Revolutionizing Azaarene Functionalization: A Transition-Metal-Free Route for Commercial Scale-Up
Revolutionizing Azaarene Functionalization: A Transition-Metal-Free Route for Commercial Scale-Up
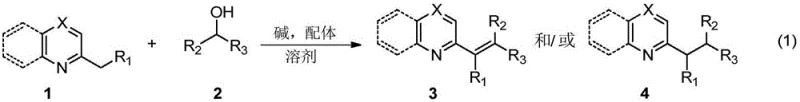
The landscape of organic synthesis for nitrogen-containing heterocycles is undergoing a significant transformation, driven by the urgent need for greener and more cost-effective manufacturing processes. Patent CN111253305A introduces a groundbreaking methodology for the alkenylation or alkylation of alkyl-substituted azaarenes, fundamentally challenging the reliance on precious transition metal catalysts that has dominated the field for decades. This innovation leverages a synergistic system comprising a simple base promoter and a nitrogen or phosphine-containing ligand to activate C(sp3)-H bonds directly using alcohols as coupling reagents. For R&D directors and procurement specialists in the pharmaceutical industry, this represents a paradigm shift towards sustainable chemistry that does not compromise on efficiency or yield. The ability to construct complex carbon-carbon bonds under mild heating conditions without generating stoichiometric heavy metal waste addresses critical pain points in modern API production. As we delve deeper into the technical specifics, it becomes evident that this approach offers a robust platform for the scalable synthesis of high-value fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the functionalization of alkyl chains on nitrogen heterocycles has been fraught with significant chemical and economic challenges that hinder efficient large-scale production. Classical alkylation strategies typically necessitate the pre-functionalization of electrophiles, requiring the use of alkyl halides, allyl carbonates, or ester compounds which generate substantial stoichiometric waste streams. Furthermore, many contemporary C-H activation protocols rely heavily on expensive transition metal catalysts such as ruthenium, iridium, or palladium complexes to facilitate hydrogen transfer strategies. These metals not only inflate the raw material costs but also introduce severe downstream processing burdens, as residual metal levels in pharmaceutical intermediates must be reduced to parts-per-million specifications to meet regulatory safety standards. The removal of these trace metals often requires specialized scavenging resins or complex purification steps, adding time, cost, and environmental toxicity to the overall manufacturing workflow. Consequently, there is a pressing demand for alternative methodologies that can bypass these limitations while maintaining high reaction efficiency and selectivity.
The Novel Approach
The methodology disclosed in the patent data presents an elegant solution by replacing costly transition metals with an accessible base-ligand cooperative system. By utilizing alkali bases such as potassium tert-butoxide or potassium carbonate in conjunction with ligands like 1,10-phenanthroline or bipyridines, the reaction successfully activates the C(sp3)-H bond of the methyl group on the azaarene ring. This approach allows alcohols to serve directly as alkenylating or alkylating agents through a hydrogen borrowing or transfer mechanism that operates effectively under thermal conditions. The elimination of transition metals simplifies the reaction setup and drastically reduces the environmental footprint, aligning perfectly with green chemistry principles. Moreover, the system exhibits remarkable versatility, accommodating a wide range of substrates including quinolines, pyrazines, and pyridines, while tolerating diverse functional groups on the alcohol coupling partners. This flexibility ensures that the process can be adapted for the synthesis of various complex molecular scaffolds required in drug discovery and development without the need for extensive re-optimization.

Mechanistic Insights into Base-Promoted C-H Activation
Understanding the mechanistic underpinnings of this transition-metal-free transformation is crucial for R&D teams aiming to implement this technology in their own laboratories. The reaction likely proceeds through a deprotonation-initiated pathway where the strong base abstracts a proton from the methyl group of the azaarene, generating a reactive carbanion or stabilized intermediate species. The presence of the nitrogen or phosphine ligand plays a pivotal role in stabilizing transient species and facilitating the hydride transfer steps necessary for the coupling with the alcohol substrate. Unlike metal-catalyzed cycles that involve oxidative addition and reductive elimination steps, this organocatalytic-style mechanism relies on the electronic modulation provided by the ligand to lower the activation energy barrier for C-C bond formation. The alcohol undergoes dehydrogenation in situ to form an aldehyde intermediate, which then condenses with the activated azaarene species, followed by dehydration to yield the final alkenylated product. This intricate interplay between the base and ligand ensures high atom economy, as the only byproducts are typically water or hydrogen gas, avoiding the generation of halogenated waste salts common in traditional substitution reactions.
Impurity control is another critical aspect where this mechanism offers distinct advantages over conventional routes. In metal-catalyzed processes, side reactions such as homocoupling of the alcohol or over-reduction of the double bond can occur due to the variable oxidation states of the metal center. However, the base-promoted system described here demonstrates excellent chemoselectivity, primarily yielding the desired alkenylated or alkylated products with minimal byproduct formation. The reaction conditions, particularly temperature and ligand choice, provide a handle to tune the selectivity between the unsaturated alkenyl product and the saturated alkyl product. For instance, lower temperatures tend to favor the retention of the double bond, while higher temperatures and extended reaction times can drive the reaction towards full saturation. This level of control allows process chemists to direct the synthesis towards the specific structural motif required for their target molecule, thereby simplifying purification and improving overall process robustness.
How to Synthesize 2-Styrylquinoline Derivatives Efficiently
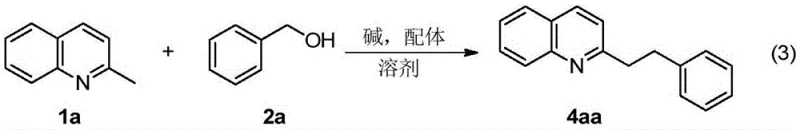
Implementing this novel synthesis route requires careful attention to reaction parameters to maximize yield and selectivity, as detailed in the experimental examples provided in the patent documentation. The standard protocol involves charging a reaction vessel with the alkyl-substituted azaarene substrate and the chosen alcohol coupling partner in a molar ratio that typically ranges from 1:1.5 to 1:4 to ensure complete conversion of the limiting reagent. A strong base, such as potassium tert-butoxide, is added along with a catalytic amount of a chelating ligand like 1,10-phenanthroline, dissolved in a high-boiling solvent such as tert-amyl alcohol or o-xylene. The mixture is then heated under an inert atmosphere or even in air, depending on the specific substrate sensitivity, at temperatures between 80°C and 140°C for a duration of 12 to 48 hours. Detailed standardized synthesis steps for optimizing this transformation are provided in the guide below.
- Prepare the reaction mixture by combining alkyl-substituted azaarenes and alcohol substrates with a strong base promoter such as potassium tert-butoxide in a suitable solvent like tert-amyl alcohol or o-xylene.
- Add a nitrogen-containing or phosphine-containing ligand, such as 1,10-phenanthroline or bipyridine derivatives, to facilitate the C-H activation process without transition metals.
- Heat the reaction mixture under an inert or air atmosphere at temperatures between 50°C and 150°C for 12 to 48 hours to achieve high yields of alkenylated or alkylated products.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this transition-metal-free technology translates into tangible strategic benefits that extend far beyond the laboratory bench. The most immediate impact is seen in the reduction of raw material costs, as the process eliminates the need for purchasing expensive precious metal catalysts which are subject to volatile market pricing and supply constraints. Furthermore, the simplicity of the workup procedure, which often involves basic filtration and solvent removal without the need for specialized metal scavengers, significantly lowers the operational expenditure associated with downstream processing. This streamlined workflow not only accelerates the production timeline but also reduces the consumption of auxiliary materials and solvents, contributing to a leaner and more cost-efficient manufacturing operation. By integrating this methodology into the supply chain, companies can achieve substantial cost savings while simultaneously enhancing their sustainability profiles, a factor that is increasingly important for meeting corporate social responsibility goals and regulatory compliance standards.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes a major cost driver from the bill of materials, as precious metals like palladium or ruthenium represent a significant portion of the expense in traditional cross-coupling reactions. Additionally, the avoidance of metal removal steps means that manufacturers do not need to invest in expensive scavenging resins or dedicate reactor time to purification processes designed to lower metal residues. This qualitative improvement in process economics allows for a more competitive pricing structure for the final pharmaceutical intermediates, providing a clear margin advantage in a crowded market. The use of commodity chemicals like potassium tert-butoxide and simple nitrogen ligands further stabilizes the supply chain against price fluctuations associated with specialized catalytic reagents.
- Enhanced Supply Chain Reliability: Relying on widely available inorganic bases and organic ligands mitigates the risk of supply disruptions that often plague the sourcing of complex transition metal complexes. Since the reagents required for this synthesis are produced on a massive industrial scale for various chemical applications, lead times are generally shorter and availability is more consistent compared to niche catalytic systems. This reliability ensures that production schedules can be maintained without the fear of delays caused by catalyst backorders or quality issues with sensitive metal reagents. Moreover, the robustness of the reaction conditions, which can tolerate air and moisture better than many sensitive metal-catalyzed systems, reduces the logistical burden of storing and handling reagents under strict inert conditions, simplifying warehouse management and transportation.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic heavy metals make this process inherently safer and easier to scale from kilogram to multi-ton production volumes. Regulatory bodies are increasingly scrutinizing the environmental impact of pharmaceutical manufacturing, and a metal-free process significantly reduces the burden of wastewater treatment and hazardous waste disposal. The lack of heavy metal contamination in the final product simplifies the regulatory filing process, as extensive data on metal clearance is not required, accelerating the time to market for new drug candidates. This alignment with green chemistry principles not only future-proofs the manufacturing process against tightening environmental regulations but also enhances the brand reputation of the supplier as a responsible and sustainable partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this base-promoted alkenylation technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational feasibility and strategic value of adopting this metal-free route for your specific synthesis needs. Understanding these details will help stakeholders make informed decisions about integrating this methodology into their existing production pipelines.
Q: Does this alkenylation method require expensive transition metal catalysts?
A: No, the method described in patent CN111253305A specifically avoids the use of transition metal catalysts. Instead, it utilizes a combination of inexpensive inorganic or organic bases and nitrogen/phosphine ligands to drive the reaction, significantly reducing raw material costs and eliminating heavy metal contamination risks.
Q: Can the selectivity between alkenylation and alkylation be controlled?
A: Yes, the reaction selectivity is highly tunable based on reaction conditions. Lower temperatures and specific ligand systems tend to favor alkenylation products (forming double bonds), while higher temperatures and extended reaction times can shift the pathway toward alkylation products (saturated chains), offering flexibility for different synthetic targets.
Q: What types of substrates are compatible with this synthesis route?
A: The protocol demonstrates broad substrate tolerance, accommodating various alkyl-substituted azaarenes such as quinolines, pyrazines, and pyridines. It also works with diverse alcohol coupling partners, including benzyl alcohols with electron-withdrawing or donating groups, making it suitable for synthesizing a wide range of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Styrylquinoline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free alkenylation technology described in patent CN111253305A and are fully equipped to leverage it for your custom synthesis projects. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-styrylquinoline derivatives meets the highest quality standards required by the global pharmaceutical industry. Our commitment to technical excellence allows us to navigate the complexities of C-H activation chemistry, delivering high-purity intermediates that accelerate your drug development timelines.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this innovative synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in base-promoted catalysis can add value to your portfolio of pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
