Advanced Biocatalytic Synthesis of Chiral Statin Intermediates for Commercial Scale
Advanced Biocatalytic Synthesis of Chiral Statin Intermediates for Commercial Scale
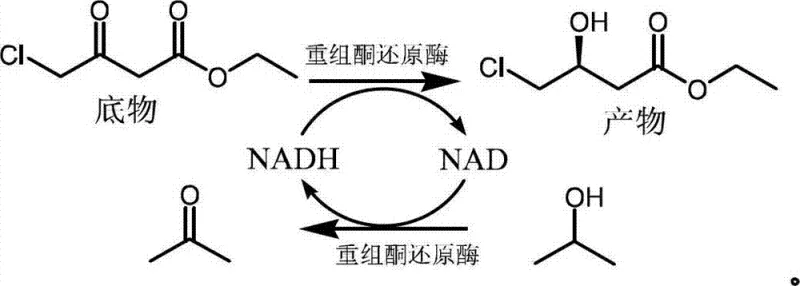
The pharmaceutical industry's relentless pursuit of efficient, sustainable, and cost-effective manufacturing routes for high-value active pharmaceutical ingredients (APIs) has placed biocatalysis at the forefront of process innovation. Specifically, the synthesis of chiral building blocks for HMG-CoA reductase inhibitors, commonly known as statins, represents a critical challenge where stereochemical purity dictates therapeutic efficacy. Patent CN102925501A introduces a transformative biological preparation method for (S)-4-chloro-3-hydroxybutyrate ethyl, a pivotal chiral intermediate in the statin value chain. This technology leverages a specialized recombinant ketoreductase to facilitate asymmetric reduction under mild conditions, effectively addressing historical bottlenecks related to cofactor regeneration and substrate loading. By integrating this advanced enzymatic approach, manufacturers can achieve transformation efficiencies exceeding 99% with exceptional optical purity, positioning this method as a superior alternative to traditional chemical reduction or earlier generation biocatalytic processes that suffered from low efficiency and complex operational requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of (S)-4-chloro-3-hydroxybutyrate ethyl has relied heavily on chemical reduction methods using agents like sodium borohydride or metal-catalyzed hydrogenation, which often result in racemic mixtures requiring costly and yield-loss-inducing chiral resolution steps. Even within the realm of biocatalysis, prior art methods disclosed in various patent applications have struggled with significant inefficiencies, particularly regarding cofactor dependency and substrate tolerance. Traditional enzymatic routes frequently necessitate the addition of glucose as a hydrogen donor coupled with a second enzyme system for cofactor regeneration, which complicates the reaction matrix, increases the burden on downstream purification, and inflates raw material costs. Furthermore, these legacy biological processes typically operate at low substrate concentrations, often below 30%, to avoid enzyme inhibition, which drastically reduces volumetric productivity and necessitates larger reactor volumes to achieve the same output, thereby negatively impacting the capital expenditure and operational efficiency of the manufacturing facility.
The Novel Approach
The innovative methodology described in the patent data overcomes these entrenched limitations by employing a robust recombinant ketoreductase capable of utilizing isopropanol directly as a hydrogen donor, effectively eliminating the need for auxiliary glucose-dehydrogenase systems. This strategic shift simplifies the reaction components significantly, allowing for a streamlined process where the enzyme catalyzes both the reduction of the ketone substrate and the regeneration of the essential NADPH cofactor through the oxidation of isopropanol to acetone. Crucially, this novel approach enables the operation of the reaction in a biphasic system comprising an aqueous buffer and toluene, which dramatically improves the solubility of the organic substrate and mitigates potential toxicity issues. This architectural change in the process design permits substrate concentrations to be increased substantially, up to 40% (w/v), which represents a massive leap in space-time yield compared to conventional methods, ultimately driving down the unit cost of production while maintaining the rigorous stereochemical control required for pharmaceutical applications.
Mechanistic Insights into Recombinant Ketoreductase-Catalyzed Asymmetric Reduction
At the heart of this high-efficiency synthesis lies the precise mechanistic action of the recombinant ketoreductase, which facilitates the stereoselective transfer of a hydride ion from the reduced cofactor NADPH to the prochiral carbonyl carbon of the 4-chloro-3-keto ester substrate. The enzyme's active site is engineered to strictly orient the substrate, ensuring that hydride delivery occurs exclusively from one face of the planar carbonyl group, thereby guaranteeing the formation of the desired (S)-enantiomer with high fidelity. This biocatalytic cycle is sustained by the continuous regeneration of NADPH from NADP+, a process driven by the concurrent oxidation of isopropanol, which serves as a sacrificial reductant. The thermodynamic equilibrium of this coupled reaction is favorably shifted towards product formation by the removal or volatility of the acetone byproduct, ensuring that the reaction proceeds to near-completion without the accumulation of inhibitory intermediates. The use of a biphasic solvent system further stabilizes the enzyme structure and modulates the local concentration of the hydrophobic substrate at the interface, optimizing the kinetic parameters for maximal turnover frequency.
Beyond the primary reduction event, the impurity profile of the final product is meticulously controlled through the inherent specificity of the biocatalyst, which exhibits negligible activity towards side reactions such as dehalogenation or over-reduction that are common pitfalls in chemical catalysis. The mild pH range of 7.0 to 9.0 and moderate temperature conditions (25°C to 45°C) preserve the integrity of the sensitive chloro-ester functionality, preventing hydrolysis or elimination reactions that could generate difficult-to-remove impurities. Furthermore, the biphasic extraction capability inherent in the toluene-water system allows for the immediate partitioning of the product into the organic phase as it forms, effectively protecting it from any potential reverse reactions or enzymatic degradation. This dual function of the solvent system acts as an in-situ purification step, ensuring that the crude product stream is already of high purity before formal work-up, significantly reducing the load on subsequent crystallization or distillation units and enhancing the overall robustness of the manufacturing process.
How to Synthesize (S)-4-chloro-3-hydroxybutyrate ethyl Efficiently
The implementation of this biocatalytic route requires careful attention to the preparation of the biphasic reaction medium and the sequential addition of reagents to maximize enzyme stability and activity. The process begins with the formulation of a phosphate buffer system adjusted to a physiological pH, which is then mixed with toluene to create the necessary organic-aqueous interface for substrate solubilization. Following the establishment of the reaction environment, the substrate, hydrogen donor, and biocatalyst are introduced under controlled agitation to ensure efficient mass transfer between phases. For a comprehensive understanding of the precise operational parameters, including specific mixing rates, induction protocols for enzyme expression, and detailed work-up procedures, please refer to the standardized synthesis guide provided below.
- Prepare a biphasic reaction system consisting of phosphate buffer (pH 7.0-9.0) and toluene, ensuring optimal solubility for the substrate.
- Introduce the substrate 4-chloro-3-carbonyl ethyl butyrate at high concentrations (up to 40% w/v) along with isopropanol as the hydrogen donor.
- Add recombinant ketoreductase and cofactor NADP+, maintaining temperature at 30°C until conversion exceeds 99%.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement and supply chain perspective, the adoption of this patented biocatalytic technology offers profound advantages that extend far beyond simple yield improvements, fundamentally altering the cost structure and reliability of the supply base for statin intermediates. The elimination of the glucose-cofactor regeneration system removes a significant variable cost component, as isopropanol is a commodity chemical with stable pricing and widespread availability compared to specialized enzymatic substrates. Moreover, the ability to run reactions at significantly higher substrate concentrations translates directly into reduced solvent usage and smaller reactor footprints for the same production volume, leading to substantial savings in utility consumption and waste treatment costs. These operational efficiencies compound to create a more resilient supply chain that is less susceptible to raw material volatility and better equipped to handle sudden surges in demand without requiring proportional capital investment in new infrastructure.
- Cost Reduction in Manufacturing: The transition to an isopropanol-driven cofactor regeneration system drastically simplifies the bill of materials by removing the need for expensive glucose and secondary enzymes, resulting in a leaner and more cost-effective production process. Additionally, the high substrate loading capacity minimizes the volume of water and buffer required per kilogram of product, which significantly lowers the energy costs associated with heating, cooling, and wastewater treatment. The simplified downstream processing, facilitated by the biphasic system, further reduces the consumption of extraction solvents and purification media, contributing to a lower overall cost of goods sold (COGS) and improved margin potential for the final API.
- Enhanced Supply Chain Reliability: By relying on robust, commercially available reagents like isopropanol and toluene rather than specialized biological feedstocks, manufacturers can mitigate the risk of supply disruptions and ensure consistent production scheduling. The high conversion rates and optical purity achieved in a single step reduce the dependency on complex multi-step synthetic sequences, shortening the overall manufacturing lead time and allowing for more agile response to market fluctuations. This process stability ensures a steady flow of high-quality intermediate, securing the upstream supply for statin production and reducing the likelihood of batch failures that could jeopardize downstream formulation timelines.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous-based nature of the biocatalytic step align perfectly with green chemistry principles, reducing the environmental footprint associated with hazardous waste generation and volatile organic compound emissions. The process is inherently scalable from laboratory benchtop to industrial multi-ton reactors without significant re-optimization, allowing for seamless capacity expansion as market demand grows. Furthermore, the high selectivity of the enzyme minimizes the formation of toxic byproducts, simplifying regulatory compliance and reducing the costs associated with environmental health and safety (EHS) management and waste disposal.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this technology into their existing manufacturing portfolios, we have compiled a set of frequently asked questions based on the specific technical disclosures and experimental data found within the patent literature. These inquiries address critical aspects regarding process robustness, impurity control, and the practical implications of scaling this biocatalytic route for commercial production. The answers provided below are derived directly from the documented embodiments and comparative examples, ensuring accuracy and relevance for your technical due diligence.
Q: What is the primary advantage of using isopropanol over glucose in this biocatalytic process?
A: Using isopropanol eliminates the need for a secondary enzyme system (like glucose dehydrogenase) and expensive glucose substrates, significantly simplifying the reaction mixture and reducing raw material costs while driving the equilibrium forward through acetone formation.
Q: How does the biphasic toluene-water system improve reaction efficiency?
A: The biphasic system enhances the solubility of the hydrophobic substrate and product, reduces substrate inhibition at high concentrations, and facilitates easier downstream extraction, thereby improving overall space-time yield.
Q: What optical purity can be achieved with this recombinant ketoreductase method?
A: The patented method consistently achieves an optical purity greater than 99% ee, meeting the stringent quality requirements for downstream statin API synthesis without the need for complex chiral resolution steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-4-chloro-3-hydroxybutyrate ethyl Supplier
As the global demand for high-purity statin intermediates continues to rise, partnering with a technically proficient CDMO is essential for securing a competitive edge in the pharmaceutical market. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and compliant with international quality standards. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of (S)-4-chloro-3-hydroxybutyrate ethyl meets the exacting requirements for chirality and chemical purity necessary for downstream API synthesis.
We invite you to engage with our technical procurement team to discuss how this advanced biocatalytic technology can be tailored to your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this enzymatic route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both the quality and cost-efficiency of your statin manufacturing operations.
