Advanced Iridium-Catalyzed Synthesis of Unsaturated Primary Alcohols for Commercial Scale-up
Advanced Iridium-Catalyzed Synthesis of Unsaturated Primary Alcohols for Commercial Scale-up
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for greener, safer, and more economically viable synthetic pathways. Patent CN110015947B introduces a groundbreaking methodology for the synthesis of unsaturated primary alcohols, a critical class of compounds serving as essential building blocks in the pharmaceutical and agrochemical industries. This innovative approach leverages a transition metal iridium catalyst to facilitate hydrogen transfer reactions, fundamentally shifting away from traditional, hazardous reduction methods. By utilizing isopropanol as both the solvent and the hydrogen source, this technology addresses long-standing challenges regarding safety, waste management, and process efficiency. For R&D directors and procurement managers seeking a reliable unsaturated primary alcohol supplier, understanding the technical nuances of this patent is crucial for optimizing supply chains and reducing manufacturing costs in pharmaceutical intermediate manufacturing. The following analysis provides a deep dive into the mechanistic advantages and commercial implications of this novel synthetic route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of unsaturated primary alcohols has relied heavily on conventional reduction techniques that pose substantial operational and environmental risks. Traditional methods often involve high-temperature and high-pressure hydrogenation using molecular hydrogen gas, which requires specialized, expensive equipment and rigorous safety protocols to mitigate explosion hazards. Alternatively, the use of inorganic reducing agents such as sodium borohydride generates significant amounts of inorganic waste salts, complicating downstream purification and increasing the burden on waste treatment facilities. Furthermore, these conventional routes frequently suffer from poor chemoselectivity, where the carbon-carbon double bond is inadvertently reduced alongside the carbonyl group, leading to complex impurity profiles that are difficult and costly to separate. The reliance on harsh conditions and hazardous reagents not only inflates the operational expenditure but also creates bottlenecks in the commercial scale-up of complex fine chemical intermediates, limiting the agility of the supply chain.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN110015947B offers a refined and sustainable alternative through transfer hydrogenation. This novel approach utilizes isopropanol, a cheap, safe, and non-toxic alcohol, as the hydrogen donor, effectively eliminating the need for high-pressure hydrogen gas or stoichiometric inorganic reductants. The reaction is catalyzed by a specific transition metal iridium complex, which operates efficiently under mild conditions with remarkably low catalyst loading. This shift in methodology drastically simplifies the reaction setup, allowing for standard glass-lined or stainless steel reactors without the need for high-pressure ratings. The use of isopropanol also serves a dual purpose as the reaction solvent, streamlining the process workflow and reducing the volume of organic solvents required. Consequently, this method aligns perfectly with the principles of green chemistry, offering a pathway for cost reduction in pharmaceutical intermediate manufacturing while enhancing overall process safety and environmental compliance.

Mechanistic Insights into Iridium-Catalyzed Transfer Hydrogenation
The core of this technological breakthrough lies in the sophisticated interaction between the unsaturated aldehyde substrate and the iridium catalyst complex within the isopropanol medium. The reaction mechanism involves a catalytic cycle where the iridium center coordinates with the alcohol solvent to facilitate a beta-hydride elimination, generating a reactive metal-hydride species in situ. This active hydride species then selectively transfers hydrogen to the carbonyl carbon of the unsaturated aldehyde, reducing it to the corresponding primary alcohol without affecting the conjugated or isolated carbon-carbon double bonds. This high level of chemoselectivity is paramount for R&D directors focused on purity and impurity profiles, as it prevents the formation of saturated alcohol byproducts that are common in traditional hydrogenation. The preservation of the unsaturated moiety is critical for downstream functionalization in drug synthesis, ensuring that the intermediate retains the necessary reactivity for subsequent coupling reactions. The efficiency of this cycle allows for high atom economy, minimizing waste and maximizing the yield of the target high-purity unsaturated primary alcohol.
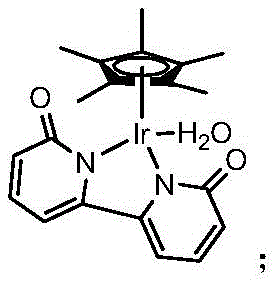
Furthermore, the specific structure of the iridium complex plays a pivotal role in controlling the reaction kinetics and selectivity. The ligand environment surrounding the iridium metal center is designed to stabilize the active catalytic species while preventing catalyst deactivation or aggregation during the reaction course. This stability allows the catalyst to function effectively at low concentrations, typically around 0.2 mol% relative to the substrate, which significantly reduces the cost associated with precious metal usage. The reaction conditions are optimized to operate at temperatures not lower than 82°C, ensuring sufficient energy for the hydrogen transfer while remaining well below the thresholds that might trigger thermal decomposition or side reactions. For supply chain heads, this robustness translates to consistent batch-to-batch reproducibility and reduced lead time for high-purity unsaturated primary alcohols. The ability to run the reaction under nitrogen protection with simple workup procedures further enhances the operational feasibility, making it an attractive option for both laboratory-scale development and large-scale commercial production.
How to Synthesize Unsaturated Primary Alcohol Efficiently
Implementing this synthesis route requires a clear understanding of the operational parameters to maximize yield and purity while maintaining safety standards. The process begins with the precise charging of unsaturated aldehyde, the iridium catalyst complex, and isopropanol into a reaction vessel, ensuring an inert atmosphere to prevent oxidation. Detailed standardized synthesis steps are essential for reproducibility, covering aspects such as addition rates, temperature ramping, and monitoring techniques. The reaction mixture is then heated in an oil bath for a duration of not less than 6 hours, allowing the hydrogen transfer to proceed to completion. Following the reaction, the workup involves cooling the mixture to room temperature and removing the solvent via rotary evaporation, followed by column chromatography to isolate the objective compound. For a comprehensive guide on the specific stoichiometry and purification protocols, please refer to the standardized procedure outlined below.
- Prepare the reaction mixture by adding unsaturated aldehyde, transition metal iridium catalyst complex, and isopropanol into a reaction vessel under nitrogen protection.
- Heat the reaction mixture in an oil bath to a temperature not lower than 82°C and maintain for at least 6 hours to ensure complete hydrogen transfer.
- Cool the mixture to room temperature, remove the solvent via rotary evaporation, and perform column chromatography separation to isolate the pure unsaturated primary alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this iridium-catalyzed transfer hydrogenation method offers substantial strategic advantages for procurement managers and supply chain leaders. The elimination of high-pressure hydrogenation equipment significantly reduces capital expenditure (CAPEX) requirements, allowing manufacturers to utilize existing standard reactor infrastructure for production. This flexibility enhances supply chain reliability by reducing dependency on specialized facilities that may have limited availability or long booking lead times. Additionally, the use of isopropanol as a benign reagent simplifies regulatory compliance and waste disposal processes, leading to substantial cost savings in environmental management. The low catalyst loading further contributes to cost reduction in manufacturing by minimizing the consumption of expensive precious metals, which is a critical factor in maintaining competitive pricing for fine chemical intermediates. Overall, this technology provides a robust framework for scaling production while mitigating operational risks.
- Cost Reduction in Manufacturing: The transition to this catalytic method eliminates the need for costly inorganic reducing agents and high-pressure infrastructure, directly lowering both material and equipment costs. By using isopropanol as a dual-purpose solvent and hydrogen source, the process reduces the volume of raw materials required and simplifies the supply logistics. The low loading of the iridium catalyst ensures that precious metal costs are minimized, contributing to a more favorable cost structure for the final product. These efficiencies collectively drive down the unit cost of production, enabling more competitive pricing strategies in the global market for pharmaceutical intermediates without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable reagents like isopropanol ensures a consistent supply of raw materials, reducing the risk of production delays caused by reagent shortages. The mild reaction conditions and robust catalyst performance enhance process stability, leading to higher success rates in batch production and fewer interruptions due to technical failures. This reliability is crucial for maintaining continuous supply to downstream customers, particularly in the pharmaceutical sector where consistency is paramount. By streamlining the synthesis process, manufacturers can respond more quickly to market demand fluctuations, ensuring that high-purity unsaturated primary alcohols are available when needed.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this method facilitate easier scale-up from laboratory to commercial production without significant process re-engineering. The reduction in hazardous waste generation simplifies environmental compliance and lowers the costs associated with waste treatment and disposal. This environmental advantage is increasingly important as regulatory pressures mount globally, making sustainable manufacturing practices a key differentiator. The ability to scale efficiently while maintaining a low environmental footprint positions this technology as a future-proof solution for the production of complex fine chemical intermediates, aligning with corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of unsaturated primary alcohols using this patented method. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, providing clarity on safety, selectivity, and scalability. Understanding these aspects is vital for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios. The insights provided here aim to bridge the gap between theoretical chemical potential and practical industrial application, ensuring informed decision-making.
Q: What are the safety advantages of using isopropanol over traditional reducing agents?
A: Unlike sodium borohydride or high-pressure hydrogen gas, isopropanol serves as both a safe, non-toxic solvent and hydrogen donor, eliminating explosion risks and hazardous waste disposal issues associated with inorganic reducing agents.
Q: How does the iridium catalyst affect the selectivity of the reaction?
A: The specific iridium complex facilitates chemoselective reduction of the carbonyl group while preserving the carbon-carbon double bond, ensuring high purity of the unsaturated primary alcohol without over-reduction side products.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the mild reaction conditions, low catalyst loading of 0.2 mol%, and absence of high-pressure equipment make this process highly scalable and economically viable for commercial manufacturing of fine chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Unsaturated Primary Alcohol Supplier
The technological potential of this iridium-catalyzed synthesis route represents a significant opportunity for optimizing the production of key pharmaceutical and fine chemical intermediates. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative method to fruition. Our facility is equipped with stringent purity specifications and rigorous QC labs to ensure that every batch of unsaturated primary alcohol meets the highest international standards. We understand the critical nature of these intermediates in the drug development pipeline and are committed to delivering consistent quality and reliability. Our technical team is well-versed in handling complex organometallic catalysis, ensuring that the transition from patent to commercial reality is seamless and efficient.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that evaluates how this green method can optimize your specific supply chain. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your target molecules. Our goal is to provide not just a product, but a comprehensive solution that enhances your R&D efficiency and commercial competitiveness. Let us help you navigate the complexities of fine chemical manufacturing with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
