Advanced Synthesis of Entecavir Intermediate N4 via Safe Halohydrin Route for Commercial Scale-Up
Advanced Synthesis of Entecavir Intermediate N4 via Safe Halohydrin Route for Commercial Scale-Up
The global demand for effective antiviral therapies, particularly for the treatment of chronic Hepatitis B, continues to drive innovation in the synthesis of key nucleoside analogs. A pivotal development in this sector is disclosed in patent CN115850280A, which outlines a robust and safer preparation method for Entecavir intermediate N4 (CAS 142217-77-4). This intermediate serves as a critical building block in the manufacture of Entecavir, a potent guanosine analogue that inhibits the hepatitis B virus polymerase. The significance of this patent lies in its departure from traditional, hazardous synthetic pathways, offering a streamlined approach that enhances both operational safety and chemical efficiency. By replacing dangerous epoxidation steps with a controlled halohydrin formation strategy, this technology addresses long-standing challenges in the commercial production of high-purity antiviral intermediates. For pharmaceutical manufacturers and procurement specialists, understanding this shift is essential for securing a reliable supply chain that meets stringent regulatory standards while optimizing production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Entecavir intermediate N4 has relied on pathways described in earlier patents, such as CN102477036A, which involve the formation of an epoxy compound intermediate. This conventional route typically necessitates the use of tert-butyl peroxide for oxidation, a reagent known for its thermal instability and significant explosion hazards, thereby imposing severe safety constraints on industrial facilities. Furthermore, the subsequent ring-opening of the epoxide requires strong, pyrophoric bases like lithium hydride, n-butyl lithium, or sodium hydride under strictly anhydrous conditions. These reagents not only pose fire risks due to hydrogen gas generation but also demand specialized equipment and rigorous moisture control, complicating the manufacturing process. Additionally, the quenching of these strong bases generates substantial volumes of high-salinity wastewater, creating environmental compliance burdens and increasing waste disposal costs. The cumulative effect of these factors is a process that is operationally risky, environmentally taxing, and difficult to scale efficiently for mass market demand.
The Novel Approach
In stark contrast, the methodology presented in patent CN115850280A introduces a paradigm shift by bypassing the epoxide intermediate entirely in favor of a direct halohydrin strategy. This innovative route utilizes mild halogenating agents, such as N-bromosuccinimide (NBS) or elemental bromine, in a mixed solvent system of tetrahydrofuran and water. This allows for the stereoselective introduction of the hydroxyl and bromine functionalities under significantly milder conditions, typically ranging from -5°C to 0°C. Crucially, the final coupling step with 6-benzyloxyguanine can be performed using weaker, non-pyrophoric bases like sodium carbonate, eliminating the need for hazardous lithium or sodium hydrides. This transition not only drastically reduces the safety risks associated with reagent handling but also simplifies the downstream processing and purification stages. The result is a synthetic pathway that is inherently more stable, environmentally friendlier, and ideally suited for the rigorous demands of modern Good Manufacturing Practice (GMP) facilities.
Mechanistic Insights into Halohydrin Formation and N-Alkylation
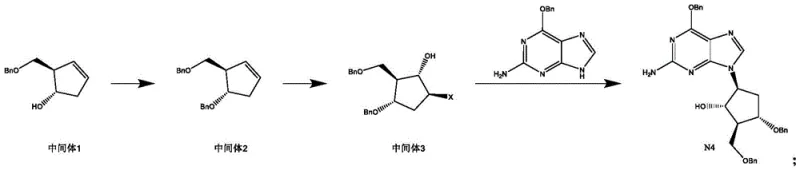
The core chemical innovation of this process resides in the stereoselective formation of the cyclopentane scaffold via electrophilic addition. In the second step of the synthesis, the alkene functionality of intermediate 2 reacts with a halogen source in the presence of water. Mechanistically, the halogen (bromine) acts as an electrophile, forming a cyclic bromonium ion intermediate across the double bond. The subsequent nucleophilic attack by water occurs from the opposite face (anti-addition), ensuring the correct trans-stereochemistry required for the biological activity of the final drug. This controlled addition is superior to the epoxide route because it avoids the strain energy and potential rearrangement issues associated with three-membered oxygen rings. The use of a polar aprotic solvent mixed with water facilitates this ionization while maintaining the solubility of the organic substrate, allowing for high conversion rates without the need for cryogenic temperatures below -20°C.
Following the construction of the functionalized cyclopentane ring, the final step involves a nucleophilic substitution reaction where the purine base displaces the bromine atom. This SN2-type reaction is facilitated by the activation of the guanine nitrogen through deprotonation by a base. The patent demonstrates that even mild bases like sodium carbonate are sufficient to drive this reaction to completion at elevated temperatures (60-120°C), likely due to the high reactivity of the secondary bromide in the specific steric environment of the cyclopentane ring. This mechanistic efficiency minimizes side reactions such as elimination or over-alkylation, which are common pitfalls in nucleoside synthesis. By carefully controlling the basicity and temperature, the process ensures that the chiral integrity of the molecule is preserved, resulting in a product with exceptional enantiomeric excess, a critical quality attribute for antiviral efficacy.
How to Synthesize Entecavir Intermediate N4 Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and purity, particularly during the halogenation and coupling phases. The process begins with the protection of the starting alcohol, followed by the critical low-temperature bromination which dictates the stereochemical outcome. Finally, the coupling with the guanine derivative must be managed to prevent degradation of the sensitive purine ring. While the general chemical transformations are straightforward, the specific workup procedures—such as the removal of succinimide byproducts or the efficient extraction of the final product from DMF—are vital for achieving pharmaceutical-grade quality. For detailed operational parameters, stoichiometry, and purification techniques, please refer to the standardized guide below.
- Protect the hydroxyl group of intermediate 1 using benzyl bromide and a base like triethylamine in anhydrous THF to form intermediate 2.
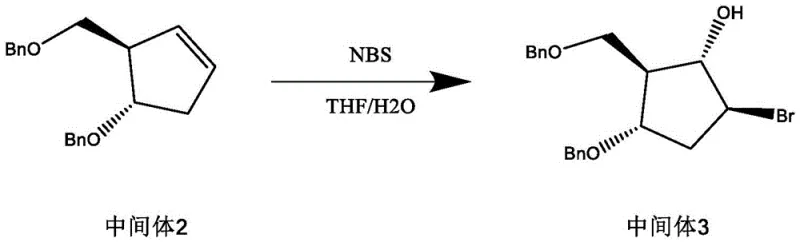
- React intermediate 2 with N-bromosuccinimide (NBS) or bromine in a THF/water mixture at low temperature (-5 to 0°C) to generate the bromohydrin intermediate 3.
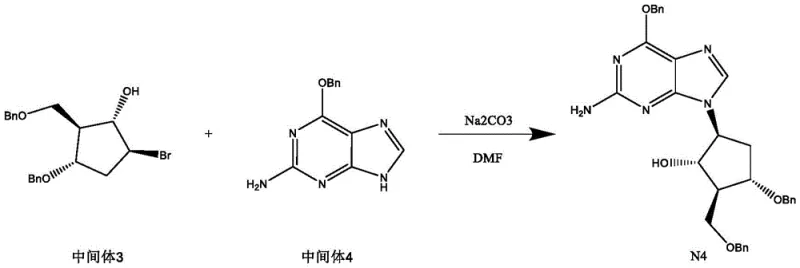
- Couple intermediate 3 with 6-benzyloxyguanine in DMF using sodium carbonate or sodium hydride as a base at elevated temperatures (60-120°C) to yield the final product N4.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond simple chemical yield. The primary advantage lies in the drastic simplification of the safety infrastructure required for production. By eliminating pyrophoric reagents like n-butyl lithium and unstable peroxides, facilities can reduce their insurance premiums, lower the cost of specialized safety equipment, and minimize the downtime associated with rigorous safety audits. Furthermore, the use of common, commodity chemicals such as NBS or liquid bromine ensures a stable and resilient supply of raw materials, shielding manufacturers from the volatility often seen in the market for exotic organometallic reagents. This stability translates directly into more predictable lead times and a reduced risk of production stoppages due to material shortages.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive and hazardous reagents. Traditional methods require costly strong bases and the infrastructure to handle them safely, whereas this new method utilizes inexpensive inorganic bases like sodium carbonate. Additionally, the avoidance of high-salinity wastewater generation significantly lowers waste treatment costs, a major hidden expense in pharmaceutical manufacturing. The simplified workup procedures also reduce solvent consumption and energy usage during purification, contributing to a leaner overall cost structure for the production of high-purity antiviral intermediates.
- Enhanced Supply Chain Reliability: From a logistics perspective, the robustness of this chemistry ensures consistent output. The tolerance for mild conditions means that the process is less susceptible to minor fluctuations in temperature or moisture, which often plague sensitive organometallic reactions. This reliability allows for tighter production scheduling and more accurate delivery commitments to downstream API manufacturers. Moreover, the use of standard solvents like THF and DMF, which are widely available globally, mitigates the risk of supply chain disruptions that can occur with niche or highly regulated chemical precursors.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this route is inherently designed for expansion. The absence of explosive peroxides and the use of aqueous workups make the transition from pilot plant to multi-ton commercial production much smoother. Environmentally, the reduction in hazardous waste and the ability to treat effluent more easily align with increasingly strict global environmental regulations. This compliance capability is a crucial asset for suppliers aiming to serve multinational pharmaceutical clients who enforce rigorous sustainability standards across their vendor networks.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the practical implementation and quality outcomes of this synthesis. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for process development scientists. Understanding these details is key to assessing the feasibility of integrating this intermediate into your existing manufacturing workflows.
Q: How does this new method improve safety compared to traditional epoxidation routes?
A: The novel method eliminates the use of hazardous oxidizing agents like tert-butyl peroxide and pyrophoric bases such as lithium hydride or n-butyl lithium. By utilizing mild halogenation reagents like NBS or liquid bromine in aqueous conditions, the process significantly reduces explosion risks and simplifies operational safety protocols.
Q: What are the purity specifications achievable with this halohydrin pathway?
A: Experimental data from the patent indicates that the final Entecavir intermediate N4 can be achieved with HPLC purity exceeding 99% and chiral purity greater than 99.5%. The mild reaction conditions help preserve stereochemical integrity, minimizing the formation of diastereomeric impurities common in harsh epoxide ring-opening reactions.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial scalability. It avoids the generation of large amounts of high-salinity wastewater associated with quenching strong bases and uses common solvents like THF and DMF. The elimination of strict anhydrous requirements for the halogenation step further facilitates easier handling and scale-up in standard reactor setups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Entecavir Intermediate N4 Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is critical for the future of antiviral drug manufacturing. As a leading CDMO and supplier in the fine chemical sector, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of nucleoside chemistry, ensuring that every batch of Entecavir intermediate N4 meets stringent purity specifications. With our rigorous QC labs and commitment to process excellence, we guarantee a supply of high-purity pharmaceutical intermediates that support the uninterrupted production of life-saving medications.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced technology for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project benefits from the highest standards of quality and efficiency in the industry.
