemical integrity of the chiral center derived from the L-tryptophan starting material.
Following the reduction, the nitrogen atom of the indoline ring undergoes oxidation using a sodium tungstate and 30% hydrogen peroxide system to form an N-oxide intermediate. This N-oxide species is thermodynamically unstable and serves as the key driver for the subsequent rearrangement. Under acidic conditions, the hydroxyl group on the nitrogen migrates to the 5-position of the phenyl ring via a mechanism akin to a Meisenheimer-type rearrangement or similar oxidative migration. This elegant transformation effectively installs the pharmacologically active hydroxyl group exactly where it is needed. Finally, acidic hydrolysis removes the acetyl and ester protecting groups, yielding the target levo-5-hydroxytryptophan. This mechanistic pathway avoids the use of heavy metal catalysts often found in direct hydroxylation methods, thereby simplifying the removal of toxic residues and ensuring a cleaner final product profile.
How to Synthesize Levo-5-Hydroxytryptophan Efficiently
The synthesis of this high-value neurochemical intermediate requires strict adherence to reaction parameters to maximize yield and optical purity. The process involves a sequential five-step protocol that transforms bulk L-tryptophan into the final crystalline product. Each step, from the initial formation of the hydrochloride salt to the final crystallization, is optimized to minimize byproduct formation and facilitate easy isolation. The detailed standardized synthesis steps, including specific molar ratios, temperature controls, and workup procedures, are outlined in the guide below to assist R&D teams in replicating this efficient pathway.
- Perform methyl or ethyl esterification on L-tryptophan using thionyl chloride and alcoholic solvent to obtain L-tryptophan ester hydrochloride.
- Conduct dehydrochlorination under alkaline conditions followed by acetylation to form N-acetyl-L-tryptophan ester.
- Reduce the indole ring using a triethylsilane-trifluoroacetic acid system, then oxidize the nitrogen position using sodium tungstate and hydrogen peroxide.
- Execute acidic rearrangement to transfer the hydroxyl group to the 5-position of the phenyl ring and remove the acetyl protecting group.
- Purify the final product through cooling crystallization and macroporous adsorption resin chromatography to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from extraction-based sourcing to this chemical synthesis route represents a strategic opportunity to de-risk the supply of critical active ingredients. The traditional reliance on African plant seeds introduces uncontrollable variables into the supply chain, whereas chemical synthesis offers a predictable, year-round production capability. By adopting this method, companies can secure a more resilient supply line that is immune to crop failures or harvest delays. Furthermore, the use of commodity chemicals like L-tryptophan as the starting material shields the production cost from the speculative volatility often seen in botanical raw material markets, providing a stable foundation for long-term budget planning and cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive and logistically complex botanical extraction steps. By utilizing L-tryptophan, which is produced on a massive industrial scale via fermentation globally, the raw material costs are significantly lower and more transparent than sourcing niche plant seeds. Additionally, the synthetic route avoids the need for specialized enzymatic catalysts or supercritical fluid extraction equipment, which require high capital expenditure and maintenance. The simplified downstream processing, which relies on standard crystallization and resin adsorption rather than complex chromatographic separations of biological sludge, further drives down operational expenses, resulting in substantial cost savings for the final API production.
- Enhanced Supply Chain Reliability: One of the most compelling advantages for supply chain heads is the decoupling of production from agricultural seasons. Plant-based extraction is inherently limited by the annual growth cycle of Griffonia Simplicifolia, creating bottlenecks that can delay projects for months. In contrast, this chemical synthesis can be run continuously in a reactor environment, allowing for just-in-time manufacturing and rapid response to market demand spikes. The robustness of the reaction conditions ensures high reproducibility batch-to-batch, reducing the risk of production failures that could disrupt the continuity of supply for downstream pharmaceutical formulations.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method offers a cleaner profile compared to traditional extraction which generates massive amounts of plant waste. The solvents used in this synthesis, such as methanol, ethanol, and dichloromethane, are well-understood and easily recoverable through distillation, aligning with green chemistry principles of solvent recycling. The absence of heavy metal catalysts simplifies the validation process for regulatory filings, as there is no need for extensive testing for residual metals like palladium or platinum. This ease of scale-up from laboratory to commercial tonnage ensures that the process remains efficient and compliant even as production volumes increase to meet global market needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the comparative data and experimental results presented in the patent documentation, focusing on purity, yield, and operational feasibility. Understanding these details is crucial for technical teams evaluating the viability of switching from extraction to synthetic sourcing for their 5-HTP requirements.
Q: What are the primary advantages of this chemical synthesis route over plant extraction?
A: Unlike plant extraction from Griffonia Simplicifolia, which suffers from seasonal instability and raw material scarcity, this chemical synthesis uses readily available L-tryptophan. It ensures consistent production cycles不受 harvest seasons, significantly enhancing supply chain reliability and reducing raw material price volatility.
Q: How does this method address impurity profiles compared to fermentation?
A: Fermentation methods often introduce complex metabolic byproducts from bacterial strains that are difficult to separate. This chemical route utilizes specific protection and deprotection strategies (acetylation/esterification) that minimize side reactions, allowing for easier purification via crystallization and resin columns to achieve purity levels exceeding 99%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the use of low-toxicity solvents and simple reaction operations suitable for industrial scale-up. The total yield exceeds 45%, and the avoidance of expensive biological enzymes or rare plant materials makes it economically viable for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levo-5-Hydroxytryptophan Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of neurological intermediates like 5-HTP. Our team of expert chemists has extensively analyzed advanced synthetic routes, including the one detailed in patent CN102351775A, to ensure we offer the most efficient production technologies available. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 5-HTP we deliver meets the highest international standards for pharmaceutical and nutraceutical applications.
We invite you to collaborate with us to leverage these technological advancements for your product pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our synthetic capabilities can optimize your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for high-purity pharmaceutical intermediates in the global market.
