Advanced Green Manufacturing of 2,5-Diaminotoluene for Global Pharmaceutical and Cosmetic Markets
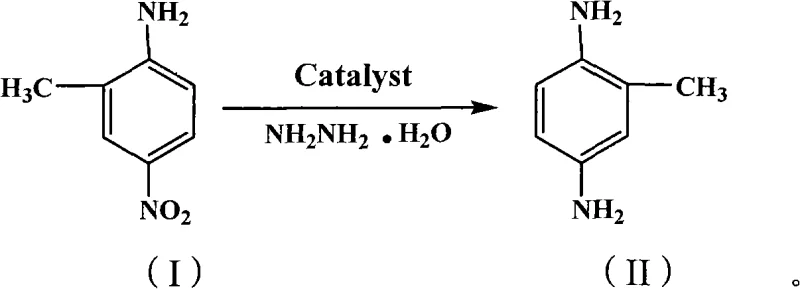
The global demand for high-purity aromatic diamines, particularly 2,5-diaminotoluene, continues to surge across the pharmaceutical and specialty cosmetic sectors, driving an urgent need for sustainable manufacturing technologies. Patent CN101659620A introduces a groundbreaking green synthetic method that fundamentally redefines the production landscape for this critical intermediate. By leveraging a novel magnetic Mg/Al-hydrotalcite solid alkali catalyst in conjunction with hydrazine hydrate, this technology replaces hazardous traditional reagents with a cleaner, more efficient system. The innovation lies not just in the chemical transformation but in the engineering of the catalyst itself, which combines basic catalytic activity with magnetic separability. This dual functionality addresses two of the most persistent challenges in fine chemical manufacturing: waste generation and catalyst recovery. For R&D directors and process engineers, this patent represents a viable pathway to achieving "zero release" goals while maintaining rigorous quality standards required by international regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2,5-diaminotoluene has relied heavily on classical reduction techniques that are increasingly untenable in the modern regulatory environment. The most common method involves the use of iron powder in acidic media, a process notorious for generating vast quantities of iron sludge and acidic wastewater that require expensive treatment and disposal protocols. Alternatively, catalytic hydrogenation is employed, which, while cleaner, necessitates high-pressure reactors and specialized safety infrastructure, leading to prohibitive capital expenditures for many manufacturers. These conventional routes often suffer from issues related to over-reduction, metal contamination in the final product, and difficult work-up procedures that compromise overall yield. The environmental footprint of these legacy methods is substantial, creating significant liability for supply chain managers who must navigate tightening global emissions standards and waste disposal regulations.
The Novel Approach
In stark contrast, the methodology disclosed in CN101659620A utilizes a hydrazine hydrate reduction system mediated by a magnetically modified solid base catalyst. This approach operates under mild conditions, typically between 30°C and 50°C, eliminating the need for high-pressure equipment and reducing energy consumption significantly. The use of hydrazine hydrate as a reducing agent provides a clean source of hydrogen equivalents, producing nitrogen gas and water as benign by-products rather than toxic sludge. Crucially, the magnetic nature of the Mg/Al-hydrotalcite catalyst allows for rapid separation from the reaction mixture using external magnetic fields, bypassing the need for complex filtration or centrifugation steps. This streamlined workflow not only simplifies the operational protocol but also enhances the recovery rate of the catalyst, enabling multiple reuse cycles and drastically reducing raw material costs over the long term.
Mechanistic Insights into Magnetic Mg/Al-Hydrotalcite Catalyzed Reduction
The core of this technological advancement lies in the unique structural and functional properties of the magnetic Mg/Al-hydrotalcite solid alkali. Hydrotalcites are layered double hydroxides known for their tunable basicity and thermal stability, but the modification with magnetic components transforms them into multifunctional heterogeneous catalysts. In the reduction of 2-methyl-4-nitroaniline, the basic sites on the hydrotalcite surface likely facilitate the activation of hydrazine, promoting the transfer of hydrogen species to the nitro group. The magnetic component, typically derived from iron oxides integrated into the lattice, does not merely serve as a handle for separation; it may also participate electronically in the redox cycle, enhancing the overall catalytic efficiency. This synergistic effect ensures that the reduction proceeds selectively to the amine without affecting other sensitive functional groups, a critical factor for maintaining the integrity of complex molecular architectures in downstream applications.
Furthermore, the heterogeneity of the catalyst plays a pivotal role in impurity control, a primary concern for R&D directors overseeing quality assurance. Unlike homogeneous catalysts that dissolve into the reaction medium and require extensive purification to remove trace metals, this solid base catalyst remains physically distinct throughout the process. The hot filtration step effectively removes the catalyst before crystallization, ensuring that the final product is free from heavy metal contamination. This is particularly vital for applications in hair cosmetics and pharmaceuticals, where strict limits on residual metals are enforced. The mechanism also minimizes the formation of azo or azoxy by-products, which are common pitfalls in nitro reductions, thereby simplifying the purification process and boosting the overall yield of the desired 2,5-diaminotoluene. The ability to recycle the catalyst further stabilizes the process chemistry, leading to consistent batch-to-batch reproducibility.
How to Synthesize 2,5-Diaminotoluene Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production, emphasizing simplicity and safety. The process begins with the suspension of the magnetic catalyst in an alcohol solvent, typically ethanol, followed by the addition of the nitro-aniline starting material. Upon heating to the optimal range of 30°C to 50°C, hydrazine hydrate is introduced gradually to control the exotherm and ensure smooth gas evolution. The reaction progress is monitored until completion, after which the magnetic catalyst is separated, and the product is isolated via cooling crystallization. For detailed standard operating procedures and specific parameter optimization, please refer to the step-by-step guide below.
- Prepare the reaction mixture by combining 2-methyl-4-nitroaniline, ethanol solvent, and magnetic Mg/Al-hydrotalcite solid alkali catalyst in a reactor equipped with stirring and heating capabilities.
- Heat the mixture to 30-50°C and slowly add hydrazine hydrate as the reducing agent, maintaining the temperature for 1 to 20 hours to ensure complete reduction of the nitro group.
- Filter the hot reaction mixture to recover the magnetic catalyst, cool the filtrate to below 0°C to crystallize the product, and dry the resulting white crystals to obtain high-purity 2,5-diaminotoluene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this green synthetic route offers compelling economic and logistical benefits that extend beyond mere regulatory compliance. The elimination of corrosive acids and the reduction of hazardous waste streams directly translate to lower operational expenditures related to waste treatment and facility maintenance. Moreover, the mild reaction conditions reduce the wear and tear on reactor vessels and ancillary equipment, extending asset life and minimizing downtime for repairs. The simplicity of the work-up procedure, characterized by magnetic separation and straightforward crystallization, reduces labor hours and increases throughput capacity, allowing manufacturers to respond more agilely to market demand fluctuations without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The implementation of a recyclable magnetic catalyst fundamentally alters the cost structure of 2,5-diaminotoluene production. By enabling the recovery and reuse of the catalyst over multiple cycles, the consumption of expensive catalytic materials is drastically reduced, leading to significant long-term savings. Additionally, the avoidance of high-pressure hydrogenation equipment eliminates the need for costly specialized infrastructure and the associated insurance premiums, while the use of ethanol as a solvent ensures compatibility with existing recovery systems, further optimizing solvent costs.
- Enhanced Supply Chain Reliability: The reliance on readily available raw materials such as hydrazine hydrate and ethanol, coupled with a robust catalyst system, mitigates the risk of supply disruptions often associated with specialized reagents. The process operates at atmospheric pressure, removing the dependency on hydrogen gas supply chains which can be volatile and logistically challenging in certain regions. This decentralization of resource requirements enhances the resilience of the manufacturing network, ensuring consistent delivery schedules even during periods of global raw material scarcity or logistical bottlenecks.
- Scalability and Environmental Compliance: From a scalability perspective, the transition from batch to continuous flow processing is facilitated by the heterogeneous nature of the catalyst, which can be packed into fixed-bed reactors for larger scale operations. The green credentials of this process, characterized by minimal waste generation and the absence of heavy metal effluents, ensure seamless compliance with increasingly stringent environmental regulations such as REACH and TSCA. This proactive alignment with sustainability goals not only future-proofs the supply chain against regulatory shocks but also enhances the brand value of the end products in eco-conscious markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of using magnetic Mg/Al-hydrotalcite over traditional iron powder reduction?
A: The magnetic Mg/Al-hydrotalcite catalyst offers superior environmental performance by eliminating the massive iron sludge waste associated with iron powder reduction. Furthermore, its magnetic properties allow for effortless separation and recycling without complex filtration, significantly lowering operational costs and improving process sustainability.
Q: How does this green synthesis method impact product purity for sensitive applications like hair dyes?
A: This method achieves high product purity, often exceeding 98%, by avoiding the side reactions and metal contamination common in acidic reduction processes. The mild reaction conditions and selective reduction capability of the hydrazine system ensure a clean impurity profile, which is critical for high-grade cosmetic and pharmaceutical specifications.
Q: Is this process scalable for industrial production of 2,5-diaminotoluene?
A: Yes, the process is designed for industrial scalability. It operates at atmospheric pressure and moderate temperatures (30-50°C), removing the need for expensive high-pressure hydrogenation equipment. The simplicity of the work-up procedure and the recyclability of the catalyst make it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Diaminotoluene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the green synthetic methods described in CN101659620A and are fully equipped to bring this technology to commercial fruition. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to plant floor is seamless and efficient. Our state-of-the-art facilities are designed to handle sensitive catalytic processes with precision, supported by rigorous QC labs that enforce stringent purity specifications to meet the exacting demands of the global pharmaceutical and cosmetic industries.
We invite forward-thinking partners to collaborate with us to leverage this advanced manufacturing capability for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your sourcing strategy is built on a foundation of innovation, reliability, and sustainable growth.
