Revolutionizing Statin Side Chain Synthesis: Advanced Biocatalytic Reduction for Commercial Scale Manufacturing
The global demand for HMG-CoA reductase inhibitors, commonly known as statins, continues to drive significant innovation in the synthesis of their chiral side chains. Patent CN102965403A introduces a transformative biological preparation method for tert-butyl(3R,5S)-6-chloro-3,5-dihydroxyhexanoate, a critical intermediate in the production of these life-saving medications. This technology leverages a specialized recombinant ketoreductase to facilitate a highly stereoselective reduction reaction, marking a substantial departure from traditional chemical synthesis routes that often rely on hazardous reagents and extreme conditions. By utilizing a robust enzymatic system coupled with an efficient cofactor regeneration mechanism, this patent addresses long-standing challenges in process economics and environmental sustainability. For pharmaceutical manufacturers seeking a reliable pharmaceutical intermediates supplier, understanding the nuances of this biocatalytic advancement is essential for optimizing supply chain resilience and product quality. The method not only ensures high optical purity but also streamlines the manufacturing workflow, making it an attractive candidate for large-scale commercial adoption in the competitive landscape of fine chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral hydroxy esters like tert-butyl(3R,5S)-6-chloro-3,5-dihydroxyhexanoate has been fraught with significant technical and safety hurdles when relying on conventional chemical reduction methodologies. Traditional approaches frequently necessitate the use of highly reactive and pyrophoric reducing agents, such as borane complexes, which must be handled at cryogenic temperatures often reaching as low as minus 70 degrees Celsius to maintain stereocontrol. These extreme conditions impose severe constraints on reactor design and energy consumption, drastically increasing the capital expenditure required for safe operation. Furthermore, the handling of such hazardous materials introduces substantial occupational health risks and complicates waste disposal protocols, creating a heavy burden on environmental compliance teams. In addition to safety concerns, chemical methods often struggle to achieve the requisite enantiomeric excess without complex resolution steps, leading to yield losses and increased material costs. The reliance on organic solvents and harsh reagents also generates significant volumes of toxic waste, conflicting with the growing industry mandate for greener manufacturing practices. Consequently, there is an urgent need for alternative synthetic strategies that can deliver high purity without compromising safety or sustainability.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in patent CN102965403A utilizes a recombinant ketoreductase to catalyze the reduction under remarkably mild and environmentally benign conditions. This biological pathway operates effectively in an aqueous phase buffer solution with a pH range of 6.0 to 9.0, eliminating the need for volatile organic solvents and extreme temperature controls. The core innovation lies in the specific selection of the biocatalyst and the cofactor regeneration system, which together enable a highly efficient transformation of the substrate tert-butyl(S)-6-chloro-5-carbonyl-3-hydroxyhexanoate. By employing isopropyl alcohol as the hydrogen donor, the process avoids the complications associated with auxiliary enzyme systems found in earlier biocatalytic attempts. This streamlined configuration not only enhances reaction efficiency but also simplifies the downstream processing requirements, as the byproducts generated are far easier to manage than those from chemical or older biological routes. The result is a robust manufacturing process that aligns perfectly with modern principles of green chemistry while delivering the high purity demanded by regulatory agencies for active pharmaceutical ingredients.
Mechanistic Insights into Recombinant Ketoreductase Catalysis
The heart of this technological breakthrough is the sophisticated catalytic cycle driven by the recombinant ketoreductase, which exhibits exceptional specificity for the target carbonyl group. The enzyme facilitates the transfer of a hydride ion from the reduced cofactor NAD(P)H to the substrate, simultaneously protonating the oxygen atom to form the desired hydroxyl group with precise stereochemistry. Crucially, the system incorporates an elegant cofactor regeneration loop where isopropyl alcohol serves as the sacrificial hydrogen donor, converting the oxidized NAD(P)+ back into its active reduced form. This internal recycling mechanism ensures that only catalytic amounts of the expensive cofactor are required, significantly driving down the raw material costs associated with the reaction. The elimination of glucose dehydrogenase and glucose, which were standard in previous generations of biocatalytic processes, prevents the accumulation of gluconic acid. This is a pivotal improvement, as the absence of acidic byproducts removes the necessity for continuous alkali addition to maintain pH stability, thereby preserving enzyme activity over extended reaction periods. The kinetic profile of this reaction allows for high conversion rates even at moderate temperatures, demonstrating the superior catalytic efficiency of the engineered enzyme variant.
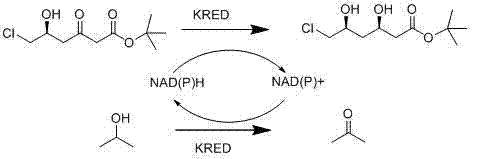
Beyond the primary reduction event, the impurity control mechanism inherent in this process is equally critical for ensuring the quality of the final pharmaceutical intermediate. In traditional glucose-dependent systems, the co-production of gluconic acid necessitates neutralization with bases, which can lead to the formation of salts that complicate product isolation and potentially degrade the enzyme. By switching to an isopropyl alcohol-driven system, the only major byproduct generated is acetone, which is highly volatile and can be easily removed during the solvent evaporation or extraction stages. This fundamental shift in reaction chemistry minimizes the risk of side reactions that could compromise the stereochemical integrity of the product. Furthermore, the aqueous buffer environment provides a stable matrix that protects the protein structure of the ketoreductase from denaturation, ensuring consistent performance throughout the batch cycle. The combination of high selectivity and clean byproduct profiles means that the crude product requires less rigorous purification, reducing the overall number of unit operations and minimizing material loss. For R&D directors focused on impurity profiles, this mechanistic clarity offers a clear path to achieving purity specifications that exceed 99 percent diastereomeric excess without resorting to costly chromatographic separations.
How to Synthesize tert-Butyl(3R,5S)-6-chloro-3,5-dihydroxyhexanoate Efficiently
Implementing this synthesis route requires careful attention to the preparation of the biocatalyst and the optimization of reaction parameters to maximize yield and throughput. The process begins with the cultivation of recombinant E. coli expressing the specific ketoreductase gene, followed by cell lysis and lyophilization to produce a stable enzyme powder suitable for industrial use. Operators must precisely control the pH of the reaction buffer, with experimental data indicating that a pH of 8.0 yields optimal transformation efficiency compared to more acidic or alkaline conditions. The ratio of substrate to hydrogen donor is also a critical variable, with the patent specifying a molar excess of isopropyl alcohol to drive the equilibrium towards product formation. Detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the operational protocol. Adhering to these parameters ensures that the reaction proceeds to completion within a practical timeframe, typically around 24 hours, while maintaining the structural integrity of the sensitive chlorohydrin moiety. This level of procedural detail is essential for technology transfer teams aiming to replicate the laboratory success of the patent in a pilot or production plant setting.
- Prepare the reaction system by dissolving the substrate tert-butyl(S)-6-chloro-5-carbonyl-3-hydroxyhexanoate in an aqueous buffer solution adjusted to pH 8.0.
- Add recombinant ketoreductase lyophilized powder, NAD+ cofactor, and isopropyl alcohol as the hydrogen donor to the mixture.
- Maintain the reaction at 30°C with stirring until conversion exceeds 99%, then extract the product using ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this biocatalytic technology presents a compelling value proposition centered on cost reduction in pharmaceutical intermediates manufacturing and enhanced operational reliability. The elimination of hazardous chemical reagents and the shift to aqueous processing significantly lowers the barriers to entry for safe manufacturing, reducing the need for specialized containment infrastructure and expensive safety monitoring systems. By removing the dependency on glucose dehydrogenase and glucose, the process simplifies the bill of materials, reducing the number of qualified suppliers needed and mitigating the risk of raw material shortages. The mild reaction conditions also translate to lower energy consumption for heating and cooling, contributing to a smaller carbon footprint and reduced utility costs over the lifecycle of the product. These factors combine to create a more resilient supply chain capable of withstanding market fluctuations and regulatory pressures. The ability to source high-purity intermediates through a greener, more efficient pathway positions companies to meet the increasingly stringent sustainability criteria imposed by global healthcare customers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the reaction system and the reduction in downstream processing complexity. By avoiding the formation of gluconic acid, the need for stoichiometric amounts of base for neutralization is completely eliminated, which reduces both material costs and the volume of inorganic salt waste generated. Furthermore, the use of isopropyl alcohol as a cheap and readily available hydrogen donor replaces more expensive cofactor regeneration substrates, lowering the variable cost per kilogram of product. The high conversion efficiency reported in the patent means that less substrate is wasted, improving the overall atom economy of the synthesis. These cumulative savings allow for a more competitive pricing structure without sacrificing margin, providing a distinct advantage in price-sensitive generic drug markets. The removal of complex purification steps further reduces labor and equipment time, accelerating the overall production cycle.
- Enhanced Supply Chain Reliability: From a logistics perspective, the stability of the lyophilized enzyme powder and the commonality of the reagents used enhance the robustness of the supply chain. Isopropyl alcohol and phosphate buffers are commodity chemicals with established global supply networks, reducing the risk of disruption compared to specialty reagents required for chemical synthesis. The mild storage and transport requirements for the biocatalyst simplify inventory management and reduce the risk of degradation during transit. Additionally, the scalability demonstrated in the patent, moving seamlessly from gram to hectogram scales, indicates a low technical risk for capacity expansion. This predictability allows supply chain planners to forecast production timelines with greater accuracy, ensuring consistent availability of the intermediate for downstream API synthesis. The reduced hazard profile also simplifies regulatory compliance for shipping and storage, further smoothing logistical operations.
- Scalability and Environmental Compliance: The environmental advantages of this technology are substantial, aligning with global initiatives to reduce the ecological impact of pharmaceutical manufacturing. The aqueous nature of the reaction minimizes the release of volatile organic compounds into the atmosphere, improving workplace air quality and reducing the load on abatement systems. The absence of heavy metals and toxic reducing agents simplifies wastewater treatment, allowing for easier compliance with environmental discharge limits. As production scales up, these benefits become increasingly pronounced, making the process suitable for multi-ton annual production capacities without proportional increases in environmental liability. The energy efficiency of running reactions at near-ambient temperatures further contributes to sustainability goals. For organizations committed to corporate social responsibility, adopting this green chemistry approach enhances brand reputation and meets the expectations of environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic process, derived directly from the patent specifications and industry best practices. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios. The answers provided reflect the specific advantages of the recombinant ketoreductase system over alternative methods, highlighting its suitability for high-volume production. These insights are intended to clarify the operational benefits and assist in decision-making processes related to process development and vendor selection. Comprehensive technical support is available to address any specific customization requirements.
Q: How does this biocatalytic method improve upon traditional chemical reduction?
A: Unlike chemical methods requiring hazardous boranes at cryogenic temperatures (-70°C), this patented biological process operates under mild conditions (30-45°C) in aqueous media, significantly enhancing operational safety and environmental compliance.
Q: What is the advantage of using isopropyl alcohol over glucose for cofactor regeneration?
A: Using isopropyl alcohol eliminates the formation of gluconic acid byproducts associated with glucose dehydrogenase systems. This avoids the need for equivalent alkali neutralization, thereby simplifying pH control and downstream purification while protecting enzyme activity.
Q: What purity levels can be achieved with this recombinant ketoreductase route?
A: The patent data demonstrates that this optimized route consistently achieves transformation efficiencies greater than 99% and product stereochemical purity (d.e.) exceeding 99%, meeting the stringent specifications required for high-value API intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable tert-Butyl(3R,5S)-6-chloro-3,5-dihydroxyhexanoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing a stable and high-quality supply of critical statin intermediates for the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of tert-butyl(3R,5S)-6-chloro-3,5-dihydroxyhexanoate meets the exacting standards required for API synthesis. Our commitment to technological excellence means we can leverage advanced biocatalytic methods like the one described in CN102965403A to offer superior products with consistent quality. Partnering with us provides access to deep technical expertise and a robust manufacturing infrastructure designed to support long-term growth and innovation in the healthcare sector.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits of switching to this biocatalytic platform for your supply chain. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments that demonstrate our capability to deliver on our promises. Our goal is to establish a collaborative relationship that drives value through efficiency, quality, and reliability. Let us help you optimize your production strategy and secure a competitive edge in the marketplace through superior chemical manufacturing solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
