Advanced Synthesis of 2-Methoxyallyl Sulfonate for Scalable Pharmaceutical Intermediate Production
Advanced Synthesis of 2-Methoxyallyl Sulfonate for Scalable Pharmaceutical Intermediate Production
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for safer, more efficient, and scalable synthetic routes. A significant breakthrough in this domain is detailed in patent CN114163364A, which introduces a novel compound, 2-methoxyallyl sulfonate, and its robust synthesis method. This innovation addresses critical bottlenecks in the production of acetylmethyl-functionalized drug precursors, which are ubiquitous in high-value therapeutic agents. The acetylmethyl group serves as a vital precursor for constructing five-membered ring backbones, a structural motif found in essential medications such as Warfarin sodium, the antimalarial agent febrifugine, and the anticancer natural product claulansine E. By providing a stable and reactive alternative to traditional reagents, this technology offers a strategic advantage for reliable pharma intermediates suppliers aiming to optimize their production pipelines.
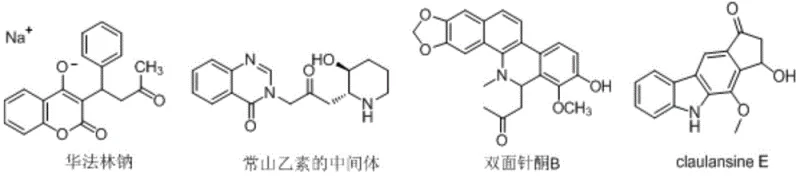
The significance of this patent extends beyond mere chemical novelty; it represents a paradigm shift in how complex heterocyclic systems are assembled. Traditional methods often rely on reagents that are either too reactive to control or too unstable to store, leading to supply chain disruptions and safety hazards. The introduction of 2-methoxyallyl sulfonate provides a solution that balances reactivity with stability, enabling the efficient synthesis of diverse drug intermediates. For R&D directors and process chemists, understanding the nuances of this new reagent is crucial for designing next-generation synthetic routes that are both economically viable and environmentally sustainable.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of acetylmethyl groups into organic frameworks has been fraught with challenges, primarily due to the limitations of available reagents. Haloacetones, while common, are restricted in their utility as they can only react with weaker nucleophilic reagents, such as active methylene groups or heteroatoms like oxygen. This limitation severely restricts the structural diversity accessible to medicinal chemists. Furthermore, the synthesis of 2-methoxyallyl bromide, a more versatile reagent capable of reacting with stronger nucleophiles like carbon ortho to carbonyl groups, involves extremely harsh conditions. The legacy method reported by Jacobson in 1977 requires temperatures as high as 190°C, resulting in a product mixture where the desired 2-methoxyallyl bromide constitutes only 60-70% of the content. Moreover, this bromide analog suffers from poor stability and exhibits strong irritation to the eyes, posing significant occupational health and safety risks in a manufacturing environment.
The Novel Approach
In stark contrast to these legacy methods, the novel approach described in patent CN114163364A utilizes 2-methoxyallyl sulfonate, a compound that combines high reactivity with exceptional stability. This new reagent is synthesized under remarkably mild conditions, typically between -40°C and 40°C, eliminating the need for energy-intensive high-temperature processes. The resulting sulfonate ester is a colorless transparent liquid that can be stored for long periods without degradation, solving the stability issues associated with its bromide counterpart. Crucially, this compound retains the ability to undergo nucleophilic substitution with stronger nucleophiles, thereby unlocking synthetic pathways that were previously difficult or dangerous to execute. The structural integrity and safety profile of this new compound make it an ideal candidate for high-purity pharmaceutical intermediates production.
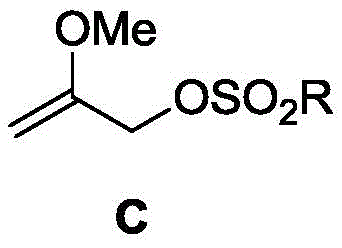
Mechanistic Insights into Sulfonate Ester Formation
The synthesis of 2-methoxyallyl sulfonate relies on a classic yet highly optimized nucleophilic substitution mechanism, specifically the esterification of an alcohol with a sulfonyl chloride. In this reaction, 2-methoxyallyl alcohol acts as the nucleophile, attacking the sulfur atom of the sulfonyl chloride (such as p-toluenesulfonyl chloride) in the presence of a base. The base, which can range from organic amines like triethylamine to inorganic carbonates like potassium carbonate, serves to scavenge the hydrochloric acid byproduct, driving the equilibrium towards the formation of the sulfonate ester. The choice of solvent plays a pivotal role in this mechanism; polar aprotic solvents like tetrahydrofuran (THF) or dichloromethane (DCM) are preferred as they effectively solvate the reactants while maintaining the reactivity of the nucleophile. The reaction proceeds smoothly at temperatures ranging from -10°C to 40°C, indicating a low activation energy barrier compared to the radical or high-thermal processes required for halide synthesis.
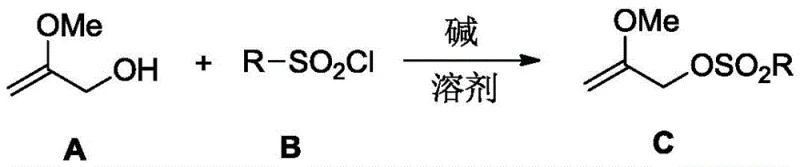
From an impurity control perspective, this mechanism offers distinct advantages. The byproduct of the reaction is typically a salt (e.g., triethylamine hydrochloride), which is easily removed during the aqueous workup phase. The patent specifies quenching with saturated sodium bicarbonate followed by extraction with ethyl acetate, a standard protocol that ensures high purity of the final organic layer. Unlike the bromide synthesis which yields complex mixtures requiring difficult separation, the sulfonate formation is clean and selective. The sulfonate group itself is an excellent leaving group, superior to halides in many contexts, which facilitates the subsequent downstream reactions where the acetylmethyl group is introduced. This mechanistic clarity allows process chemists to predict and control side reactions effectively, ensuring consistent quality in commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 2-Methoxyallyl Sulfonate Efficiently
The synthesis protocol outlined in the patent provides a straightforward, three-step operational procedure that is amenable to both laboratory and industrial scales. The process begins with the dissolution of the starting materials in an anhydrous solvent, followed by the controlled addition of a base to initiate the reaction. Monitoring is achieved via thin-layer chromatography (TLC), ensuring that the reaction is stopped precisely upon the disappearance of the starting material to maximize yield and minimize decomposition. The workup involves standard liquid-liquid extraction and drying techniques, culminating in the recovery of the product via reduced pressure distillation. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized guide below.
- Mix 2-methoxyallyl alcohol and sulfonyl chloride in an organic solvent such as tetrahydrofuran or dichloromethane.
- Add a base like triethylamine or potassium carbonate and react at temperatures between -40°C to 40°C for 8 to 12 hours.
- Quench the reaction with saturated sodium bicarbonate, extract with ethyl acetate, dry over anhydrous sodium sulfate, and concentrate to obtain the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of 2-methoxyallyl sulfonate synthesis represents a significant opportunity to optimize costs and mitigate risks. The transition from hazardous, unstable reagents to this stable sulfonate ester simplifies logistics and storage requirements. Traditional reagents like 2-methoxyallyl bromide often require specialized handling and have short shelf lives, leading to potential waste and supply interruptions. In contrast, the new compound can be produced in large batches and stored indefinitely, allowing for better inventory management and reducing lead time for high-purity pharmaceutical intermediates. Furthermore, the use of commodity chemicals such as sulfonyl chlorides and simple alcohols ensures a robust raw material supply chain that is less susceptible to market volatility.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of extreme processing conditions and expensive purification steps. By operating at near-ambient temperatures (-40°C to 40°C), the process significantly reduces energy consumption compared to the 190°C required for bromide synthesis. Additionally, the high yields reported in the patent examples (ranging from roughly 86% to 95%) indicate a highly efficient atom economy, minimizing raw material waste. The absence of transition metal catalysts or exotic reagents further lowers the direct material costs, contributing to substantial cost savings in pharmaceutical manufacturing without compromising on quality or safety standards.
- Enhanced Supply Chain Reliability: Stability is a key factor in supply chain resilience. The ability to store 2-methoxyallyl sulfonate for long periods means that manufacturers can build strategic stockpiles to buffer against demand fluctuations or raw material shortages. The synthesis uses widely available solvents like THF, DCM, and toluene, which are staples in the fine chemical industry, ensuring that production is not bottlenecked by niche reagent availability. This reliability is crucial for maintaining continuous production schedules for critical drug intermediates, ensuring that downstream API manufacturing is never delayed due to intermediate shortages.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly superior to conventional methods. The mild reaction conditions reduce the risk of thermal runaways, enhancing process safety on a large scale. The workup procedure generates aqueous waste streams that are relatively easy to treat, and the organic solvents used are recoverable and recyclable. By avoiding the generation of irritating vapors associated with alkyl bromides, the process improves workplace safety and reduces the need for extensive personal protective equipment (PPE) and ventilation infrastructure. These factors collectively facilitate easier regulatory approval and smoother commercial scale-up from pilot plants to multi-ton production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 2-methoxyallyl sulfonate. These insights are derived directly from the technical specifications and experimental data provided in patent CN114163364A. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing manufacturing workflows.
Q: What are the advantages of 2-methoxyallyl sulfonate over 2-methoxyallyl bromide?
A: Unlike 2-methoxyallyl bromide, which requires harsh conditions (190°C) and is unstable and irritating, 2-methoxyallyl sulfonate is synthesized under mild conditions (-40 to 40°C), is stable for long-term storage, and poses significantly less safety risk to operators.
Q: What is the primary application of this compound in drug synthesis?
A: It serves as a superior reagent for introducing acetylmethyl groups into organic molecules, facilitating the construction of five-membered ring backbones found in drugs like Warfarin sodium and antimalarial agents.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process uses cheap, readily available reagents and simple workup procedures (extraction and distillation), making it highly suitable for commercial scale-up from kilograms to tons without complex purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methoxyallyl Sulfonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced intermediate synthesis technologies like the one described in CN114163364A. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust industrial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We operate rigorous QC labs equipped with state-of-the-art analytical instruments to guarantee stringent purity specifications for every batch of 2-methoxyallyl sulfonate we produce, adhering to the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this cost-effective and safe synthesis route for your drug development projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you secure a reliable supply of high-quality intermediates that drive your innovation forward.
