Advanced Synthesis of N-(4-formyl-2-methylphenyl)trimethylacetamide for Scalable Telmisartan Production
Introduction to Novel Pharmaceutical Intermediate Synthesis
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing critical antihypertensive agents, and patent CN113024399A presents a significant breakthrough in the production of Telmisartan precursors. This intellectual property discloses a robust method for preparing N-(4-formyl-2-methylphenyl)trimethylacetamide, a pivotal building block in the angiotensin II receptor blocker (ARB) class of medications. The core innovation lies in utilizing a modified Duff reaction to introduce a formyl group directly onto the aromatic ring, bypassing the cumbersome multi-step sequences traditionally required for such transformations. By leveraging urotropine and trifluoroacetic acid, this process achieves high regioselectivity and yield while maintaining an environmentally friendlier profile compared to legacy chemistries. For R&D directors and procurement specialists, understanding this specific molecular architecture is crucial for securing reliable supply chains for next-generation cardiovascular therapies.
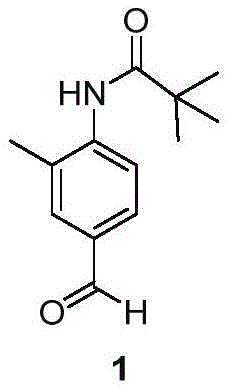
The structural integrity of this intermediate is paramount, as it serves as the foundation for constructing the complex benzimidazole scaffold found in Telmisartan. The molecule features a protected amine group via a pivaloyl moiety, which ensures stability during subsequent coupling reactions while directing electrophilic substitution to the desired para-position relative to the nitrogen. This strategic protection-deprotection strategy is essential for minimizing impurities that could complicate downstream purification. As a reliable pharmaceutical intermediate supplier, recognizing the nuances of this specific substitution pattern allows for better quality control and process optimization. The ability to produce this compound with consistent stereochemical and chemical purity directly impacts the efficacy and safety profile of the final API, making this patent a valuable asset for any manufacturer aiming to dominate the ARB market sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, introducing a formyl group onto a benzene ring has been fraught with chemical challenges and operational inefficiencies that hinder large-scale manufacturing. Traditional approaches often rely on Blanc chloromethylation followed by Sommelet oxidation, a sequence that is not only lengthy but also involves handling hazardous chloromethyl ethers and generating significant amounts of toxic waste. Alternatively, the Vilsmeier-Haack reaction, while popular, necessitates the use of phosphorus oxychloride and dimethylformamide, creating severe corrosion issues in reactors and complicating wastewater treatment due to high phosphorus content. These legacy methods frequently suffer from poor regioselectivity, leading to difficult-to-separate isomers that reduce overall yield and increase the cost of goods sold. Furthermore, the harsh conditions required for these transformations can degrade sensitive functional groups, necessitating additional protection steps that further elongate the synthetic timeline and erode profit margins for contract manufacturing organizations.
The Novel Approach
In stark contrast, the methodology outlined in CN113024399A offers a streamlined, one-pot solution that dramatically simplifies the introduction of the aldehyde functionality. By employing the Duff reaction conditions using hexamethylenetetramine (urotropine) in a trifluoroacetic acid medium, the process achieves direct formylation with exceptional precision. This approach eliminates the need for corrosive phosphorus reagents and toxic chlorinating agents, thereby reducing the environmental footprint and lowering the barrier for regulatory compliance. The reaction proceeds under relatively mild thermal conditions compared to extreme alternatives, preserving the integrity of the pivaloyl protecting group and ensuring a cleaner reaction profile. For supply chain heads, this translates to a more resilient production schedule with fewer bottlenecks caused by equipment maintenance or waste disposal logistics, ultimately facilitating cost reduction in pharmaceutical intermediate manufacturing through operational excellence.
Mechanistic Insights into Duff Reaction Formylation
The success of this synthesis hinges on the precise mechanistic pathway of the Duff reaction, which operates through an electrophilic aromatic substitution mechanism facilitated by the formation of an reactive iminium species. In the presence of strong acid like trifluoroacetic acid, urotropine decomposes to generate formaldehyde imine equivalents that act as potent electrophiles. These species attack the electron-rich aromatic ring of the N-(o-tolyl)trimethylacetamide substrate, specifically targeting the position para to the activating amide group due to steric and electronic directing effects. The bulky tert-butyl group on the amide nitrogen further enhances this selectivity by blocking ortho-positions, ensuring that the formyl group is installed exclusively at the C4 position. This high degree of regiocontrol is critical for minimizing the formation of regioisomeric impurities that would otherwise require costly chromatographic separation steps later in the synthesis of Telmisartan.
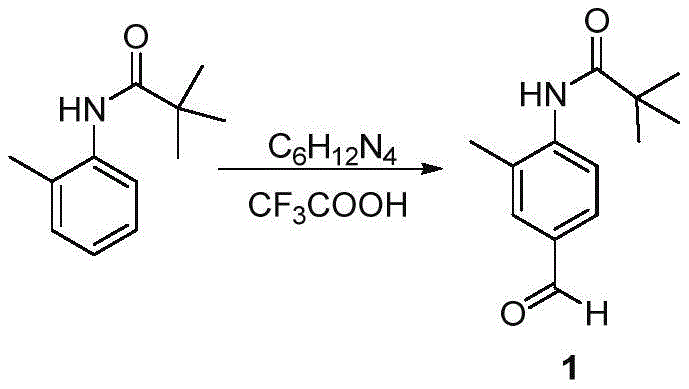
Following the initial electrophilic attack, the resulting intermediate undergoes hydrolysis upon the addition of water, releasing the free aldehyde and regenerating ammonia derivatives. The use of trifluoroacetic acid serves a dual purpose: it acts as both the solvent and the catalyst, protonating the urotropine to facilitate the generation of the electrophile while also solubilizing the organic substrates. The subsequent hydrolysis step is carefully controlled by temperature and time to ensure complete conversion of the aminomethyl intermediate to the aldehyde without promoting side reactions such as aldol condensation or over-oxidation. Understanding these kinetic parameters allows process chemists to fine-tune the reaction for maximum throughput. The result is a high-purity product where the impurity profile is dominated by easily removable starting materials rather than complex structural analogs, significantly easing the burden on quality assurance teams during batch release testing.
How to Synthesize N-(4-formyl-2-methylphenyl)trimethylacetamide Efficiently
Executing this synthesis requires careful attention to stoichiometry and thermal management to replicate the high yields reported in the patent literature. The process begins with the sequential addition of the substrate, acid, and urotropine, followed by a controlled heating phase to drive the formylation to completion. Once the reaction is deemed complete via TLC monitoring, a hydrolysis step involving water addition and reflux is critical to unmask the aldehyde functionality. The workup procedure utilizes standard liquid-liquid extraction techniques with toluene, allowing for the recovery of the expensive trifluoroacetic acid from the aqueous layer, which aligns with green chemistry principles. Detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are essential for maintaining consistency.
- Combine N-(o-tolyl)trimethylacetamide with excess trifluoroacetic acid and urotropine in a reactor.
- Heat the mixture to 120-125°C and maintain reaction for 2-3 hours to ensure complete conversion.
- Hydrolyze the intermediate by adding water, reflux, then extract with toluene and purify to obtain the final solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this novel synthetic route offers tangible benefits that extend beyond mere chemical elegance. The shift away from hazardous reagents like phosphorus oxychloride reduces the need for specialized corrosion-resistant equipment, allowing manufacturers to utilize standard glass-lined or stainless steel reactors. This flexibility lowers capital expenditure requirements and expands the pool of potential contract manufacturing partners who can execute the chemistry safely. Moreover, the simplification of the synthetic sequence from multiple steps to a single pot operation drastically reduces the total processing time and labor costs associated with isolation and purification between stages. These efficiencies contribute to a more competitive pricing structure for the final intermediate, enabling pharmaceutical companies to optimize their cost of goods sold without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents combined with a reduced number of unit operations leads to substantial savings in raw material and utility costs. By avoiding the use of phosphorus-based chemicals, facilities save significantly on waste treatment fees and neutralization agents, which are often hidden costs in traditional formylation processes. The high yield reported in the examples minimizes the loss of valuable starting materials, ensuring that every kilogram of input translates effectively into saleable output. Additionally, the ability to recover and recycle trifluoroacetic acid from the aqueous waste stream further enhances the economic viability of the process, creating a closed-loop system that maximizes resource utilization.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as urotropine and trifluoroacetic acid ensures a stable and secure supply of raw materials, mitigating the risk of shortages that often plague specialty reagent markets. Since these inputs are produced on a massive global scale, price volatility is minimal, allowing for accurate long-term budgeting and forecasting. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by sensitivity to moisture or oxygen, common issues with organometallic catalysts. This reliability is crucial for maintaining continuous API production lines, preventing costly downtime and ensuring that downstream customers receive their orders on time, every time.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with thermal profiles that are manageable even in large-volume reactors, reducing the risk of runaway exotherms. The reduction in toxic byproducts simplifies the environmental permitting process for manufacturing sites, facilitating faster approval for new production lines. Waste streams are primarily aqueous and organic solvents that can be treated using conventional methods, avoiding the complex disposal protocols required for heavy metal or phosphorus-containing wastes. This alignment with increasingly stringent global environmental regulations future-proofs the supply chain against potential legislative changes, ensuring long-term operational continuity for the manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this key intermediate. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this Duff reaction route over traditional formylation methods?
A: This method eliminates the need for hazardous phosphorus oxychloride used in Vilsmeier reactions and avoids the multi-step complexity of Blanc chloromethylation, resulting in higher purity and simplified waste treatment.
Q: What is the expected yield and purity profile for this intermediate?
A: Experimental data indicates yields exceeding 80% with high product purity, achieved through a straightforward extraction and crystallization process that minimizes side reactions.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the use of commercially available raw materials like urotropine and the absence of sensitive catalytic systems make this route highly robust and easily scalable for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(4-formyl-2-methylphenyl)trimethylacetamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antihypertensive medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical supply chains. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. By adopting advanced synthetic strategies like the Duff reaction described in CN113024399A, we offer our partners a distinct competitive advantage through superior process efficiency and product consistency.
We invite you to collaborate with us to optimize your Telmisartan supply chain and achieve your cost reduction goals. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your project timelines and quality objectives effectively.
