Revolutionizing Atorvastatin Intermediate Production via Advanced One-Pot Biocatalysis
Revolutionizing Atorvastatin Intermediate Production via Advanced One-Pot Biocatalysis
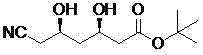
The global demand for high-quality statin intermediates continues to surge as cardiovascular therapies remain a cornerstone of modern medicine. A pivotal advancement in this sector is detailed in patent CN103695486A, which discloses a highly efficient biological preparation method for (3R, 5R)-6-cyano-3,5-dihydroxyhexanoic acid tert-butyl ester. This compound serves as a critical chiral building block for the synthesis of Atorvastatin (Lipitor), one of the world's best-selling pharmaceutical agents. The patented technology leverages a sophisticated multi-enzyme cascade to transform a chloro-substituted ketone precursor directly into the desired diol product under mild conditions. By integrating recombinant ketoreductase, glucose dehydrogenase, and dehalogenase into a single reaction vessel, this innovation eliminates the need for hazardous chemical reductants and extreme temperatures. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic elegance and commercial viability of this route is essential for securing a competitive edge in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key statin intermediate has been plagued by significant safety and economic hurdles. Prior art, such as that disclosed in patent WO2004094243A1, relies heavily on chemical reduction strategies employing borane complexes and sodium borohydride. These traditional protocols necessitate rigorous exclusion of moisture and oxygen, often requiring reactions to be conducted at cryogenic temperatures as low as -50°C. Such harsh conditions not only escalate energy costs but also introduce severe safety risks associated with the handling of pyrophoric borane reagents on an industrial scale. Furthermore, alternative biocatalytic routes described in documents like WO2008042876A2 utilize (5R)-6-cyano-5-hydroxy-3-oxohexanoic acid tert-butyl ester as the starting material. While enzymatic, this approach suffers from the high cost and limited availability of the cyano-substituted precursor, creating a bottleneck in the supply chain. The complexity of protecting group manipulations and the need for separate reduction and substitution steps in older methodologies further diminish their attractiveness for large-scale cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in CN103695486A represents a paradigm shift towards sustainable and economical synthesis. This novel approach utilizes the more abundant and affordable (5R)-6-chloro-5-hydroxy-3-oxohexanoic acid tert-butyl ester as the feedstock. The brilliance of this strategy lies in its "one-pot" design, where reduction of the ketone moiety and nucleophilic substitution of the chlorine atom occur simultaneously within the same reactor. By employing a tri-enzyme system comprising ketoreductase (KRED), glucose dehydrogenase (GDH), and dehalogenase, the process achieves high conversion rates at room temperature and a neutral pH range of 6 to 8. This eliminates the capital expenditure associated with cryogenic reactors and the operational expenses of inert gas protection. The result is a streamlined workflow that drastically simplifies downstream processing, offering a compelling value proposition for commercial scale-up of complex pharmaceutical intermediates while maintaining exceptional stereochemical integrity.
Mechanistic Insights into Multi-Enzyme Cascade Catalysis
The core of this technological breakthrough is the synergistic action of three distinct biocatalysts working in harmony. The recombinant ketoreductase (KRED) is responsible for the stereoselective reduction of the C3 ketone group to a hydroxyl group, establishing the critical (3R) configuration. This reduction consumes NADH as a cofactor, which is continuously regenerated in situ by the glucose dehydrogenase (GDH) through the oxidation of glucose to gluconolactone. Simultaneously, the dehalogenase enzyme facilitates the displacement of the chlorine atom at the C6 position with a cyano group from the cyanating agent (e.g., sodium cyanide), preserving the (5R) stereochemistry. This concurrent catalysis prevents the accumulation of unstable intermediates and drives the equilibrium towards the final product. The use of a triethanolamine hydrochloride buffer system ensures optimal enzyme stability and activity throughout the reaction duration, typically spanning 8 to 12 hours. This precise orchestration of biocatalytic events allows for the direct transformation of a chloro-ketone into a cyano-diol without the need for isolation of intermediate species.
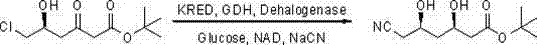
From an impurity control perspective, this enzymatic route offers superior selectivity compared to chemical alternatives. The high specificity of the enzymes minimizes the formation of side products such as over-reduced alcohols or elimination byproducts that are common in chemical reductions. The patent data indicates that the process achieves a cis-selectivity of greater than 99%, ensuring that the resulting intermediate possesses the exact spatial arrangement required for subsequent cyclization into the lactone ring of Atorvastatin. Furthermore, the mild reaction conditions prevent the degradation of the sensitive tert-butyl ester protecting group, which can be susceptible to hydrolysis under acidic or basic chemical conditions. The ability to maintain high optical purity (>98% content) directly from the reactor reduces the burden on purification teams, allowing for simpler crystallization or extraction protocols. This level of control is paramount for meeting the stringent regulatory requirements of global health authorities regarding chiral purity in active pharmaceutical ingredients.
How to Synthesize (3R, 5R)-6-cyano-3,5-dihydroxyhexanoic acid tert-butyl ester Efficiently
Implementing this biocatalytic process requires careful attention to enzyme loading and reaction parameters to maximize yield and efficiency. The patent provides a robust framework for executing this synthesis, emphasizing the importance of maintaining the correct mass ratios between the biocatalysts and the substrate. The procedure involves suspending the lyophilized enzyme powders in a buffered solution, adding the cofactor and glucose source, and then introducing the substrate along with the cyanating agent to maintain pH. Detailed standard operating procedures for enzyme preparation, including fermentation conditions and lysis methods, are critical for ensuring consistent batch-to-batch performance. For process chemists looking to adopt this technology, adhering to the specified molar equivalents and temperature controls is essential to replicate the high yields reported in the examples. The following guide outlines the standardized synthesis steps derived from the patent data to facilitate technology transfer and pilot plant trials.
- Prepare a reaction system in triethanolamine hydrochloride buffer containing recombinant ketoreductase, glucose dehydrogenase prozyme, dehalogenase, cofactor (NAD), and glucose.
- Introduce (5R)-6-chloro-5-hydroxy-3-oxohexanoic acid tert-butyl ester as the substrate and maintain pH between 6 and 8 using a cyanating reagent such as sodium cyanide.
- Allow the reaction to proceed at room temperature until completion, followed by extraction with ethyl acetate and solvent removal to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this biocatalytic route translates into tangible strategic benefits that extend beyond mere technical feasibility. The shift from hazardous chemical reagents to benign enzymatic catalysts fundamentally alters the risk profile of the manufacturing process. By eliminating the need for borane and cryogenic cooling, facilities can operate with lower insurance premiums and reduced safety compliance overheads. The use of readily available starting materials, specifically the chloro-substituted ketone, mitigates supply chain volatility often associated with specialized cyano-precursors. This accessibility ensures a more stable flow of raw materials, reducing the risk of production stoppages due to vendor shortages. Furthermore, the simplified "one-pot" nature of the reaction reduces the number of unit operations, leading to shorter cycle times and increased throughput capacity within existing infrastructure. These factors collectively contribute to a more resilient and cost-effective supply chain for high-value statin intermediates.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the substitution of expensive and dangerous reagents with cost-effective biological alternatives. The elimination of borane reagents removes the necessity for specialized handling equipment and waste disposal protocols associated with hazardous chemicals. Additionally, operating at room temperature significantly lowers energy consumption compared to processes requiring sustained cryogenic conditions. The high yield and selectivity reported in the patent examples mean that less raw material is wasted on byproducts, directly improving the overall material balance and reducing the cost per kilogram of the final API. By consolidating the reduction and substitution steps into a single vessel, manufacturers can also save on solvent usage and labor costs associated with intermediate isolation and purification.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on niche starting materials that have limited global suppliers. This technology circumvents that risk by utilizing a chloro-ketone precursor that is synthetically more accessible and widely produced than its cyano-counterpart. The robustness of the enzymatic system, which tolerates a broad pH range and ambient temperatures, makes the process less susceptible to minor fluctuations in utility supplies or environmental conditions. This resilience ensures that production schedules can be maintained consistently, reducing lead time for high-purity pharmaceutical intermediates and allowing manufacturers to respond more agilely to market demand. The ability to produce enzymes via fermentation also adds a layer of vertical integration potential, further securing the supply chain against external disruptions.
- Scalability and Environmental Compliance: As regulatory pressure mounts for greener manufacturing practices, this biocatalytic route offers a clear path to sustainability. The aqueous nature of the reaction medium and the biodegradability of the enzymes align perfectly with green chemistry principles, minimizing the generation of toxic heavy metal waste or halogenated organic solvents. Scaling this process from laboratory to commercial tonnage is facilitated by the simplicity of the operation, which does not require exotic high-pressure or low-temperature reactors. The reduction in hazardous waste streams simplifies effluent treatment and lowers the environmental footprint of the facility. This compliance advantage is increasingly becoming a deciding factor for multinational corporations when selecting partners for high-purity pharmaceutical intermediate production, as it future-proofs the supply chain against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic synthesis route. These answers are derived directly from the experimental data and claims presented in patent CN103695486A, providing a factual basis for decision-making. Understanding these nuances is critical for R&D teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of this supply source. The clarity provided here aims to bridge the gap between academic patent language and practical industrial application, ensuring all stakeholders have a unified understanding of the process capabilities.
Q: What are the primary advantages of this biocatalytic route over traditional chemical reduction?
A: Unlike traditional methods requiring toxic borane reagents and cryogenic conditions (-50°C), this enzymatic process operates at room temperature and neutral pH, significantly improving operational safety and reducing energy consumption.
Q: How does the starting material cost compare to previous enzymatic methods?
A: This method utilizes (5R)-6-chloro-5-hydroxy-3-oxohexanoic acid tert-butyl ester, which is generally more accessible and cost-effective than the cyano-substituted precursors required in earlier biocatalytic patents like WO2008042876A2.
Q: What level of stereochemical control does this process achieve?
A: The combined action of specific ketoreductases and dehalogenases ensures high cis-selectivity (≥99%) and excellent optical purity, yielding the critical (3R, 5R) configuration required for Atorvastatin activity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (3R, 5R)-6-cyano-3,5-dihydroxyhexanoic acid tert-butyl ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic route described in CN103695486A for the global statin market. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this innovative patent into a robust commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to delivering products that meet stringent purity specifications, backed by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom enzyme engineering or optimization of the fermentation parameters, our R&D division is ready to collaborate on tailoring this process to your specific quality targets.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your API cost structure. By leveraging our capabilities, you can secure a stable supply of this critical intermediate while benefiting from the safety and efficiency of green manufacturing. Please contact us today to request a Customized Cost-Saving Analysis tailored to your project volume. We are prepared to provide specific COA data and comprehensive route feasibility assessments to support your regulatory filings and accelerate your time to market. Let us be your trusted partner in navigating the complexities of modern pharmaceutical synthesis.
