Advanced Biocatalytic Synthesis of Statin Intermediates: Scalable Production & Cost Efficiency
The pharmaceutical industry's relentless pursuit of efficient statin synthesis has found a significant breakthrough in patent CN102978249A, which discloses a novel biological preparation method for 6-cyano-(3R, 5R)-dihydroxy-hexanoic acid tert-butyl ester. This compound serves as a pivotal chiral intermediate in the manufacturing of blockbuster cholesterol-lowering drugs like atorvastatin. Traditional synthetic routes have long been plagued by harsh reaction conditions, hazardous reagents, and complex purification steps that inflate production costs and environmental burdens. This patent introduces a streamlined biocatalytic approach that leverages a specific recombinant ketoreductase (KRED) to drive the asymmetric reduction of the keto-ester substrate. By integrating substrate reduction and cofactor regeneration into a single enzymatic step, the technology offers a robust pathway for reliable pharmaceutical intermediates supplier networks seeking to modernize their production capabilities. The shift from chemical catalysis to this advanced bio-engineered solution represents a paradigm shift in green chemistry, promising not only superior stereocontrol but also a drastic simplification of the overall manufacturing workflow.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this critical statin intermediate has relied heavily on chemical reduction methods or less optimized enzymatic processes that impose severe operational constraints. For instance, prior chemical arts, such as those disclosed in US Patent No. 2009/0216029 A1, necessitate the use of borane compounds under extreme cryogenic conditions below -70°C. These requirements demand specialized, energy-intensive equipment and pose significant safety risks due to the flammability and toxicity of the reagents involved. Furthermore, even under these rigorous conditions, the stereoisomeric purity often struggles to exceed 98%, necessitating additional recrystallization steps that erode overall yield. Similarly, earlier biocatalytic attempts, like the Codexis route (WO 2008/042876 A2), while avoiding harsh chemicals, introduced new complexities by requiring a dual-enzyme system. This approach utilized both a ketoreductase and a glucose dehydrogenase, leading to the accumulation of gluconic acid byproducts. The presence of this acidic byproduct complicates pH regulation, requiring the continuous addition of alkaline solutions, and creates substantial difficulties in downstream processing and impurity removal, ultimately hindering cost reduction in API manufacturing.
The Novel Approach
The methodology presented in CN102978249A fundamentally resolves these bottlenecks by employing a singular, highly efficient recombinant ketoreductase capable of managing the entire redox cycle independently. This innovative strategy eliminates the need for a secondary enzyme and its associated substrates, thereby removing the generation of non-volatile acidic byproducts like gluconic acid. The reaction proceeds smoothly in an aqueous phase buffering liquid at a neutral pH of 6.5 to 7.5, under normal temperature and pressure, creating a much safer and more controllable environment. Crucially, the system utilizes isopropyl alcohol as a hydrogen donor, which is oxidized to acetone during the cofactor regeneration process. Unlike gluconic acid, acetone is highly volatile and can be easily removed from the reaction system through simple evaporation or extraction, greatly simplifying the post-processing work. This elegance in design allows for the production of high-purity OLED material precursors or pharmaceutical intermediates with minimal waste, marking a significant advancement over the cumbersome legacy technologies.
Mechanistic Insights into KRED-Catalyzed Asymmetric Reduction
At the heart of this process lies the sophisticated mechanism of the recombinant ketoreductase (KRED), which exhibits exceptional stereoselectivity for the (3R, 5R) configuration. The enzyme facilitates the hydride transfer from the reduced cofactor (NADPH) to the prochiral ketone group of the substrate, 6-cyano-(5R)-hydroxyl-3-oxo hecanoic acid t-butyl ester. What distinguishes this specific patent implementation is the ingenious coupling of the reduction reaction with an in-situ cofactor regeneration cycle driven by isopropyl alcohol. As the KRED reduces the substrate to the desired diol product, the oxidized cofactor (NADP+) is immediately regenerated back to its active form (NADPH) by oxidizing the isopropyl alcohol. This creates a closed catalytic loop that requires only catalytic amounts of the expensive cofactor, rather than stoichiometric quantities, driving down material costs significantly.
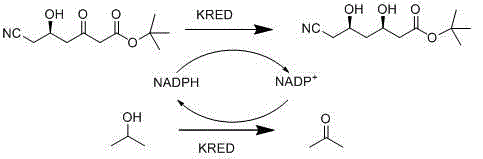
This mechanistic efficiency directly translates to superior impurity control, a critical parameter for R&D directors focused on quality. In previous dual-enzyme systems, the secondary enzymatic reaction produced gluconic acid, which not only shifted the pH but also remained dissolved in the aqueous phase, complicating isolation. In contrast, the byproduct of this novel system is acetone. Due to its high volatility and distinct physical properties compared to the product and substrate, acetone does not interfere with the reaction equilibrium and is effortlessly separated during the workup phase. This clean reaction profile ensures that the final product achieves an optical purity (de value) greater than 99% and a conversion rate exceeding 98%, as validated by LC-MS and HPLC analysis in the patent examples. The absence of difficult-to-remove acidic impurities means fewer purification columns and less solvent consumption, enhancing the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 6-Cyano-(3R,5R)-dihydroxy-hexanoic acid tert-butyl ester Efficiently
The practical implementation of this biocatalytic route is designed for seamless integration into existing manufacturing facilities, requiring only standard reactor setups capable of maintaining mild thermal conditions. The process begins with the preparation of the reaction vessel, where the substrate is suspended in a phosphate-buffered saline solution adjusted to a pH of roughly 7.5. Isopropyl alcohol is added as the hydrogen source, followed by the introduction of the lyophilized recombinant ketoreductase powder and the NADP cofactor. The mixture is then agitated at a controlled temperature between 25°C and 35°C, allowing the enzymatic cascade to proceed without the need for exotic pressure vessels or cryogenic cooling. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction system by suspending the substrate 6-cyano-(5R)-hydroxyl-3-oxo hecanoic acid t-butyl ester in an aqueous phosphate buffer (pH 6.5-7.5) with isopropyl alcohol as the hydrogen donor.
- Introduce the recombinant ketoreductase (KRED) and the cofactor NADP/NADPH into the mixture, maintaining the temperature between 25°C and 35°C.
- Monitor the reaction via LC-MS until conversion exceeds 90%, then extract with ethyl acetate and evaporate the solvent to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this biocatalytic protocol offers tangible strategic benefits that extend far beyond simple yield improvements. The elimination of hazardous borane reagents and the shift away from extreme low-temperature operations significantly reduce the safety compliance burden and insurance costs associated with production. Furthermore, the reliance on a single enzyme system simplifies the raw material supply chain, reducing the number of critical biological components that need to be sourced and qualified. The mild aqueous conditions also mean that standard stainless steel reactors can be utilized without the need for specialized glass-lined or Hastelloy equipment often required for corrosive chemical reductions, representing a substantial capital expenditure saving for manufacturers looking to expand capacity.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the drastic simplification of the downstream processing (DSP) workflow. By replacing the dual-enzyme system with a single KRED, the process avoids the formation of gluconic acid, a non-volatile byproduct that traditionally necessitates complex ion-exchange chromatography or extensive washing steps to remove. Additionally, the byproduct acetone is volatile and easily stripped from the mixture, reducing energy consumption during solvent recovery. The ability to use catalytic rather than stoichiometric amounts of the expensive NADP cofactor further lowers the bill of materials, ensuring that cost reduction in pharmaceutical intermediates manufacturing is achieved through fundamental process intensification rather than mere supplier negotiation.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions—operating at ambient pressure and moderate temperatures—greatly enhances the reliability of production schedules. Unlike chemical routes susceptible to delays due to the handling hazards of pyrophoric reagents or the logistical challenges of maintaining cryogenic supply chains, this bioprocess is inherently safer and more stable. The use of commercially available isopropyl alcohol as a hydrogen donor ensures that key reagents are commoditized and readily accessible globally, mitigating the risk of supply disruptions. This stability allows for more accurate lead time predictions and ensures reducing lead time for high-purity pharmaceutical intermediates becomes a achievable reality for partners.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the aqueous nature of the reaction aligns perfectly with modern green chemistry mandates. The process generates minimal hazardous waste, as the primary organic byproduct (acetone) is easily recovered and potentially recycled. The high conversion rates (>98%) mean that raw material utilization is maximized, reducing the volume of unreacted starting material that must be treated as waste. This efficiency facilitates the commercial scale-up of complex statin intermediates from pilot kilogram batches to multi-ton annual production without encountering the exponential increase in waste treatment costs often seen in traditional chemical synthesis, making it an ideal candidate for sustainable long-term supply contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented biocatalytic technology. These answers are derived directly from the experimental data and claims within patent CN102978249A, providing a factual basis for evaluating the process viability. Understanding these specifics is crucial for technical teams assessing the fit of this route within their existing portfolio of synthetic capabilities.
Q: How does this single-enzyme system improve upon previous dual-enzyme methods?
A: Unlike prior art requiring two enzymes (ketoreductase plus glucose dehydrogenase), this patent utilizes a single recombinant ketoreductase that simultaneously performs substrate reduction and cofactor regeneration. This eliminates the formation of gluconic acid byproducts, thereby simplifying pH control and downstream purification significantly.
Q: What are the specific reaction conditions required for this biocatalytic process?
A: The process operates under mild, environmentally friendly conditions: an aqueous phase buffer at pH 6.5-7.5, normal atmospheric pressure, and temperatures ranging from 25°C to 35°C. It avoids the hazardous reagents and extreme cryogenic temperatures (-70°C) associated with traditional chemical reductions.
Q: How is the optical purity of the final statin intermediate controlled?
A: The specificity of the recombinant ketoreductase ensures high stereoselectivity. Experimental data from the patent indicates that the process consistently achieves a substrate conversion rate greater than 98% and an optical purity (de value) exceeding 99%, meeting stringent requirements for chiral pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Cyano-(3R,5R)-dihydroxy-hexanoic acid tert-butyl ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic route described in CN102978249A for the global statin market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this single-enzyme system are fully realized in large-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 6-cyano-(3R, 5R)-dihydroxy-hexanoic acid tert-butyl ester meets the exacting standards required for GMP pharmaceutical applications. We are committed to leveraging this green chemistry innovation to deliver consistent quality and supply security to our global partners.
We invite forward-thinking pharmaceutical companies to collaborate with us to optimize their supply chains using this advanced technology. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data from our recent pilot runs and to discuss detailed route feasibility assessments for your upcoming projects, ensuring a seamless transition to this superior manufacturing platform.
