Advanced Synthetic Route for 1,3-Oxazine Fluorides Enabling Commercial Scale-Up
The landscape of modern medicinal chemistry is increasingly defined by the strategic incorporation of fluorine atoms into bioactive scaffolds, a trend driven by the unique ability of fluorine to modulate metabolic stability, lipophilicity, and binding affinity. In this context, the development of efficient methodologies for constructing fluorinated heterocycles is paramount for the rapid advancement of drug discovery pipelines. Patent CN111574471B introduces a significant breakthrough in this domain by disclosing a novel synthetic method for 1,3-oxazine fluorides, a privileged structure found in various biologically active molecules including BACE-1 inhibitors. This technology leverages a tandem cyclization-fluorination strategy starting from homoallyl imidates, offering a distinct advantage over traditional multi-step condensations. For global pharmaceutical manufacturers, this innovation represents not just a chemical curiosity but a viable pathway to streamline the production of complex intermediates, potentially reducing the overall cost of goods and accelerating time-to-market for new therapeutic candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of fluorinated 1,3-oxazine cores has relied heavily on the condensation of pre-functionalized fluorinated precursors with amino-alcohol derivatives. These conventional pathways are often plagued by significant logistical and economic hurdles, primarily stemming from the high cost and limited commercial availability of specialized fluorinated building blocks. Furthermore, these multi-step sequences frequently necessitate harsh reaction conditions, such as strong acids or bases, which can lead to poor atom economy and the generation of complex waste streams that are difficult to treat. The reliance on protecting group strategies in these older methods further elongates the synthetic timeline, introducing additional unit operations that increase both capital expenditure and operational complexity. From a supply chain perspective, the dependency on niche fluorinated starting materials creates vulnerability, as any disruption in the supply of these specific reagents can halt entire production lines, making the conventional approach less attractive for large-scale commercial manufacturing.
The Novel Approach
In stark contrast, the methodology described in the patent utilizes a direct, one-pot transformation of readily accessible homoallyl imidates into the desired fluorinated heterocycles. This approach fundamentally shifts the paradigm by installing the fluorine atom concurrently with the ring closure, effectively merging two synthetic steps into a single operation. The use of commercially abundant electrophilic fluorinating agents, such as Selectfluor, allows for the activation of the alkene moiety, triggering an intramolecular nucleophilic attack by the imidate nitrogen. This cascade process not only simplifies the operational workflow but also significantly enhances the overall yield by minimizing intermediate isolation losses. The versatility of this method is underscored by its tolerance for a wide range of substituents on both the aromatic ring and the alkene terminus, allowing for the rapid generation of diverse libraries of analogs for structure-activity relationship studies without the need for extensive process re-optimization.
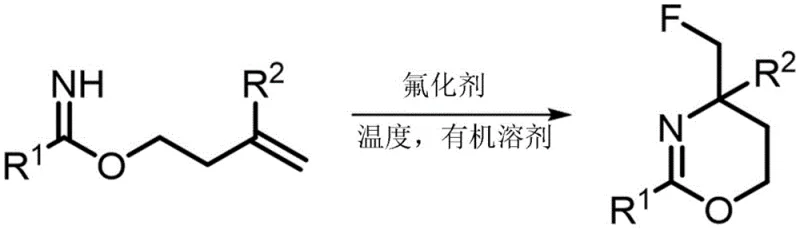
Mechanistic Insights into Electrophilic Fluorination-Induced Cyclization
The core of this technological advancement lies in the elegant mechanism of electrophilic fluorination-induced cyclization, which proceeds through a highly organized transition state. Upon addition of the fluorinating agent to the reaction mixture, the electron-rich double bond of the homoallyl imidate undergoes electrophilic attack, generating a transient fluoronium ion intermediate. This cationic species is immediately intercepted by the neighboring imidate nitrogen lone pair, driving the formation of the six-membered 1,3-oxazine ring with high regioselectivity. The reaction kinetics are finely tuned by the choice of solvent and temperature, ensuring that the cyclization outcompetes potential side reactions such as polymerization or elimination. Understanding this mechanistic pathway is crucial for R&D directors aiming to optimize the process further, as it highlights the importance of maintaining precise stoichiometric control to prevent over-fluorination or decomposition of the sensitive intermediates.
From an impurity control perspective, this mechanism offers inherent advantages by limiting the formation of regioisomers that are common in stepwise syntheses. The intramolecular nature of the ring-closing step imposes geometric constraints that favor the formation of the thermodynamically stable 1,3-oxazine scaffold over alternative cyclic structures. Moreover, the use of mild organic solvents like acetonitrile helps to solubilize both the organic substrate and the ionic fluorinating salt, creating a homogeneous reaction environment that promotes consistent heat and mass transfer. This homogeneity is critical for scaling up the reaction, as it prevents the formation of hot spots that could lead to thermal runaway or the generation of colored impurities. Consequently, the resulting crude product typically exhibits a cleaner profile, reducing the burden on downstream purification units and facilitating the attainment of stringent pharmaceutical purity specifications.
How to Synthesize 1,3-Oxazine Fluorides Efficiently
Implementing this synthesis in a practical setting requires careful attention to reagent quality and reaction parameters to ensure reproducibility and safety. The protocol begins with the dissolution of the homoallyl imidate substrate in a dry, aprotic solvent, with acetonitrile being the preferred medium due to its ability to stabilize the ionic intermediates. The fluorinating agent, preferably Selectfluor, is then added sequentially to control the exotherm associated with the initial electrophilic attack. As illustrated in the specific embodiment below, the reaction is typically conducted at elevated temperatures to overcome the activation energy barrier for cyclization.
- Dissolve the homoallyl imidate substrate in anhydrous acetonitrile under inert atmosphere.
- Add Selectfluor (1-chloromethyl-4-fluoro-1,4-diazoniabicyclo[2.2.2]octane bis(tetrafluoroborate)) sequentially to the reaction mixture.
- Heat the mixture to 60-80°C for approximately 12 hours, then proceed with standard aqueous workup and purification.
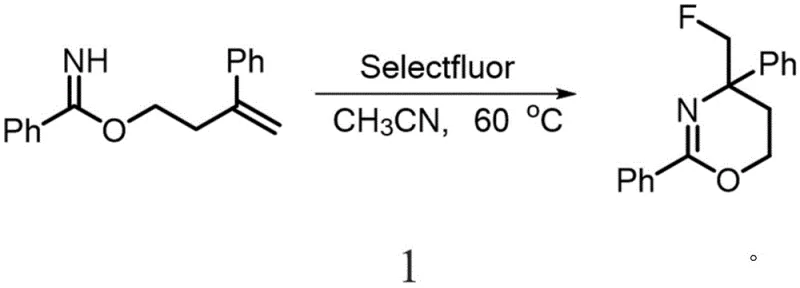
The detailed standardized synthesis steps for this process are outlined in the guide below, providing a clear roadmap for laboratory and pilot plant execution.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond simple yield improvements. By shifting the synthesis to rely on bulk commodity chemicals like homoallyl alcohols and amines rather than exotic fluorinated precursors, the raw material cost base is significantly lowered. This substitution effect decouples the production cost from the volatile pricing of specialized fluorine sources, providing greater budget predictability for long-term manufacturing contracts. Furthermore, the simplification of the process flow reduces the number of required reactors and purification columns, directly translating to lower capital intensity and reduced utility consumption per kilogram of product produced.
- Cost Reduction in Manufacturing: The elimination of pre-fluorinated starting materials removes a major cost driver from the bill of materials, as these specialty reagents often carry substantial price premiums due to complex manufacturing processes. Additionally, the one-pot nature of the reaction reduces solvent usage and waste disposal costs, as fewer workup and isolation steps are required compared to linear synthetic routes. The high atom economy of the cyclization ensures that a larger proportion of the input mass is converted into the final valuable product, minimizing the financial loss associated with byproduct formation. Overall, these factors combine to create a leaner, more cost-effective manufacturing process that enhances the margin potential for the final API.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly simplified because the key starting materials, homoallyl imidates, can be synthesized from widely available allylic alcohols and nitriles which are produced on a massive global scale. This abundance ensures that supply disruptions are unlikely, providing a secure foundation for continuous manufacturing operations. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the risk of batch failures and the need for costly re-processing. Consequently, manufacturers can maintain tighter delivery schedules and offer more reliable lead times to their downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The use of acetonitrile, a solvent with well-established recovery and recycling protocols, aligns well with modern green chemistry initiatives and environmental regulations. The moderate temperature range of 10-120°C avoids the need for energy-intensive cryogenic cooling or high-pressure equipment, making the process easier to scale from kilogram to multi-ton quantities. The reduction in reaction steps inherently lowers the E-factor (mass of waste per mass of product), facilitating compliance with increasingly strict environmental discharge limits. This scalability ensures that the technology can grow with the demand of the drug, supporting seamless transitions from clinical trial material to commercial launch volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorination technology, derived directly from the patent specifications and practical application scenarios. These insights are intended to clarify the operational feasibility and strategic value of adopting this route for your specific project needs.
Q: What are the primary advantages of this fluorination method over traditional condensation routes?
A: This method utilizes a direct cyclization-fluorination cascade on readily available homoallyl imidates, eliminating the need for pre-fluorinated building blocks which are often expensive and difficult to source, thereby simplifying the synthetic sequence.
Q: Is the process scalable for industrial production of API intermediates?
A: Yes, the reaction employs common organic solvents like acetonitrile and operates at moderate temperatures (60-80°C), avoiding cryogenic conditions or hazardous reagents, which facilitates safe scale-up from laboratory to multi-ton production.
Q: What is the typical yield and purity profile for this synthesis?
A: Exemplified embodiments demonstrate isolated yields around 82% with high structural fidelity, producing colorless liquids that require standard purification techniques, indicating a robust impurity profile suitable for downstream pharmaceutical processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Oxazine Fluoride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and optimizing production costs. Our team of expert chemists has thoroughly analyzed the potential of this patent-protected route and is fully prepared to translate this laboratory innovation into a robust commercial process. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,3-oxazine fluoride delivered meets the highest industry standards for pharmaceutical intermediates.
We invite you to engage with our technical procurement team to discuss how this technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and our proven track record of excellence in fine chemical manufacturing.
