Advanced Enzymatic Resolution for High-Purity Alzheimer's Disease Intermediate Manufacturing
Advanced Enzymatic Resolution for High-Purity Alzheimer's Disease Intermediate Manufacturing
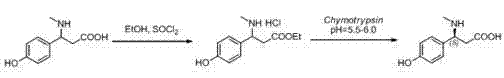
The pharmaceutical industry's relentless pursuit of effective treatments for neurodegenerative disorders, particularly Alzheimer's disease, has placed a premium on the availability of high-purity chiral amino acid intermediates. A pivotal development in this domain is detailed in patent CN103131747A, which outlines a sophisticated method for synthesizing (S)-3-(4-hydroxy phenyl)-3-methylamino propionic acid through biological enzyme chiral resolution. This compound serves as a crucial precursor in the synthesis of bioactive peptides and potential therapeutics for senile dementia, representing a significant advancement over traditional chemical synthesis routes. The innovation lies in leveraging the stereospecific catalytic characteristics of biological enzymes, specifically Chymotrypsin, to achieve exceptional optical purity while maintaining mild reaction conditions. For R&D directors and procurement specialists alike, this technology offers a compelling pathway to secure reliable supplies of complex amino acid derivatives that meet the stringent quality standards required for modern drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure alpha-non-natural amino acids has been fraught with challenges related to harsh reaction conditions and inefficient separation processes. Traditional chemical resolution methods often rely on the formation of diastereomeric salts using expensive chiral resolving agents, which necessitates multiple crystallization steps to achieve acceptable enantiomeric excess. Furthermore, these conventional pathways frequently involve the use of heavy metal catalysts or aggressive reagents that can introduce toxic impurities, complicating the downstream purification process and increasing the environmental burden. The reliance on such rigorous conditions not only escalates operational costs due to energy consumption and waste treatment but also poses significant risks to the structural integrity of sensitive functional groups present in the molecule. Consequently, manufacturers have long sought a more elegant solution that could bypass these inefficiencies while delivering the high fidelity required for pharmaceutical applications.
The Novel Approach
In stark contrast to these legacy methods, the novel approach described in the patent utilizes a biocatalytic strategy that transforms the synthesis landscape through precision and efficiency. By starting with the racemic 3-(4-hydroxy phenyl)-3-methylamino propionic acid, the process first employs a straightforward esterification step to generate the ethyl ester substrate, which is then subjected to highly selective enzymatic hydrolysis. This biological resolution exploits the inherent ability of Chymotrypsin to distinguish between enantiomers, selectively hydrolyzing the desired isomer while leaving the other intact or easily separable. The result is a streamlined workflow that eliminates the need for complex chiral chromatography or excessive recrystallization cycles, directly addressing the pain points of yield loss and purity compromise. This shift towards biocatalysis represents a paradigm change in how we approach the commercial scale-up of complex pharmaceutical intermediates, offering a robust alternative to chemically intensive protocols.
Mechanistic Insights into Chymotrypsin-Catalyzed Hydrolysis
The core of this technological breakthrough resides in the precise mechanistic action of Chymotrypsin (bovine, EC:3.4.21.1), a serine protease known for its specificity towards peptide bonds adjacent to large hydrophobic amino acids. In this specific application, the enzyme facilitates the hydrolysis of the ethyl ester bond in the racemic substrate, but with a profound preference for the stereochemistry of the (S)-enantiomer. The reaction mechanism involves the formation of an acyl-enzyme intermediate, where the active site serine residue attacks the carbonyl carbon of the ester group. The geometry of the enzyme's binding pocket ensures that only the (S)-configured substrate fits correctly to undergo nucleophilic attack, effectively filtering out the (R)-enantiomer which remains largely unreacted or reacts at a negligible rate. This kinetic resolution is further optimized by maintaining the reaction environment at a physiological temperature of 37°C, which maximizes enzyme turnover without denaturing the protein structure.
Crucially, the control of the reaction pH plays a decisive role in determining the final optical purity and yield of the product. The patent specifies a narrow pH window of 5.5 to 6.0, which is meticulously maintained using Lithium Hydroxide rather than the more common Sodium Hydroxide. This specific choice of base is not arbitrary; Lithium ions appear to suppress non-enzymatic background hydrolysis, a side reaction that would otherwise erode the enantiomeric excess by randomly cleaving ester bonds regardless of chirality. By minimizing this non-selective hydrolysis, the process ensures that the observed optical rotation is driven almost exclusively by the enzymatic activity, resulting in an impressive ee value of 98%. This level of impurity control is vital for regulatory compliance, as it drastically reduces the burden on downstream purification units to remove unwanted isomers or degradation products.
How to Synthesize (S)-3-(4-hydroxy phenyl)-3-methylamino propionic acid Efficiently
Implementing this synthesis route requires careful attention to the preparation of the ester substrate and the subsequent enzymatic conditions to ensure reproducibility at scale. The process begins with the conversion of the racemic acid into its ethyl ester hydrochloride salt using thionyl chloride in dehydrated alcohol, a step that proceeds with near-quantitative yield and sets the stage for the biocatalytic resolution. Following isolation, the substrate is dissolved in water and the pH is carefully adjusted before the introduction of the enzyme, creating an environment where the biocatalyst can operate at peak efficiency. The detailed standardized synthetic steps, including specific reagent ratios and workup procedures, are outlined in the guide below to assist technical teams in replicating this high-efficiency protocol.
- Perform esterification of racemic 3-(4-hydroxy phenyl)-3-methylamino propionic acid using thionyl chloride in ethanol under reflux to obtain the ethyl ester hydrochloride substrate.
- Dissolve the ethyl ester substrate in water and adjust the pH to 5.5-6.0 using lithium hydroxide to minimize non-enzymatic hydrolysis.
- Add Chymotrypsin (bovine) at 37°C and maintain pH stability for selective hydrolysis, yielding the high-purity (S)-enantiomer with 98% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enzymatic resolution method offers substantial strategic advantages that extend beyond mere technical feasibility. The elimination of harsh chemical reagents and the reduction in processing steps translate directly into a more resilient and cost-effective supply chain model. By adopting this technology, organizations can mitigate the risks associated with volatile raw material markets for chiral resolving agents and reduce their dependency on complex waste management infrastructure. The inherent simplicity of the process also shortens the overall production cycle time, allowing for faster response to market demands and improved inventory turnover rates without compromising on the quality of the final active pharmaceutical ingredient.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the workflow and the reduction in auxiliary material consumption. By replacing expensive chiral resolving agents and heavy metal catalysts with a reusable biological enzyme, the direct material costs are significantly lowered. Furthermore, the mild reaction conditions eliminate the need for extreme heating or cooling, resulting in reduced energy expenditure for HVAC and reactor maintenance. The high yield of the initial esterification step combined with the efficient resolution means less raw material is wasted, optimizing the overall atom economy of the synthesis and driving down the cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: From a logistics perspective, the use of readily available starting materials and robust biocatalysts enhances the stability of the supply chain. Unlike specialized chemical catalysts that may have long lead times or single-source dependencies, Chymotrypsin is a commercially established enzyme with a stable supply network. The process tolerance allows for flexibility in sourcing solvents, as ethanol can potentially be substituted with tetrahydrofuran if supply constraints arise. This adaptability ensures continuous production capabilities even during periods of global chemical shortages, providing a buffer against external market disruptions and securing the continuity of supply for critical drug intermediates.
- Scalability and Environmental Compliance: The environmental profile of this enzymatic route aligns perfectly with modern green chemistry principles, facilitating easier regulatory approval and community acceptance. The aqueous nature of the enzymatic step reduces the volume of organic solvents required, thereby lowering the facility's VOC emissions and hazardous waste generation. Scaling this process from laboratory to industrial production is straightforward because the reaction does not generate exothermic spikes or require high-pressure equipment, reducing capital expenditure on specialized reactors. This ease of scale-up allows manufacturers to rapidly increase capacity to meet growing demand for Alzheimer's therapeutics while maintaining a minimal environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic resolution technology. These insights are derived directly from the patent specifications and are intended to clarify the operational parameters and quality outcomes expected from this synthesis route. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing lines.
Q: What is the optical purity achieved by this enzymatic method?
A: The patented process utilizing Chymotrypsin achieves an enantiomeric excess (ee) value of 98%, which is critical for pharmaceutical applications requiring high stereochemical integrity.
Q: Why is Lithium Hydroxide preferred for pH adjustment in this reaction?
A: Lithium Hydroxide is specifically recommended over other bases like Sodium Hydroxide because it significantly reduces non-enzymatic hydrolysis, thereby preserving the optical purity of the final product.
Q: What are the optimal reaction conditions for the enzymatic resolution step?
A: The enzymatic hydrolysis should be conducted at a controlled temperature of 37°C with the pH strictly maintained between 5.5 and 6.0 to ensure maximum enzyme activity and selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-3-(4-hydroxy phenyl)-3-methylamino propionic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing high-quality intermediates for the development of next-generation neurotherapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical partners. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify enantiomeric excess and impurity profiles. Our capability to implement complex biocatalytic processes like the one described in CN103131747A positions us as a strategic partner capable of bridging the gap between innovative research and commercial reality.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on transparent and verified performance metrics. Let us collaborate to bring life-saving medications to market faster and more efficiently.
